Advanced Synthesis Of Phenylimidazolidine Derivatives For Commercial Pharmaceutical Applications
Advanced Synthesis Of Phenylimidazolidine Derivatives For Commercial Pharmaceutical Applications
The pharmaceutical and agrochemical industries continuously demand high-purity intermediates that possess complex substitution patterns, particularly those involving trifluoromethyl groups which enhance metabolic stability and lipophilicity. Patent CN1207732A introduces a robust and versatile preparation method for phenylimidazolidine derivatives, specifically targeting structures defined by Formula (I). This technology addresses critical challenges in constructing the bond between the substituted aniline moiety and the hydantoin ring system. The core innovation lies in a multi-step sequence that begins with the selective functionalization of the aniline precursor, followed by a copper-catalyzed coupling reaction. 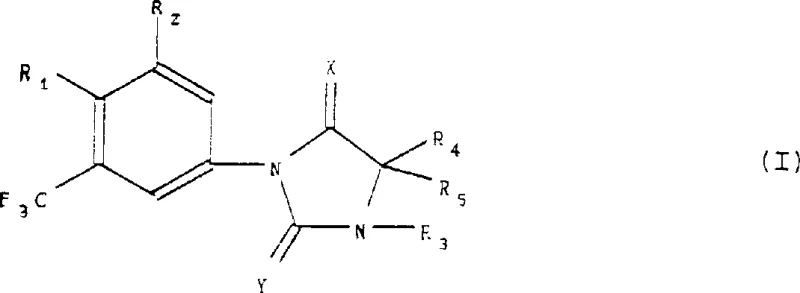 As illustrated in the general structure, the versatility of this method allows for the introduction of diverse functional groups such as cyano, halo, and amino substituents, making these compounds invaluable building blocks for reliable pharmaceutical intermediate supplier networks seeking to diversify their API pipelines.
As illustrated in the general structure, the versatility of this method allows for the introduction of diverse functional groups such as cyano, halo, and amino substituents, making these compounds invaluable building blocks for reliable pharmaceutical intermediate supplier networks seeking to diversify their API pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for attaching hydantoin rings to electron-deficient aromatic systems often suffer from harsh reaction conditions and poor regioselectivity. Conventional nucleophilic aromatic substitution typically requires extremely high temperatures and strong bases, which can lead to the degradation of sensitive functional groups like esters or nitriles present on the aromatic ring. Furthermore, achieving selective halogenation on the aniline precursor prior to coupling is notoriously difficult; standard brominating agents often result in over-halogenation or attack at the ortho-position rather than the desired para-position relative to the amino group. These inefficiencies create significant impurities that are costly and technically challenging to remove during downstream purification, ultimately impacting the overall yield and commercial viability of the process for cost reduction in API intermediate manufacturing.
The Novel Approach
The methodology disclosed in the patent overcomes these hurdles through a refined two-stage strategy focusing on mild activation and efficient coupling. A key breakthrough is the use of dibromo-dimethylhydantoin as a specialized brominating agent. 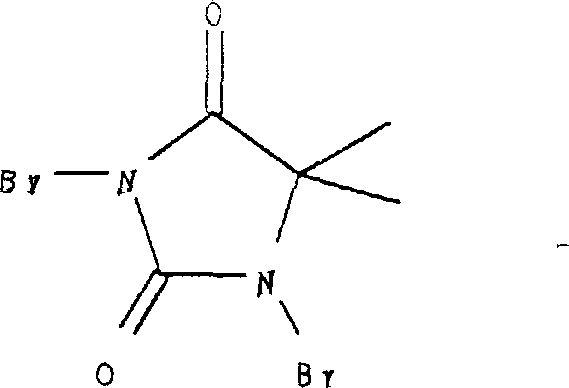 Unlike elemental bromine or N-bromosuccinimide which can be aggressive, this reagent facilitates highly selective bromination at the position para to the amino group when conducted at low temperatures around 0°C in polar aprotic solvents like N,N-dimethylacetamide. Following this activation, the coupling with the hydantoin derivative is performed using cuprous oxide as a catalyst at elevated temperatures (155°C to 165°C). This specific combination of reagents and conditions ensures high conversion rates while maintaining the integrity of the trifluoromethyl group, providing a scalable pathway for the commercial scale-up of complex pharmaceutical intermediates.
Unlike elemental bromine or N-bromosuccinimide which can be aggressive, this reagent facilitates highly selective bromination at the position para to the amino group when conducted at low temperatures around 0°C in polar aprotic solvents like N,N-dimethylacetamide. Following this activation, the coupling with the hydantoin derivative is performed using cuprous oxide as a catalyst at elevated temperatures (155°C to 165°C). This specific combination of reagents and conditions ensures high conversion rates while maintaining the integrity of the trifluoromethyl group, providing a scalable pathway for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Copper-Catalyzed Aryl-Hydantoin Coupling
The heart of this synthesis lies in the copper-mediated formation of the carbon-nitrogen bond between the aryl halide and the hydantoin nitrogen. Mechanistically, this likely proceeds via an oxidative addition-reductive elimination cycle typical of Ullmann-type couplings, although the specific involvement of cuprous oxide suggests a surface-mediated or radical mechanism may also play a role under these thermal conditions. The presence of the electron-withdrawing trifluoromethyl group on the aromatic ring actually facilitates the initial oxidative addition step by rendering the carbon-halogen bond more susceptible to nucleophilic attack or metal insertion. The solvent choice, specifically N,N-dimethylacetamide or dimethylformamide, is critical as it stabilizes the polar transition states and solubilizes the inorganic copper salts, ensuring homogeneous reaction kinetics. This mechanistic understanding allows process chemists to fine-tune the stoichiometry, typically using a slight excess of the hydantoin derivative to drive the equilibrium towards the desired product.
Beyond the primary coupling, the patent details sophisticated mechanisms for further functionalizing the aromatic core, significantly expanding the utility of these intermediates. For instance, the amino group generated or preserved in the intermediate stages can undergo diazotization to form diazonium salts, which serve as versatile handles for Sandmeyer reactions. 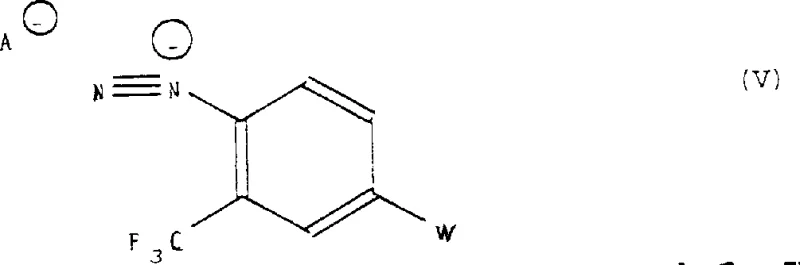 This allows for the precise replacement of the amino group with cyano, iodo, or fluoro substituents, enabling the synthesis of diverse analogues like 4-(4,4-dimethyl-2,5-dioxo-1-imidazolidyl)-2-(trifluoromethyl)benzonitrile. Additionally, the patent describes the use of Lawesson's reagent to convert carbonyl oxygens to sulfur atoms.
This allows for the precise replacement of the amino group with cyano, iodo, or fluoro substituents, enabling the synthesis of diverse analogues like 4-(4,4-dimethyl-2,5-dioxo-1-imidazolidyl)-2-(trifluoromethyl)benzonitrile. Additionally, the patent describes the use of Lawesson's reagent to convert carbonyl oxygens to sulfur atoms. 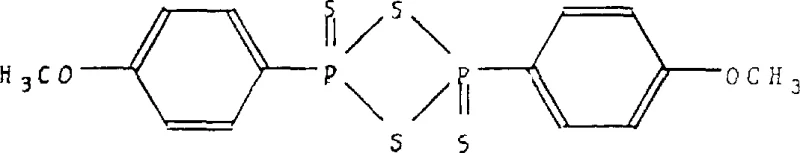 This thionation capability is crucial for generating thio-hydantoin derivatives, which often exhibit different biological activity profiles and metabolic stability compared to their oxo-counterparts, thereby offering medicinal chemists a powerful tool for structure-activity relationship (SAR) studies.
This thionation capability is crucial for generating thio-hydantoin derivatives, which often exhibit different biological activity profiles and metabolic stability compared to their oxo-counterparts, thereby offering medicinal chemists a powerful tool for structure-activity relationship (SAR) studies.
How to Synthesize 3-[4-amino-3-(trifluoromethyl)phenyl]-5,5-dimethyl-2,4-imidazolidinedione Efficiently
The synthesis of this specific high-value intermediate exemplifies the efficiency of the patented process, combining selectivity with operational simplicity. The procedure begins with the dissolution of ortho-5-trifluoromethylaniline in N,N-dimethylacetamide, followed by cooling to 0°C to control the exothermic bromination. The detailed standardized synthesis steps are provided in the guide below, outlining the precise addition rates and workup procedures required to achieve the reported yields of over 150g on a laboratory scale.
- Dissolve ortho-5-trifluoromethylaniline in N,N-dimethylacetamide and cool to 0°C. Add dibromo-dimethylhydantoin solution slowly to achieve selective bromination.
- Add dimethylhydantoin and cuprous oxide to the reaction mixture. Heat the system to reflux at approximately 155°C for 18 hours to facilitate coupling.
- Cool the reaction mixture, filter the solids, and wash with ammonia solution and deionized water to isolate the pure imidazolidinedione product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits beyond mere chemical elegance. The reliance on commodity chemicals such as cuprous oxide and dimethylhydantoin, rather than exotic ligands or precious metal catalysts like palladium, drastically simplifies the raw material sourcing landscape. This shift reduces exposure to volatile market prices associated with rare earth metals and ensures a more stable supply chain continuity. Furthermore, the use of robust solvents like N,N-dimethylacetamide, which are widely available in bulk quantities, minimizes logistical bottlenecks and storage hazards compared to more volatile or toxic alternatives often found in legacy processes.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of stoichiometric copper oxide significantly lowers the direct material costs per kilogram of product. Additionally, the high selectivity of the bromination step reduces the burden on purification units, meaning less solvent and energy are consumed in recrystallization or chromatography steps. This streamlined workflow translates directly into substantial cost savings in fine chemical manufacturing, allowing for more competitive pricing in the global API market without compromising on quality margins.
- Enhanced Supply Chain Reliability: By utilizing starting materials that are commercially available in multi-ton quantities, such as trifluoromethylaniline derivatives, the risk of supply disruption is minimized. The process does not rely on custom-synthesized reagents with long lead times; instead, it leverages established chemical supply chains. This reliability is critical for maintaining consistent production schedules for high-purity pharmaceutical intermediates, ensuring that downstream drug manufacturers receive their materials on time and preventing costly delays in clinical or commercial drug launches.
- Scalability and Environmental Compliance: The reaction conditions, while requiring elevated temperatures for the coupling step, are well within the operating parameters of standard glass-lined steel reactors found in most multipurpose chemical plants. The absence of heavy metal waste streams simplifies wastewater treatment protocols, aiding in environmental compliance and reducing the costs associated with hazardous waste disposal. This ease of scale-up from kilogram to metric ton levels ensures that the technology can meet growing market demands efficiently while adhering to increasingly stringent global environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing clarity on process capabilities and product specifications.
Q: What is the primary advantage of using dibromo-dimethylhydantoin in this synthesis?
A: According to Patent CN1207732A, using dibromo-dimethylhydantoin allows for highly selective bromination at the para-position of the amino group under mild conditions (0°C), avoiding the formation of poly-brominated byproducts common with other reagents.
Q: Can this process be adapted to produce thio-derivatives?
A: Yes, the patent describes a conversion step where carbonyl groups (>C=O) in the imidazolidine ring can be transformed into thiocarbonyl groups (>C=S) using Lawesson's reagent, expanding the chemical diversity of the final intermediates.
Q: What catalysts are required for the coupling reaction?
A: The process utilizes copper-based catalysts, specifically cuprous oxide (Cu2O) or cupric oxide (CuO), which are cost-effective and readily available compared to precious metal catalysts often used in similar aromatic substitutions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenylimidazolidine Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation therapeutics. Our technical team has extensively analyzed the pathways described in CN1207732A and possesses the expertise to optimize these reactions for industrial production. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the bench to the plant. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of phenylimidazolidine derivatives meets the exacting standards required by global regulatory bodies.
We invite you to collaborate with us to leverage this advanced chemistry for your specific drug discovery programs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate how our manufacturing capabilities can accelerate your timeline and reduce your overall project costs effectively.
