Revolutionizing Gossypol Production: A Scalable Asymmetric Synthesis Route for High-Purity Enantiomers
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access complex chiral natural products, particularly those with potent biological activities such as anticancer and antiviral properties. Patent CN111848374A introduces a groundbreaking preparation method for gossypol and its derivatives, specifically targeting the high-value single enantiomers (+)-gossypol and (-)-gossypol. Historically, obtaining these optically pure forms relied heavily on resolution techniques which are inherently inefficient. This new technology leverages a sophisticated asymmetric palladium-catalyzed coupling strategy to construct the sterically hindered biaryl axis directly. By utilizing cheap and readily available starting materials, the process achieves the target molecules in only 10 steps, representing a drastic improvement over previous methods that required more than 20 steps. This innovation not only enhances the feasibility of large-scale manufacturing but also ensures a consistent supply of high-purity intermediates essential for downstream drug development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the acquisition of single enantiomers of gossypol has been plagued by significant economic and operational inefficiencies inherent to chiral resolution strategies. The conventional approach typically involves synthesizing the racemic mixture first, followed by separation using chiral resolving agents such as amino acids to form diastereomeric Schiff bases. This process is fundamentally limited by a maximum theoretical yield of 50% for the desired enantiomer, effectively wasting half of the synthesized material. Furthermore, the requirement for stoichiometric amounts of expensive chiral auxiliaries drastically increases the raw material costs and complicates the purification workflow. The additional steps required to introduce and subsequently remove these resolving agents extend the production timeline and increase the generation of chemical waste. For procurement managers and supply chain heads, these factors translate into higher costs of goods sold (COGS) and potential bottlenecks in securing sufficient quantities of active pharmaceutical ingredients (APIs) for clinical or commercial use.
The Novel Approach
In stark contrast, the methodology disclosed in this patent employs a direct asymmetric synthesis route that constructs the chiral axis during the carbon-carbon bond-forming event. By utilizing a specialized chiral phosphine ligand in conjunction with a palladium catalyst, the process induces axial chirality during the coupling of two naphthaldehyde units. This catalytic asymmetric approach bypasses the need for resolution entirely, theoretically allowing for yields approaching 100% of the desired enantiomer depending on the catalyst efficiency. The route is remarkably concise, condensing the synthesis into just 10 linear steps from basic commodity chemicals. This streamlining eliminates the redundant protection and deprotection cycles often seen in older total synthesis routes. For R&D directors, this represents a robust platform technology that simplifies the impurity profile and enhances the overall process mass intensity (PMI), making it a superior choice for modern green chemistry initiatives and cost-effective manufacturing.
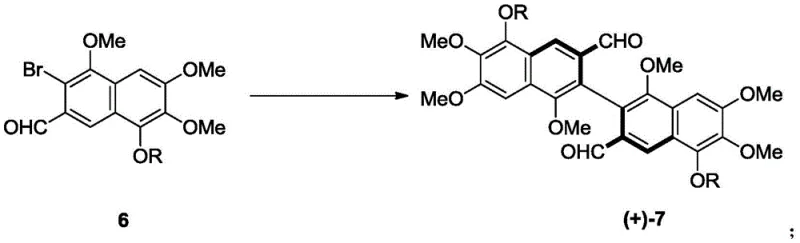
Mechanistic Insights into Pd-Catalyzed Asymmetric Biaryl Coupling
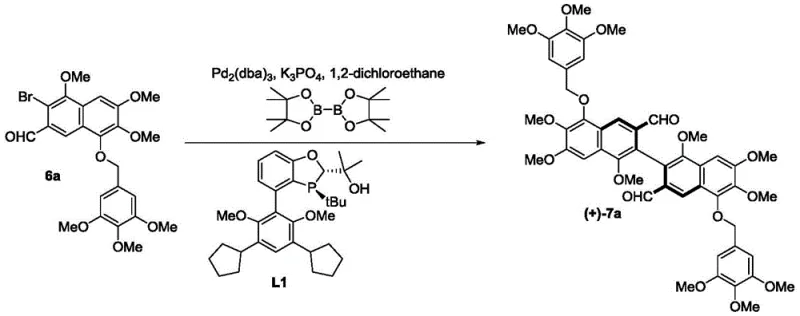
The cornerstone of this synthetic breakthrough is the enantioselective Suzuki-Miyaura type coupling reaction that joins two functionalized naphthalene halves to form the sterically crowded biaryl backbone of gossypol. The reaction utilizes a diboron reagent to activate one naphthaldehyde partner, which then couples with a brominated naphthaldehyde counterpart under the influence of a palladium catalyst and a chiral ligand such as L1 or L2. The mechanism likely proceeds through the standard catalytic cycle involving oxidative addition of the aryl bromide to the Pd(0) species, transmetallation with the organoboron compound, and finally reductive elimination to forge the C-C bond. The critical element is the chiral environment created by the ligand, which differentiates the transition states leading to the (R) or (S) atropisomers. Due to the ortho-tetrasubstitution pattern on the naphthalene rings, the resulting biaryl bond possesses a high rotational energy barrier, locking the molecule into a stable chiral conformation. This precise control over stereochemistry is vital, as the biological activities of gossypol enantiomers differ significantly, with specific isomers showing distinct anticancer or contraceptive profiles.
Furthermore, the reaction conditions are optimized to balance reactivity and selectivity, typically employing mild temperatures around 60°C in solvents like 1,2-dichloroethane. The use of inorganic bases such as potassium phosphate ensures efficient activation of the boron species without degrading the sensitive aldehyde functionalities present on the substrate. Following the coupling, the resulting intermediate (Compound 7) exhibits enantiomeric excess (ee) values up to 83%, which serves as an excellent foundation for further purification. Simple recrystallization techniques can elevate this purity to greater than 99% ee, ensuring that the final API meets the stringent regulatory requirements for chiral drugs. This mechanistic robustness allows for the reliable production of both (+)- and (-)-enantiomers simply by switching the chirality of the ligand used, providing unparalleled flexibility for manufacturing diverse gossypol derivatives.
How to Synthesize Gossypol Enantiomers Efficiently
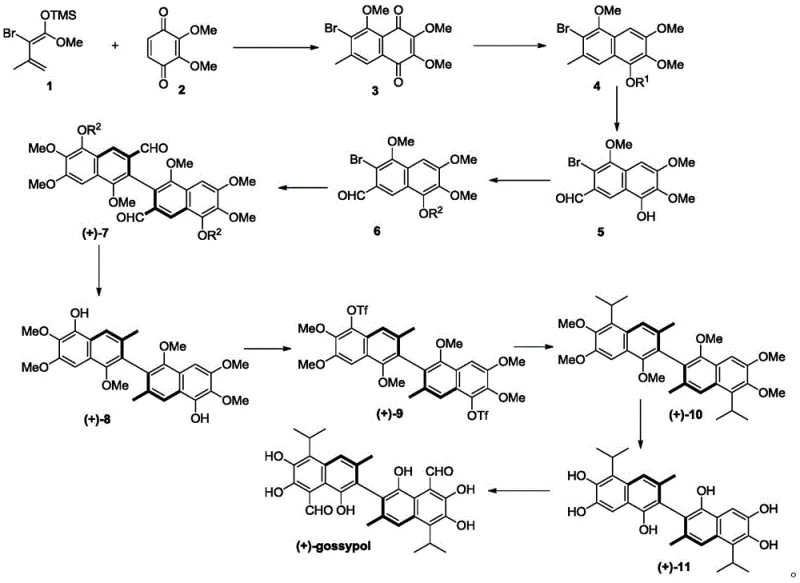
The synthesis of high-purity gossypol enantiomers via this patented route involves a logical sequence of transformations designed to maximize yield and stereocontrol. The process begins with the construction of the naphthalene core through a Diels-Alder cycloaddition, followed by functional group manipulations to install the necessary aldehyde and leaving group handles. The pivotal step is the asymmetric coupling described previously, which sets the axial chirality. Subsequent steps involve catalytic hydrogenation to reduce aldehyde groups to methyls, triflation of phenolic hydroxyls to enable further cross-coupling for side-chain installation, and finally, demethylation and formylation to reveal the native polyphenolic structure of gossypol. Each step has been optimized for scalability, using reagents and conditions that are compatible with large-scale reactor operations. For detailed operational parameters, safety data, and specific stoichiometric ratios required for GMP manufacturing, please refer to the standardized synthesis guide below.
- Construct the naphthalene core via Diels-Alder reaction of conjugated diene and benzoquinone derivatives, followed by selective reduction and acylation.
- Perform bromination and oxidation to generate the key naphthaldehyde intermediate, followed by protective group alkylation.
- Execute the critical enantioselective biaryl coupling using a chiral phosphine ligand and palladium catalyst to establish axial chirality.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthesis route offers transformative benefits regarding cost structure and supply reliability. The most immediate impact is the drastic reduction in the number of synthetic steps, dropping from over 20 in legacy methods to just 10. This compression of the supply chain directly correlates to substantial cost savings in labor, utilities, and equipment usage, as fewer unit operations are required to reach the final product. Additionally, the elimination of chiral resolution removes the need for expensive stoichiometric resolving agents and the associated loss of 50% of the material, effectively doubling the theoretical output from the same amount of starting raw materials. This efficiency gain significantly lowers the cost of goods sold (COGS), allowing for more competitive pricing in the global market for pharmaceutical intermediates. The reliance on commercially available starting materials further mitigates supply risk, ensuring that production schedules are not disrupted by the scarcity of exotic precursors.
From an environmental and scalability perspective, this process aligns perfectly with modern sustainable manufacturing goals. The use of catalytic amounts of palladium rather than stoichiometric chiral auxiliaries reduces the heavy metal load in waste streams, simplifying effluent treatment and reducing environmental compliance costs. The robustness of the palladium-catalyzed coupling allows for straightforward scale-up from kilogram to multi-ton quantities without significant re-optimization, ensuring a continuous and reliable supply of high-purity gossypol intermediates. This scalability is crucial for meeting the demands of clinical trials and eventual commercial launch of gossypol-based therapeutics. By partnering with a manufacturer capable of executing this advanced chemistry, companies can secure a long-term supply of critical intermediates while minimizing their carbon footprint and operational expenditures associated with complex chemical synthesis.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this gossypol synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is essential for R&D teams evaluating the feasibility of technology transfer and for procurement specialists assessing the quality and consistency of the supply chain. The answers reflect the current state of the art as defined by the patent holder, ensuring that all stakeholders have accurate information for decision-making.
Q: What is the primary advantage of this new gossypol synthesis method?
A: The primary advantage is the significant reduction in synthetic steps (10 steps vs. >20 steps in prior art) and the direct construction of axial chirality via asymmetric catalysis, eliminating the need for inefficient chemical resolution.
Q: Can this process be scaled for industrial API production?
A: Yes, the patent explicitly states the method is simple to operate and suitable for industrial production, utilizing commercially available raw materials and standard palladium catalytic conditions.
Q: What level of optical purity can be achieved?
A: The asymmetric coupling step yields intermediates with enantiomeric excess (ee) values up to 83%, which can be further enriched to greater than 99% ee through simple recrystallization processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gossypol Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis and contract development, possessing the technical expertise to bring complex routes like this gossypol synthesis from the laboratory bench to commercial reality. Our team of experienced chemists is well-versed in handling sensitive palladium-catalyzed reactions and managing the intricacies of chiral intermediate production. We offer extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch, adhering to international regulatory standards. Whether you require process optimization, impurity profiling, or full-scale manufacturing, our CDMO services are tailored to accelerate your drug development timeline.
We invite you to collaborate with us to leverage this innovative synthesis technology for your gossypol-based projects. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this streamlined route. We encourage potential partners to contact us to obtain specific COA data for our reference standards and to discuss route feasibility assessments tailored to your specific volume requirements. Let us help you secure a reliable, cost-effective, and high-quality supply of gossypol intermediates, empowering your research and commercial success in the competitive pharmaceutical landscape.
