Scalable Synthesis of 1,1'-Deoxygossypol: A Cost-Effective Route for Anti-HIV Intermediates
Scalable Synthesis of 1,1'-Deoxygossypol: A Cost-Effective Route for Anti-HIV Intermediates
The pharmaceutical industry continuously seeks robust and economically viable pathways for synthesizing complex bioactive molecules, particularly those with potent antiviral properties. A significant breakthrough in this domain is detailed in patent CN112679319A, which discloses a highly efficient method for the synthesis of 1,1'-deoxygossypol, a compound known for its promising anti-HIV activity. This novel approach fundamentally shifts the paradigm from relying on scarce, expensive starting materials to utilizing gossypol acetate, an abundant and cost-effective derivative of cottonseed. By streamlining the synthetic sequence to just eight steps and achieving a remarkable total yield of 45%, this technology addresses critical bottlenecks in the supply chain of high-value pharmaceutical intermediates. The methodology not only enhances economic feasibility but also significantly improves operational safety by eliminating the need for hazardous cryogenic reagents, thereby offering a sustainable solution for large-scale manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of 1,1'-deoxygossypol was plagued by severe economic and technical inefficiencies that hindered its widespread adoption in drug development. The traditional route, as referenced in existing literature, relied heavily on 1-bromo-2-isopropyl-3,4-dimethoxybenzene as the primary starting material, a chemical that is not only scarce but also exorbitantly priced, creating a prohibitive barrier for commercial production. Furthermore, the conventional synthetic pathway was excessively lengthy, requiring nine distinct reaction steps that resulted in a dismal total yield of merely 16.9%, leading to substantial material waste and inflated production costs. From a safety and operational perspective, the legacy method demanded the use of highly reactive and dangerous reagents such as n-butyllithium, necessitating strict temperature control at minus 78 degrees Celsius, which imposes heavy energy burdens and complex engineering requirements on manufacturing facilities.
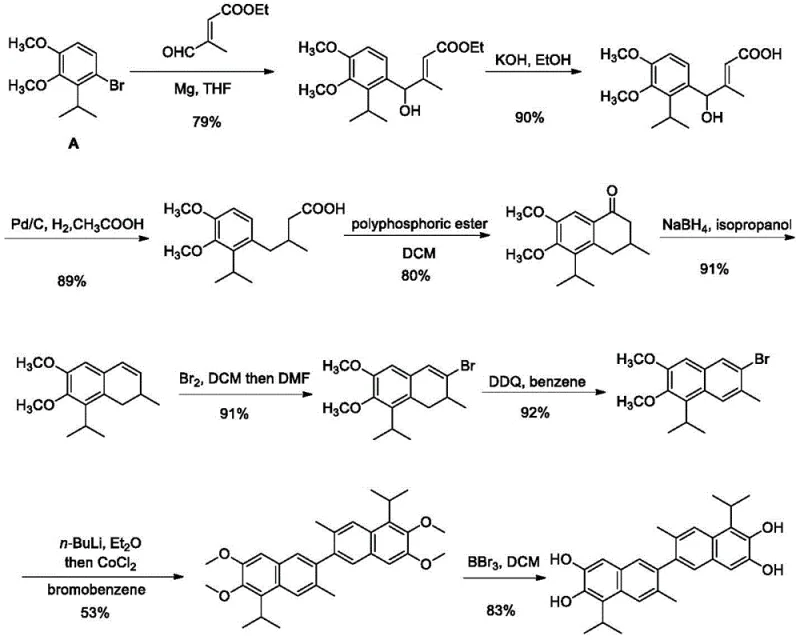
The Novel Approach
In stark contrast to the cumbersome legacy processes, the disclosed invention introduces a streamlined and economically superior strategy that leverages the natural abundance of gossypol derivatives. By initiating the synthesis with gossypol acetate, a readily available byproduct of the cottonseed oil industry, the new method drastically reduces raw material costs while simplifying the overall process flow to eight manageable steps. This optimized route achieves a total yield of 45%, representing a nearly threefold improvement over previous methods, which translates directly into higher throughput and reduced waste generation. The reaction conditions are notably mild, operating primarily at ambient or moderately elevated temperatures without the need for extreme cryogenic cooling, thus enhancing the safety profile and making the process inherently more suitable for industrial scale-up and continuous manufacturing environments.
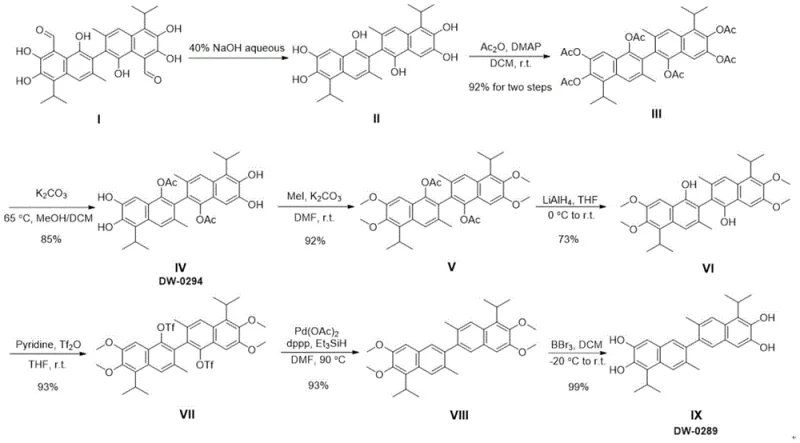
Mechanistic Insights into Selective Protection and Pd-Catalyzed Reduction
The core chemical ingenuity of this synthesis lies in its sophisticated manipulation of phenolic hydroxyl groups through a series of selective protection and deprotection strategies, culminating in a pivotal palladium-catalyzed reduction. The process begins with the deformylation of gossypol acetate using aqueous sodium hydroxide, effectively removing aldehyde groups to generate apogossypol, which is then acetylated to protect specific positions. Subsequent steps involve the precise differentiation of hydroxyl environments; for instance, selective deacetylation using potassium carbonate allows for the exposure of specific phenolic sites for methylation, while others remain protected as acetates. This orthogonal protection strategy is crucial for directing the regioselectivity of subsequent transformations, ensuring that the final molecular architecture is constructed with high fidelity and minimal formation of structural isomers or impurities.
A critical mechanistic highlight of this route is the reductive deoxygenation step, where the protected intermediate is subjected to palladium catalysis. In this transformation, the intermediate bearing triflate groups is treated with a palladium catalyst system, specifically palladium acetate and dppp, in the presence of triethylsilane as a hydride source. This catalytic cycle facilitates the cleavage of the carbon-oxygen bond at the 1 and 1' positions, effectively removing the oxygen functionality to yield the deoxy precursor. The use of a homogeneous palladium catalyst ensures high conversion rates under relatively mild thermal conditions, avoiding the harsh reducing agents often required in classical deoxygenation methods. This step is instrumental in establishing the core binaphthalene skeleton of 1,1'-deoxygossypol with the correct oxidation state, setting the stage for the final global demethylation to reveal the target polyphenol structure.
How to Synthesize 1,1'-Deoxygossypol Efficiently
The synthesis of this high-value intermediate requires precise control over reaction parameters to maximize yield and purity, particularly during the selective functionalization stages. The process integrates standard organic transformations such as acetylation, methylation, and triflation, but their sequence is carefully orchestrated to exploit the differing reactivities of the phenolic hydroxyl groups on the binaphthalene core. Operators must pay close attention to stoichiometry and temperature during the triflation and reduction steps to prevent over-reaction or catalyst deactivation. For a comprehensive understanding of the specific operational parameters, reagent ratios, and workup procedures required to replicate this high-yielding process, please refer to the standardized synthesis guide provided below.
- Deformylation of gossypol acetate using aqueous NaOH to obtain apogossypol, followed by acetylation.
- Selective deacetylation and methylation to protect specific phenolic hydroxyl groups.
- Triflation of remaining hydroxyls followed by Pd-catalyzed reductive deoxygenation and final demethylation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis method offers transformative benefits that extend far beyond simple chemical yield improvements. The most immediate impact is seen in the drastic reduction of raw material costs, as the process substitutes an expensive, specialty bromobenzene derivative with gossypol acetate, a commodity chemical derived from the massive global cottonseed oil industry. This shift not only lowers the direct cost of goods sold but also insulates the supply chain from the volatility associated with niche synthetic starting materials, ensuring a more stable and predictable sourcing environment. Furthermore, the elimination of cryogenic processing requirements significantly reduces energy consumption and capital expenditure on specialized low-temperature reactors, contributing to a leaner and more agile manufacturing infrastructure.
- Cost Reduction in Manufacturing: The transition to gossypol acetate as a feedstock represents a fundamental economic advantage, as this material is sourced from agricultural byproducts rather than multi-step synthetic sequences. By avoiding the use of precious metal catalysts in early stages and eliminating the need for expensive organolithium reagents, the overall reagent cost profile is substantially optimized. Additionally, the higher total yield means that less raw material is required to produce the same amount of final product, effectively amplifying the cost savings across the entire production volume and improving the gross margin potential for the final API.
- Enhanced Supply Chain Reliability: Relying on cottonseed-derived starting materials diversifies the supply base away from petrochemical-dependent intermediates, which are often subject to geopolitical and market fluctuations. The simplified synthetic route, with fewer unit operations and milder conditions, reduces the risk of batch failures and production delays, thereby enhancing the reliability of delivery schedules. This robustness is critical for maintaining continuity in the supply of essential antiviral intermediates, ensuring that downstream drug manufacturers can meet their own production targets without interruption.
- Scalability and Environmental Compliance: The process design inherently supports green chemistry principles by minimizing the use of hazardous reagents and reducing waste generation through higher efficiency. The absence of extreme低温 conditions and the use of common solvents like DCM, THF, and DMF facilitate easier waste treatment and solvent recovery, aligning with stringent environmental regulations. This environmental compatibility, combined with the operational simplicity of the reaction steps, makes the technology highly scalable from pilot plant to multi-ton commercial production, allowing for rapid capacity expansion to meet market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 1,1'-deoxygossypol synthesized via this novel pathway. These insights are derived directly from the patent specifications and are intended to clarify the feasibility and advantages of this method for potential partners and stakeholders. Understanding these details is essential for evaluating the integration of this intermediate into broader pharmaceutical development pipelines.
Q: What is the primary advantage of this new synthesis route over conventional methods?
A: The new route utilizes gossypol acetate, a cheap and abundant cottonseed byproduct, instead of expensive 1-bromo-2-isopropyl-3,4-dimethoxybenzene. It improves total yield from 16.9% to 45% and eliminates hazardous cryogenic conditions.
Q: How does the process ensure high purity for pharmaceutical applications?
A: The method employs robust purification techniques including column chromatography after key steps and uses selective protection strategies that minimize side reactions, ensuring stringent purity specifications suitable for API intermediates.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the process avoids extreme conditions like -78°C and uses common reagents like NaOH, Ac2O, and BBr3. The mild reaction conditions and simple post-processing make it highly adaptable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,1'-Deoxygossypol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable and high-quality supply of complex pharmaceutical intermediates like 1,1'-deoxygossypol. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive materials that meet stringent purity specifications. Our rigorous QC labs employ advanced analytical techniques to verify the identity and purity of every batch, guaranteeing that the 1,1'-deoxygossypol we supply is fully compliant with the demanding standards required for anti-HIV drug development and other therapeutic applications.
We invite you to collaborate with us to leverage this innovative synthesis technology for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this route can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us support your journey from research to commercial success with our unparalleled expertise in fine chemical manufacturing.
