Sustainable Manufacturing of Epsilon-Caprolactam via Biomass-Derived Intermediates for Global Polyamide Markets
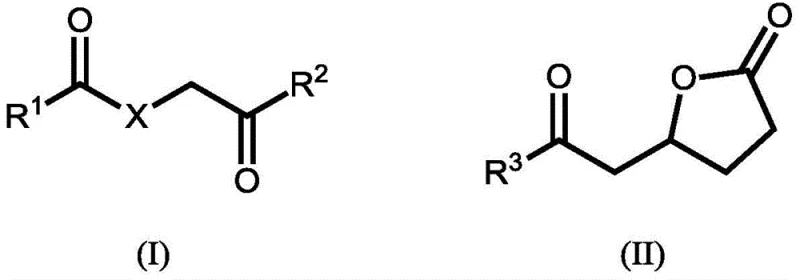
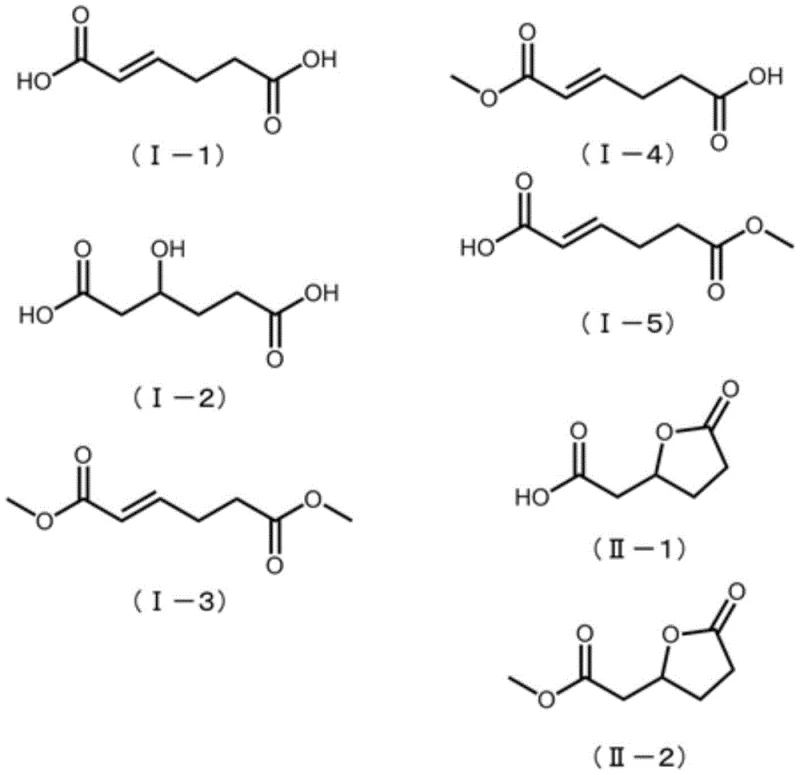
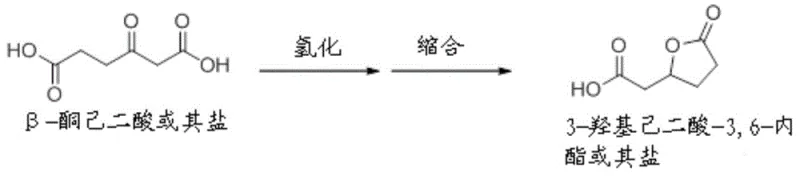
The global demand for sustainable polyamide precursors is driving a paradigm shift away from petrochemical dependence toward renewable biomass resources. Patent CN107074761B, published in April 2020, introduces a groundbreaking production method for epsilon-caprolactam, the critical monomer for Nylon 6, utilizing specific compounds derivable from biomass. This technology addresses the long-standing environmental and economic inefficiencies of the conventional Beckmann rearrangement process by employing a direct catalytic reaction of alpha-hydrogenated adipic acid, 3-hydroxyadipic acid, or their lactones with hydrogen and ammonia. By leveraging these bio-based intermediates, manufacturers can achieve high selectivity for epsilon-caprolactam while completely avoiding the generation of ammonium sulfate, a low-value by-product that plagues traditional synthesis routes. This report analyzes the technical feasibility and commercial implications of this novel pathway for R&D directors and supply chain strategists seeking to decarbonize their polymer value chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
For decades, the industrial standard for producing epsilon-caprolactam has relied on the Beckmann rearrangement of cyclohexanone oxime, a process heavily dependent on fuming sulfuric acid. While effective, this legacy technology suffers from severe atom economy issues, specifically the co-production of approximately 1.5 to 4.5 tons of ammonium sulfate for every ton of caprolactam produced. This by-product requires costly neutralization steps and creates a significant burden on waste management infrastructure, often necessitating sale into saturated fertilizer markets at low margins. Furthermore, the reliance on cyclohexanone ties the entire supply chain to volatile crude oil prices and benzene feedstocks, exposing manufacturers to geopolitical supply risks. The multi-step nature of converting benzene to cyclohexane, then to cyclohexanone, and finally to the oxime introduces multiple points of failure and energy consumption, making the overall process energetically intensive and environmentally taxing in terms of greenhouse gas emissions.
The Novel Approach
In stark contrast, the method disclosed in CN107074761B streamlines the synthesis into a more direct catalytic transformation using renewable feedstocks. The core innovation lies in the utilization of compounds represented by general formulas (I) and (II), such as alpha-hydrogenated adipic acid (also known as 2-hexenedioic acid) or 3-hydroxyadipic acid-3,6-lactone. These precursors can be sourced directly from microbial fermentation of sugars or derived from lignocellulosic biomass, effectively decoupling production from fossil fuels. The process involves a single-pot reaction where the carboxylic acid or lactone functionality reacts with ammonia and hydrogen in the presence of a noble metal catalyst. This approach not only bypasses the sulfuric acid neutralization step entirely but also demonstrates remarkable tolerance for isomeric mixtures, meaning expensive isomerization pre-treatment steps required in other bio-routes are rendered unnecessary. This results in a significantly shortened reaction sequence and a cleaner product profile.
Mechanistic Insights into Catalytic Hydrogenation and Amidation
The chemical transformation described in the patent relies on a sophisticated interplay between hydrogenation and amidation mechanisms facilitated by supported noble metal catalysts. The reaction initiates with the activation of hydrogen on the surface of the catalyst, typically palladium, platinum, or ruthenium supported on oxides like alumina or silica. In the case of alpha-hydrogenated adipic acid, the double bond is first hydrogenated to form a saturated intermediate, which subsequently undergoes amidation with ammonia to form adipamide. This adipamide intermediate is not merely a by-product but a crucial precursor that cyclizes under the reaction conditions to form the seven-membered lactam ring of epsilon-caprolactam. The patent data indicates that controlling the partial pressures of hydrogen and ammonia is critical; excessive hydrogen pressure can lead to over-reduction, forming unwanted amines like hexamethyleneimine, while insufficient ammonia limits the amidation rate. The use of solvents such as dioxane or gamma-butyrolactone further stabilizes the transition states and improves the solubility of the polar intermediates, ensuring homogeneous reaction kinetics.
Impurity control is another mechanistic highlight of this process, particularly regarding the suppression of ring-opened by-products. Traditional routes often struggle with hydrolysis of the lactam ring back to amino acids, but this catalytic system operates under conditions that favor cyclization. The patent highlights that even when adipamide is formed as a major species during the reaction, it can be recycled back into the reactor or converted in situ to the final lactam, thereby pushing the molar balance close to 100%. This high atom efficiency is achieved by carefully selecting reaction temperatures between 150°C and 300°C, which provide sufficient thermal energy for ring closure without degrading the sensitive amide bonds. The ability to use mixed cis-trans isomers of the starting diacid without prior separation further simplifies the impurity profile, as the catalyst system appears robust enough to process both geometric configurations efficiently.
How to Synthesize Epsilon-Caprolactam Efficiently
Implementing this synthesis route requires precise control over reactor parameters to maximize yield and minimize the formation of reduced amine by-products. The process begins with the preparation of the biomass-derived feedstock, ensuring that salts or free acid forms are compatible with the chosen solvent system. Operators must carefully manage the pressurization sequence, introducing ammonia before or alongside hydrogen to prevent premature saturation of the catalyst surface with hydrogen, which could favor reduction over amidation. The following guide outlines the standardized operational protocol derived from the patent examples to ensure reproducible high-quality output suitable for polymerization grade applications.
- Prepare the reactor by loading biomass-derived starting materials such as alpha-hydrogenated adipic acid or 3-hydroxyadipic acid-3,6-lactone along with a palladium-based catalyst.
- Introduce hydrogen and ammonia gases into the autoclave, adjusting partial pressures to between 0.5 MPa and 10 MPa for hydrogen and 0.1 MPa to 5 MPa for ammonia.
- Heat the reaction mixture to a temperature range of 150°C to 300°C for 0.5 to 12 hours to facilitate cyclization and amidation, followed by purification via distillation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this biomass-derived synthesis route offers transformative advantages in cost structure and risk mitigation. The most immediate impact is the drastic reduction in waste disposal costs associated with the elimination of ammonium sulfate by-products. In traditional manufacturing, the handling, storage, and sale of tons of low-value sulfate salt represent a significant logistical and financial burden; removing this step streamlines the downstream processing facility and reduces the need for large-scale neutralization infrastructure. Furthermore, sourcing raw materials from biomass fermentation opens up new supply channels that are less correlated with the volatility of the crude oil market, providing a natural hedge against petrochemical price spikes. This diversification of feedstock sources enhances supply chain resilience, ensuring continuity of production even during disruptions in the fossil fuel supply network.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the simplification of the purification train. Since the reaction selectively produces epsilon-caprolactam with minimal heavy by-products, the energy-intensive distillation columns required to separate complex mixtures in the Beckmann process can be optimized or reduced in size. Additionally, the ability to recycle intermediate adipamide back into the process loop ensures that raw material utilization is maximized, effectively lowering the cost per kilogram of the final monomer. The avoidance of corrosive fuming sulfuric acid also reduces maintenance costs for reactors and piping, extending the lifespan of capital equipment and lowering the total cost of ownership for the production facility.
- Enhanced Supply Chain Reliability: Utilizing biomass-derived intermediates such as alpha-hydrogenated adipic acid allows manufacturers to tap into the growing bio-economy, where feedstock availability is increasingly stable due to agricultural scaling. Unlike benzene, which is subject to refinery turnarounds and geopolitical tensions, sugar and starch-based feedstocks can be sourced regionally, reducing transportation lead times and carbon footprint. This localization potential is critical for multinational corporations aiming to meet regional content requirements and reduce Scope 3 emissions. The robustness of the catalyst system, which tolerates isomeric mixtures, further ensures that variations in upstream fermentation quality do not halt production, providing a buffer against raw material specification fluctuations.
- Scalability and Environmental Compliance: From a regulatory perspective, this process aligns perfectly with tightening global environmental standards regarding waste discharge and carbon intensity. The absence of sulfur-containing waste streams simplifies permitting processes and reduces the risk of non-compliance fines. Scalability is inherently supported by the use of standard high-pressure autoclave technology, which is well-understood in the fine chemical industry, allowing for straightforward scale-up from pilot plants to commercial multi-ton reactors. The use of recyclable heterogeneous catalysts further supports green chemistry principles, minimizing heavy metal contamination in the final product and reducing the environmental impact of catalyst disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biomass-based epsilon-caprolactam production technology. These insights are derived directly from the experimental data and claims within the patent documentation to assist decision-makers in evaluating the feasibility of adoption.
Q: What are the primary advantages of this biomass route over the traditional Beckmann rearrangement?
A: The primary advantage is the elimination of ammonium sulfate by-products, which are generated in massive quantities during the neutralization step of the traditional cyclohexanone oxime process. This new method significantly reduces waste treatment costs and environmental impact.
Q: Which catalysts are most effective for this hydrogenation-amidation process?
A: Noble metal catalysts, particularly palladium supported on carriers like gamma-alumina, silica, or carbon, demonstrate high selectivity. The patent specifies that palladium is particularly preferred for suppressing side reactions like the formation of hexamethyleneimine.
Q: Can this process utilize mixed isomers of the starting material?
A: Yes, unlike some prior art methods that require specific trans-trans isomers of muconic acid, this process tolerates mixtures of cis and trans isomers of alpha-hydrogenated adipic acid, simplifying the upstream purification requirements and reducing raw material costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Epsilon-Caprolactam Supplier
As the chemical industry pivots towards sustainability, NINGBO INNO PHARMCHEM stands at the forefront of translating innovative patent technologies into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required for high-performance polyamide applications, guaranteeing that every batch of epsilon-caprolactam meets the exacting standards of global textile and engineering plastic manufacturers.
We invite strategic partners to collaborate with us on optimizing this biomass-derived route for your specific supply chain needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this greener synthesis method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your move towards sustainable nylon production is backed by solid scientific data and reliable supply commitments.
