Revolutionizing Agomelatine Synthesis: A Scalable One-Pot Hydrogenation Strategy for Global Pharma Supply Chains
Revolutionizing Agomelatine Synthesis: A Scalable One-Pot Hydrogenation Strategy for Global Pharma Supply Chains
The pharmaceutical industry constantly seeks robust, scalable, and cost-effective pathways for the production of active pharmaceutical ingredients (APIs) and their key intermediates. Patent CN102942501A introduces a transformative industrial production method for preparing Agomelatine, a potent melatonin receptor agonist and serotonin 2C receptor antagonist used in the treatment of depression and sleep disorders. This innovation addresses critical bottlenecks in traditional synthesis by utilizing a novel one-pot catalytic hydrogenation strategy that simultaneously reduces a nitrile group and performs an acylation reaction. By shifting from multi-step, low-yield processes to a streamlined hydrogenation protocol, this technology offers a compelling value proposition for manufacturers aiming to optimize their supply chains for high-purity antidepressant intermediates. The method leverages readily available raw materials and standard reactor configurations to achieve exceptional purity profiles, marking a significant advancement in the commercial manufacturing of complex heterocyclic amines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Agomelatine has been plagued by inefficiencies that hinder large-scale commercial viability. Early methodologies, such as those described in patent EP0447285, relied on 7-methoxy-alpha-tetralone as a starting material, necessitating an arduous eight-step synthetic sequence with an average yield lower than 30%. Such low efficiency not only drives up the cost of goods sold (COGS) but also generates substantial chemical waste, creating environmental compliance burdens. Other approaches, like the one detailed in the Chinese Journal of Pharmaceuticals (2008), attempted to simplify the route using tetrahydrofuran as a solvent but suffered from severe selectivity issues, generating irreversible dimer impurities at levels of 10% to 15% during the initial reaction stages. Furthermore, alternative high-pressure methods described in patent CN101643433A required specialized continuous-feed hydrogenation equipment operating at 30 bar, which presents significant capital expenditure barriers and operational complexity for standard manufacturing facilities unable to support such niche infrastructure.
The Novel Approach
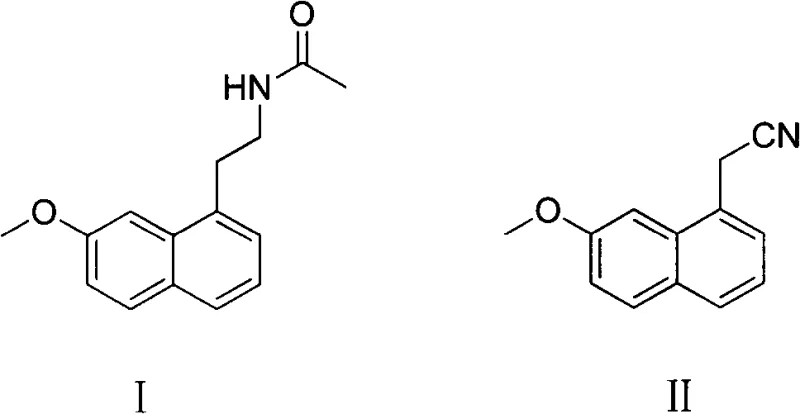
In stark contrast to these legacy techniques, the method disclosed in CN102942501A utilizes (7-methoxy-1-naphthyl) acetonitrile as a direct precursor, subjecting it to a concurrent hydrogenation and acylation process within a standard autoclave. This approach fundamentally alters the reaction landscape by maintaining a faintly alkaline environment through the addition of acid-binding agents like triethylamine or pyridine, which preserves catalyst activity and drives the reaction to completion with remarkable speed. The result is a drastic reduction in reaction time to merely 1-2 hours and a suppression of dimer impurities to less than 0.1% in the crude product, a feat previously unattainable without complex purification steps. By operating at moderate pressures of 0.5-3MPa and temperatures between 35-60°C, this process eliminates the need for exotic high-pressure equipment, thereby lowering the barrier to entry for contract development and manufacturing organizations (CDMOs) seeking to offer cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Raney-Ni Catalyzed Reductive Acylation
The core of this technological breakthrough lies in the sophisticated interplay between the Raney-Nickel catalyst, the hydrogen gas, and the acylating agent within an anhydrous solvent system. Mechanistically, the process initiates with the adsorption of hydrogen onto the Raney-Ni surface, followed by the reduction of the nitrile group (-CN) on the (7-methoxy-1-naphthyl) acetonitrile substrate to a primary amine intermediate. Crucially, the presence of the acylating reagent, such as acetic anhydride or acetyl chloride, ensures that this highly reactive amine is immediately captured to form the stable acetamide bond found in Agomelatine. This tandem reaction sequence prevents the accumulation of free amine species, which are often prone to secondary reactions that lead to the formation of unwanted dimers or oligomers. The careful selection of the acid-binding agent serves a dual purpose: it neutralizes the acid byproducts generated during acylation (such as HCl or acetic acid) and maintains the pH balance necessary to prevent the poisoning of the nickel catalyst, ensuring sustained catalytic turnover throughout the batch cycle.
From an impurity control perspective, the suppression of dimer formation is attributed to the kinetic favorability of the acylation step over the condensation of amine intermediates. In conventional methods where reduction and acylation might be separated or poorly synchronized, the transient amine can react with unreacted nitrile or other amine molecules to form heavy ends. However, in this optimized system, the high local concentration of the acylating agent and the controlled alkalinity create a thermodynamic sink that directs the reaction flux exclusively toward the target mono-acetylated product. This precise control over the reaction pathway results in a crude product purity of 98-99% as measured by HPLC, significantly simplifying downstream processing. The ability to achieve such high selectivity without cryogenic conditions or stoichiometric amounts of expensive reagents underscores the robustness of this catalytic cycle for the commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Agomelatine Efficiently
The implementation of this synthesis route requires strict adherence to anhydrous conditions and precise control over hydrogen pressure to maximize safety and yield. Operators must ensure that the reactor is thoroughly purged of oxygen prior to hydrogen introduction to prevent catalyst deactivation and potential safety hazards associated with pyrophoric Raney-Ni. The detailed standardized synthetic steps, including specific molar ratios of the acid-binding agent and catalyst loading, are critical for reproducing the high purity and yield metrics reported in the patent data.
- Mix (7-methoxy-1-naphthyl) acetonitrile with an acylating reagent (e.g., acetic anhydride) and an acid-binding agent in an anhydrous solvent like THF.
- Add Raney-Ni catalyst to the mixture and stir while introducing industrial hydrogen gas, ensuring air is displaced three times.
- Maintain reaction pressure between 0.5-3MPa and temperature between 35-60°C for 1-2 hours to complete the reduction and acylation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this hydrogenation methodology translates directly into enhanced operational resilience and significant cost optimization opportunities. By consolidating what were previously multiple reaction and isolation steps into a single unit operation, manufacturers can drastically reduce solvent consumption, energy usage, and labor hours associated with batch turnover. The elimination of complex purification protocols required to remove high levels of dimer impurities further streamlines the production timeline, allowing for faster throughput and reduced lead time for high-purity pharmaceutical intermediates. Moreover, the reliance on commodity chemicals like Raney-Ni, which can be reused multiple times with negligible cost impact, removes the dependency on scarce or volatile precious metal catalyst markets, stabilizing the raw material cost structure over the long term.
- Cost Reduction in Manufacturing: The process achieves substantial cost savings by utilizing inexpensive Raney-Ni catalysts that can be mechanically recovered and reused over 10 to 12 cycles, effectively rendering the catalyst cost negligible in the overall bill of materials. Additionally, the high crude purity of 98-99% minimizes the loss of valuable material during recrystallization and washing steps, maximizing the mass balance and overall yield to approximately 95%, which directly improves the margin profile for bulk drug production.
- Enhanced Supply Chain Reliability: By removing the requirement for specialized continuous-feed high-pressure equipment, this method allows production to be executed in standard industrial autoclaves available at most chemical manufacturing sites. This flexibility expands the pool of qualified suppliers capable of producing Agomelatine, mitigating the risk of supply disruptions caused by equipment bottlenecks or the limited availability of niche processing facilities, thereby ensuring a more robust and continuous supply for downstream formulation partners.
- Scalability and Environmental Compliance: The one-pot nature of the reaction significantly reduces the generation of three-waste pollutants (waste water, waste gas, and solid waste) compared to multi-step alternatives, simplifying environmental management and lowering disposal costs. The process is inherently safer due to the moderate pressure conditions (0.5-3MPa) and the absence of highly reactive intermediates that require isolation, making it ideally suited for safe scaling from pilot plant quantities to multi-ton annual commercial production volumes without compromising operator safety or regulatory compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this hydrogenation technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on yield expectations, equipment requirements, and impurity profiles. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into existing manufacturing portfolios.
Q: How does this new hydrogenation method improve upon previous Agomelatine synthesis routes?
A: Unlike prior art which suffered from low yields (<30%) or required complex multi-step processes, this method achieves a crude purity of 98-99% and a refined yield of 95% in a single pot, significantly reducing waste and processing time.
Q: What is the primary advantage regarding impurity control in this process?
A: The process effectively suppresses the formation of dimer impurities, keeping them below 0.1% in the crude product, whereas older methods often generated 10-15% dimer content that was difficult to remove.
Q: Does this method require specialized high-pressure equipment?
A: No, the reaction operates at moderate pressures (0.5-3MPa) using standard industrial autoclaves, eliminating the need for expensive continuous-feed high-pressure hydrogenation equipment required by other patented methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Agomelatine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the rigorous demands of the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the hydrogenation process described in CN102942501A can be seamlessly transferred from the laboratory to full-scale manufacturing. We are committed to delivering products with stringent purity specifications and maintaining rigorous QC labs to verify that every batch meets the highest standards for identity, potency, and impurity control, providing our partners with the confidence they need to advance their drug development pipelines.
We invite you to engage with our technical procurement team to discuss how this efficient synthesis route can be tailored to your specific volume requirements and quality targets. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this one-pot hydrogenation method. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability as a trusted partner in the reliable supply of high-value antidepressant intermediates.
