Advanced Synthesis of Phenylimidazolidine Derivatives for Commercial Pharmaceutical Applications
Introduction to Advanced Phenylimidazolidine Synthesis
The pharmaceutical and fine chemical industries continuously demand more efficient and selective synthetic routes for complex heterocyclic intermediates. Patent CN1131219C discloses a groundbreaking methodology for the preparation of phenylimidazolidine derivatives, specifically targeting compounds of Formula (I). These structures are pivotal in the development of various bioactive agents, necessitating a manufacturing process that balances high yield with exceptional purity profiles. The disclosed innovation addresses critical bottlenecks in traditional aromatic substitution reactions by introducing a highly controlled halogenation strategy followed by a robust copper-catalyzed coupling sequence. This approach not only streamlines the synthetic pathway but also significantly enhances the reproducibility required for Good Manufacturing Practice (GMP) environments.
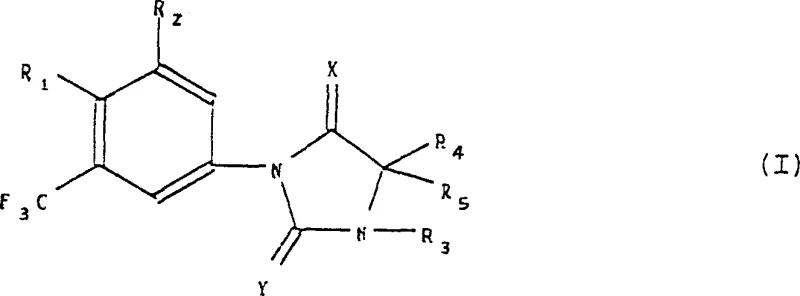
At the core of this technological advancement is the ability to precisely manipulate the substitution pattern on the phenyl ring attached to the imidazolidine core. Traditional methods often struggle with regioselectivity, leading to difficult-to-separate isomers that compromise the quality of the final active pharmaceutical ingredient (API). By leveraging specific reagents such as dibromodimethylhydantoin, the process achieves superior control over the introduction of functional groups. This level of precision is essential for R&D directors who require consistent impurity profiles to accelerate drug development timelines. Furthermore, the versatility of the method allows for the generation of a wide library of derivatives, including nitrile, iodo, and amino variants, providing a flexible platform for medicinal chemistry optimization.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of substituted phenylimidazolidines has been plagued by issues related to non-selective halogenation and harsh reaction conditions. Conventional electrophilic aromatic substitution often results in a mixture of ortho and para isomers, particularly when dealing with activated aniline derivatives. Separating these isomers typically requires energy-intensive chromatography or multiple recrystallization steps, which drastically reduces overall throughput and increases waste generation. Additionally, older coupling methodologies frequently relied on stoichiometric amounts of heavy metal reagents or extreme temperatures that degraded sensitive functional groups. These inefficiencies translate directly into higher production costs and longer lead times, creating significant friction for procurement managers seeking reliable supply chains for complex intermediates.
The Novel Approach
The novel process outlined in the patent data revolutionizes this landscape by employing a tandem strategy of selective bromination and catalytic coupling. Instead of indiscriminate halogenation, the method utilizes dibromodimethylhydantoin to selectively install a bromine atom at the para-position of the trifluoromethylaniline precursor. This intermediate is then subjected to a coupling reaction with a hydantoin derivative in the presence of cuprous oxide. This catalytic system operates effectively in polar aprotic solvents like dimethylacetamide at temperatures around 155°C to 165°C. The result is a direct, high-yielding route to the target imidazolidine scaffold with minimal byproduct formation.
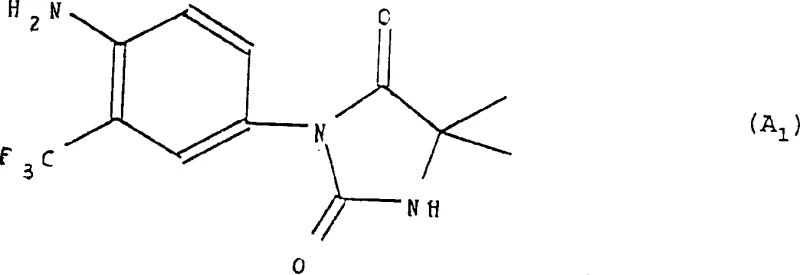
This streamlined approach eliminates the need for extensive purification between steps, as the selectivity of the initial bromination ensures that the subsequent coupling proceeds cleanly. For supply chain heads, this means a reduction in the number of unit operations and a decrease in solvent consumption, directly impacting the environmental footprint and cost of goods sold. The ability to isolate the final product through simple filtration and washing with aqueous ammonia further underscores the operational simplicity of this method. By shifting from stoichiometric reagents to catalytic processes, the technology aligns with modern green chemistry principles while delivering the commercial viability required for large-scale manufacturing.
Mechanistic Insights into Copper-Catalyzed Coupling and Selective Halogenation
The mechanistic elegance of this synthesis lies in the dual functionality of the reagents and the specific role of the copper catalyst. The initial step involves the generation of an electrophilic bromine species from dibromodimethylhydantoin, which reacts preferentially with the electron-rich para-position of the aniline ring due to steric and electronic factors. This selectivity is crucial, as it prevents the formation of ortho-substituted impurities that are notoriously difficult to remove. Following isolation or in-situ generation, the aryl bromide undergoes a nucleophilic aromatic substitution-like process facilitated by cuprous oxide. The copper catalyst likely forms a transient organometallic complex with the aryl halide, activating it towards nucleophilic attack by the nitrogen of the hydantoin ring.
Furthermore, the process allows for downstream functionalization via diazotization pathways, expanding the utility of the intermediate. The amino group on the phenyl ring can be converted into a diazonium salt, which serves as a versatile handle for introducing diverse functionalities such as cyano, iodo, or fluoro groups. For instance, treatment with sodium nitrite and hydrochloric acid generates the diazonium species, which can then be displaced by copper cyanide to install a nitrile group. This modularity is invaluable for R&D teams exploring structure-activity relationships (SAR). The control over reaction parameters, such as maintaining temperatures between 0°C and 20°C during diazotization, ensures the stability of the reactive intermediates and prevents decomposition, thereby safeguarding the integrity of the final product's impurity profile.
How to Synthesize Phenylimidazolidine Derivatives Efficiently
Implementing this synthesis requires careful attention to reaction conditions and reagent quality to maximize efficiency and safety. The process begins with the dissolution of the aniline precursor in a polar aprotic solvent, followed by the controlled addition of the brominating agent under cooling to manage exothermicity. Subsequent coupling steps demand rigorous temperature control to ensure complete conversion without degrading the hydantoin ring. The workup procedure is equally critical, utilizing specific washes to remove residual copper catalysts and inorganic salts. Detailed standardized operating procedures are essential to replicate the high yields observed in the patent examples consistently.
- Perform selective para-bromination of o-trifluoromethylaniline using dibromodimethylhydantoin in dimethylacetamide at 0°C to 20°C.
- React the resulting brominated intermediate with dimethylhydantoin in the presence of cuprous oxide catalyst at elevated temperatures (155°C to 165°C).
- Purify the crude product through aqueous workup, filtration, and recrystallization using solvents such as ethyl acetate or toluene mixtures.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers substantial advantages that resonate deeply with procurement and supply chain objectives. The primary benefit is the drastic simplification of the manufacturing process, which inherently drives down operational costs. By eliminating the need for complex separation techniques to remove regioisomers, the process reduces both labor hours and equipment utilization time. This efficiency gain allows for a more competitive pricing structure without compromising on quality margins. Moreover, the reliance on commercially available and stable starting materials, such as o-trifluoromethylaniline and dimethylhydantoin, mitigates the risk of raw material shortages. This stability is a key factor for supply chain heads who prioritize continuity and reliability in their vendor networks.
- Cost Reduction in Manufacturing: The transition to a catalytic coupling system significantly lowers the consumption of expensive reagents compared to traditional stoichiometric methods. The elimination of heavy metal scavengers and complex chromatographic purification steps further reduces the cost of goods. By optimizing solvent usage and enabling simpler workup procedures like filtration and crystallization, the overall resource intensity of the production is minimized. These cumulative efficiencies translate into a leaner cost structure, allowing for better budget allocation and improved profitability in the manufacturing of high-value pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent batch-to-batch quality, which is critical for maintaining long-term supply agreements. The use of standard industrial solvents like dimethylacetamide and dimethylformamide means that the process can be easily transferred to multiple manufacturing sites without requiring specialized infrastructure. This flexibility enhances supply chain resilience, reducing the risk of disruptions caused by equipment failures or site-specific limitations. Additionally, the high selectivity of the process minimizes the generation of hazardous waste, simplifying compliance with environmental regulations and ensuring uninterrupted production schedules.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction parameters that are easily manageable in large-scale reactors. The ability to operate at moderate temperatures and pressures reduces energy consumption and safety risks associated with high-pressure hydrogenation or cryogenic conditions. Furthermore, the reduced waste profile aligns with increasingly stringent global environmental standards. By minimizing the use of toxic reagents and facilitating easier waste treatment, the process supports sustainable manufacturing practices. This commitment to environmental stewardship not only protects the ecosystem but also enhances the brand reputation of the manufacturer in the eyes of eco-conscious clients.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its potential for integration into their supply chains. The following questions address common inquiries regarding the process capabilities, quality control measures, and adaptability to specific project requirements. These insights are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: What is the key advantage of using dibromodimethylhydantoin in this synthesis?
A: It provides exceptional regioselectivity for para-bromination of the aniline derivative, minimizing ortho-substitution byproducts and simplifying downstream purification.
Q: Can this process be scaled for industrial production?
A: Yes, the process utilizes common industrial solvents like dimethylacetamide and dimethylformamide, and standard catalysts like cuprous oxide, making it highly suitable for commercial scale-up.
Q: How is the purity of the final phenylimidazolidine derivative ensured?
A: High purity is achieved through precise temperature control during the coupling reaction and rigorous washing protocols using ammonia water and deionized water to remove metal catalysts and salts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenylimidazoline Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of pharmaceutical development programs. Our expertise in process chemistry allows us to master complex synthetic routes like the one described in CN1131219C, ensuring that we can deliver phenylimidazolidine derivatives with the highest standards of purity and consistency. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging our state-of-the-art facilities to meet the rigorous demands of the global market. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that employ advanced analytical techniques to verify every batch.
We invite you to collaborate with us to optimize your supply chain and reduce your time to market. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By partnering with us, you gain access to a reliable source of complex heterocyclic intermediates that can accelerate your drug discovery and development efforts. Please contact our technical procurement team to request specific COA data and route feasibility assessments for your next project, and let us demonstrate how our engineering capabilities can solve your sourcing challenges.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
