Advanced Asymmetric Synthesis of L-AMPB: A Breakthrough in Herbicide Intermediate Manufacturing
Advanced Asymmetric Synthesis of L-AMPB: A Breakthrough in Herbicide Intermediate Manufacturing
The global demand for high-efficiency herbicides continues to drive innovation in the synthesis of key active pharmaceutical and agrochemical ingredients. A pivotal development in this sector is detailed in patent CN101516898B, which outlines a robust and highly selective process for producing L-2-amino-4-(hydroxymethylphosphinyl)-butyric acid, commonly abbreviated as L-AMPB. This compound serves as a critical intermediate for broad-spectrum herbicides, offering significantly superior biological activity compared to its racemic DL-AMPB counterpart. The technical breakthrough lies in the utilization of a novel asymmetric Strecker reaction catalyzed by specific urea derivatives, which overcomes historical limitations regarding selectivity and cost. For R&D directors and procurement specialists, this methodology represents a paradigm shift from traditional enzymatic or precious-metal catalyzed routes towards a more sustainable and economically viable chemical synthesis platform.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the manufacturing of optically active L-AMPB has been fraught with significant technical and economic challenges that hinder large-scale commercialization. Traditional approaches often relied on enzymatic resolution or transamination reactions, which necessitate operation at extremely low substrate concentrations to maintain enzyme viability, thereby drastically reducing volumetric productivity and increasing solvent costs. Furthermore, alternative asymmetric synthesis routes frequently depended on the use of expensive chiral pool starting materials, such as D-valine or L-vinylglycine, which introduce substantial raw material costs and supply chain volatility. Perhaps most critically, some established methods utilized rhodium-based catalysts with chiral phosphine ligands for asymmetric hydrogenation; while effective, the reliance on precious rhodium metal creates a bottleneck for cost reduction and poses environmental disposal challenges due to heavy metal contamination risks in the final product.
The Novel Approach
In stark contrast to these legacy methods, the process disclosed in the patent introduces a streamlined chemical route that leverages an asymmetric Strecker reaction on a phosphorus-containing aldehyde. This innovative strategy bypasses the need for expensive chiral amino acid starters and eliminates the requirement for precious metal catalysts entirely. By reacting a specific phosphinyl-propanal derivative with an amine to form an imine intermediate, followed by cyanation in the presence of a chiral urea derivative catalyst, the process achieves high stereocontrol. This approach not only simplifies the reaction sequence but also utilizes readily available and cost-effective reagents. The ability to perform this transformation with high diastereoselectivity ensures that the resulting L-AMPB precursor requires minimal downstream purification, directly addressing the pain points of yield loss and complex isolation procedures found in older technologies.
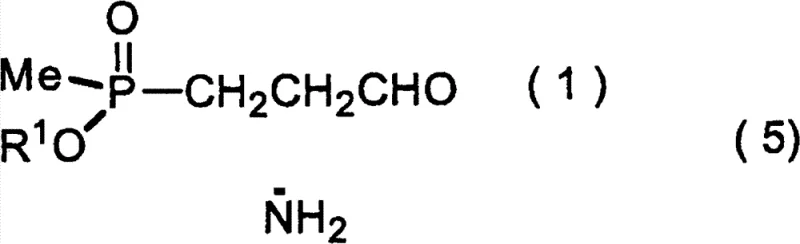
Mechanistic Insights into Urea-Derivative Catalyzed Asymmetric Strecker Reaction
The core of this technological advancement resides in the sophisticated activation mechanism facilitated by the chiral urea derivative catalysts, specifically those represented by formula (6) in the patent documentation. These organocatalysts function by simultaneously activating the imine substrate through hydrogen bonding interactions while orienting the cyanide source for a stereoselective nucleophilic attack. The dual hydrogen-bond donor capability of the thiourea or urea moiety stabilizes the transition state, effectively lowering the activation energy for the formation of the desired L-configured amino nitrile. This mechanistic precision is crucial when dealing with linear aliphatic aldehydes bearing polar phosphorus substituents, a substrate class that has historically proven difficult for asymmetric induction. The patent highlights that variations in the catalyst structure, such as modifying the amine groups attached to the urea backbone (e.g., dimethylamino vs. dibenzylamino), allow for fine-tuning of the steric environment to maximize enantiomeric excess.
From an impurity control perspective, this catalytic system offers distinct advantages by minimizing the formation of the undesired D-isomer and other side products. The high specificity of the urea catalyst ensures that the cyanide addition occurs predominantly from one face of the planar imine intermediate. Experimental data within the patent indicates that optimal catalyst configurations can achieve L-to-D ratios as high as 94:6, demonstrating exceptional stereochemical fidelity. This high level of purity at the intermediate stage significantly reduces the burden on downstream crystallization or chromatographic purification steps. For quality assurance teams, this means a more consistent impurity profile and a reduced risk of chiral contaminants carrying through to the final herbicide active ingredient, thereby ensuring compliance with stringent regulatory specifications for agrochemical products.
How to Synthesize L-AMPB Efficiently
The synthesis of L-AMPB via this patented route involves a logical three-step sequence that is amenable to standard chemical processing equipment. The process begins with the condensation of a protected phosphinyl-aldehyde with a protecting amine, such as benzylamine, to generate the reactive imine species in situ. This intermediate is then subjected to the asymmetric cyanation step under controlled low-temperature conditions to establish the chiral center. Finally, the nitrile group is hydrolyzed, and the protecting groups are removed to reveal the free amino acid. While the general chemistry is straightforward, precise control over reaction parameters such as temperature, stoichiometry, and catalyst loading is essential to replicate the high yields and selectivity reported in the literature. Detailed standardized operating procedures for scaling this pathway from laboratory to pilot plant are provided in the technical guide below.
- Condense 3-(alkoxymethylphosphinyl)-propanal with benzylamine in the presence of a dehydrating agent to form the imine intermediate.
- Perform an asymmetric Strecker reaction on the imine using hydrogen cyanide and a chiral urea derivative catalyst at low temperatures (-40°C).
- Execute acid hydrolysis of the nitrile group followed by deprotection of the hydroxyl and amino groups to yield the final L-AMPB product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers compelling strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the raw material portfolio; by shifting away from biologically derived chiral starters or precious metal catalysts, manufacturers can secure a more stable and predictable supply chain. The reagents required, such as simple alkyl amines and phosphorus-containing aldehydes, are commodity chemicals with robust global availability, mitigating the risk of supply disruptions that often plague specialty bio-reagents. Furthermore, the elimination of heavy metal catalysts like rhodium removes the need for expensive and time-consuming metal scavenging steps, which are often required to meet residual metal limits in final products. This streamlining of the process flow translates directly into reduced operational expenditures and shorter cycle times.
- Cost Reduction in Manufacturing: The economic impact of replacing precious metal catalysts with organic urea derivatives cannot be overstated. Rhodium and similar metals are subject to extreme price volatility and represent a significant capital tie-up in the catalyst inventory. By utilizing organocatalysts, the process eliminates this cost driver entirely. Additionally, the ability to run reactions at higher substrate concentrations compared to enzymatic methods improves reactor throughput, effectively lowering the cost per kilogram of production. The simplified workup procedures, which often involve basic filtration and concentration rather than complex chromatographic separations, further reduce solvent consumption and waste disposal costs, contributing to a leaner and more cost-effective manufacturing model.
- Enhanced Supply Chain Reliability: Reliance on enzymatic processes often introduces variability due to the sensitivity of biological systems to batch-to-batch differences in raw materials. In contrast, this chemical synthesis route offers superior reproducibility and robustness. The starting materials are chemically defined and stable, allowing for long-term storage and just-in-time delivery without the degradation risks associated with enzymes or sensitive chiral pools. This stability ensures consistent production schedules and reliable lead times for customers. Moreover, the scalability of the reaction conditions, which do not require specialized high-pressure hydrogenation equipment or sterile fermentation tanks, allows for flexible manufacturing across multiple sites, enhancing overall supply chain resilience against regional disruptions.
- Scalability and Environmental Compliance: From an environmental and safety standpoint, this process aligns well with modern green chemistry principles. The avoidance of heavy metals reduces the toxic load of the effluent, simplifying wastewater treatment and ensuring compliance with increasingly strict environmental regulations. The reaction conditions are generally mild, operating at temperatures that do not require extreme heating or cooling infrastructure, which lowers energy consumption. The high selectivity of the reaction minimizes the generation of isomeric byproducts, thereby improving the overall atom economy of the process. These factors combined make the technology highly scalable from pilot batches to multi-ton commercial production, facilitating a smoother technology transfer and faster time-to-market for new herbicide formulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating the process viability. Understanding these nuances is critical for technical teams assessing the feasibility of integrating this route into existing manufacturing portfolios. The answers reflect the specific advantages of the urea-catalyzed Strecker reaction over competing methodologies.
Q: What are the advantages of this asymmetric synthesis over enzymatic methods?
A: Unlike enzymatic methods which require low substrate concentrations and complex purification, this chemical catalytic route allows for higher concentrations, simpler workup procedures, and avoids the need for expensive optically active amino acids as starting materials.
Q: Does this process rely on expensive precious metal catalysts?
A: No, the process utilizes organic urea derivatives or other non-precious metal complexes (such as aluminum or titanium derivatives) as catalysts, eliminating the cost and supply chain risks associated with rhodium or other precious metals.
Q: What level of optical purity can be achieved with this method?
A: The patent data demonstrates high asymmetric yields, with L-isomer ratios reaching up to 94:6 (L:D) depending on the specific catalyst variant used, ensuring high-quality herbicide intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-AMPB Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this asymmetric synthesis technology for the agrochemical industry. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a commercial setting. Our state-of-the-art facilities are equipped to handle the specific requirements of phosphorus chemistry and asymmetric catalysis, maintaining stringent purity specifications through our rigorous QC labs. We are committed to delivering high-purity L-AMPB intermediates that meet the exacting standards required for next-generation herbicide development, providing our partners with a competitive edge in the global market.
We invite you to engage with our technical procurement team to discuss how this advanced manufacturing route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your volume requirements. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate our capability to deliver this critical intermediate with the reliability and quality that your operations demand. Together, we can accelerate the development of more efficient and sustainable agricultural solutions.
