Advanced Epsilon-Caprolactam Production via N-Protonated Ionic Liquid Catalysis for Industrial Scale-Up
The chemical manufacturing landscape is undergoing a significant transformation driven by the urgent need for sustainable and efficient synthesis pathways, particularly for high-volume polymer precursors like epsilon-caprolactam. Patent CN1305855C introduces a groundbreaking methodology that leverages N-protonated caprolactam as a cationic group within a Brönsted acidic ionic liquid system to catalyze the Beckmann rearrangement of cyclohexanone oxime. This innovation represents a paradigm shift from traditional hazardous processes to a greener, more economically viable industrial standard that aligns with modern environmental regulations and cost-efficiency mandates. By utilizing the reaction product itself as part of the catalytic ionic liquid structure, this technology solves longstanding separation challenges that have plagued the industry for decades. The result is a streamlined production workflow that delivers high-purity epsilon-caprolactam with minimal environmental footprint and superior operational safety profiles for large-scale facilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional industrial production of epsilon-caprolactam has historically relied heavily on the Beckmann rearrangement using oleum containing significant concentrations of sulfur trioxide, a process fraught with severe engineering and environmental drawbacks. This legacy method necessitates reaction temperatures between 100°C and 130°C followed by a cumbersome neutralization step using large volumes of ammonia water to liberate the caprolactam from the sulfuric acid complex. The most critical disadvantage of this conventional route is the coproduction of massive quantities of low-value ammonium sulfate by-product, often ranging from 2 to 4 tons per ton of caprolactam, which creates a substantial waste disposal burden and economic inefficiency. Furthermore, the highly corrosive nature of the oleum reaction medium imposes rigorous material requirements on reactor vessels and piping, leading to escalated capital expenditure for corrosion-resistant alloys and frequent maintenance downtime. The complexity of the downstream purification process required to remove acidic residues and salts further compounds the operational costs and energy consumption associated with traditional manufacturing.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a functionalized room temperature ionic liquid that serves dually as both the catalyst and the reaction medium, fundamentally simplifying the entire chemical architecture of the synthesis. This innovative system operates under significantly milder thermal conditions, typically between 50°C and 120°C, which drastically reduces energy input requirements and enhances the safety profile of the reactor environment. Because the ionic liquid is designed such that the product epsilon-caprolactam does not form stable complexes with the catalytic cation, the need for alkaline neutralization is completely eliminated, thereby eradicating the generation of solid ammonium sulfate waste. The simplicity of the reaction system allows for a more direct isolation of the target molecule, reducing the number of unit operations required and minimizing the potential for product loss during purification. This technological leap not only addresses the environmental liabilities of the old process but also unlocks new possibilities for cost reduction in polymer intermediate manufacturing through improved yield and resource utilization.
Mechanistic Insights into N-Protonated Caprolactam Catalyzed Beckmann Rearrangement
The core of this technological advancement lies in the unique physicochemical properties of the Brönsted acidic ionic liquid, which creates a highly favorable microenvironment for the protonation of the oxime nitrogen-oxygen bond. Unlike conventional solvents where proton availability might be limited or unevenly distributed, the strong Coulombic forces within the ionic liquid matrix stabilize the cyclic imide positive ion intermediate essential for the rearrangement to proceed efficiently. The weak coordination ability of the ionic liquid anions enhances the freedom of the dissociated hydrogen ions, effectively increasing the apparent acidity of the system without requiring harsh mineral acids. This enhanced acidity facilitates the rapid migration of the phenyl or alkyl group in the oxime substrate, driving the reaction towards high conversion rates even at lower temperatures. The dynamic equilibrium established between the protonated caprolactam cation and the free base ensures that the catalyst remains active throughout the cycle while preventing product inhibition.
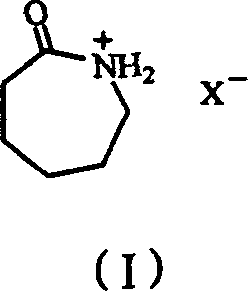
Impurity control is inherently managed through the specificity of the ionic liquid catalytic cycle, which favors the formation of epsilon-caprolactam over competing degradation pathways. The mild reaction conditions prevent the thermal decomposition of the sensitive lactam ring, which is a common issue in high-temperature gas-phase rearrangements using solid acid catalysts. Additionally, the homogeneous nature of the liquid-phase reaction ensures uniform heat and mass transfer, eliminating hot spots that could lead to localized charring or by-product formation. The primary by-product observed is cyclohexanone, which is chemically related to the starting material and can be easily recycled back into the oxime production line, creating a closed-loop system that maximizes atom economy. This level of selectivity is crucial for pharmaceutical and high-performance polymer applications where trace impurities can compromise the mechanical properties or biocompatibility of the final material.
How to Synthesize Epsilon-Caprolactam Efficiently
Implementing this synthesis route requires precise control over the molar ratios and thermal parameters to maximize the efficiency of the ionic liquid catalyst. The process begins with the preparation of the specific N-protonated caprolactam ionic liquid, followed by the introduction of cyclohexanone oxime in a controlled stoichiometric ratio ranging from 1:1 to 1:4. Detailed standardized synthesis steps see the guide below.
- Prepare the N-protonated caprolactam Brönsted acidic ionic liquid catalyst by combining caprolactam with a suitable Brönsted acid anion source.
- Mix the cyclohexanone oxime substrate with the ionic liquid catalyst in a molar ratio ranging from 1: 1 to 1:4 within a reaction vessel.
- Heat the mixture to a temperature between 50°C and 120°C for 1 to 6 hours, then separate the product using extraction methods without neutralization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this ionic liquid catalysis technology translates into tangible strategic advantages that extend far beyond simple chemical yield improvements. The elimination of the neutralization step and the associated waste stream fundamentally alters the cost structure of production by removing the need for purchasing bulk ammonia and managing hazardous waste disposal contracts. This simplification of the process flow reduces the overall processing time per batch, allowing for higher throughput within existing infrastructure without the need for significant capital expansion. The robustness of the ionic liquid catalyst, which can be reused multiple times without significant loss of activity, ensures a stable and predictable supply of the critical catalyst material, mitigating risks associated with volatile raw material markets. These factors combine to create a more resilient supply chain capable of meeting demanding delivery schedules while maintaining strict quality standards.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the drastic simplification of the downstream processing requirements and the elimination of waste treatment costs. By avoiding the generation of ammonium sulfate, manufacturers save significantly on waste disposal fees and the logistical costs associated with handling solid by-products. The reusability of the ionic liquid catalyst means that the effective cost per kilogram of catalyst consumed is substantially lower than single-use acid catalysts, contributing to long-term operational expenditure savings. Furthermore, the reduced corrosion rate extends the lifespan of reactor vessels and piping, deferring major capital replacement expenditures and lowering maintenance budgets. These cumulative efficiencies result in a more competitive cost position for the final epsilon-caprolactam product in the global market.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the reduced dependency on hazardous reagents like oleum and ammonia, which are often subject to strict transportation regulations and supply fluctuations. The milder reaction conditions reduce the risk of unplanned shutdowns due to safety incidents or equipment failures, ensuring a more consistent output volume for downstream customers. The ability to recycle the primary by-product, cyclohexanone, back into the feedstock stream further insulates the production process from raw material price volatility. This self-sustaining aspect of the chemistry provides a buffer against market disruptions, making the supplier a more reliable partner for long-term contractual agreements. Consistent quality and delivery performance are critical for maintaining the production schedules of nylon manufacturers and other downstream users.
- Scalability and Environmental Compliance: Scaling this technology to commercial production levels is facilitated by the liquid-phase nature of the reaction, which is compatible with standard chemical processing equipment used in the fine chemical industry. The absence of noxious gas emissions and solid waste generation simplifies the permitting process and ensures compliance with increasingly stringent environmental regulations across different jurisdictions. The low vapor pressure of the ionic liquid minimizes volatile organic compound (VOC) emissions, contributing to a safer workplace environment and reducing the need for complex scrubbing systems. This environmental compatibility enhances the corporate social responsibility profile of the manufacturing entity, appealing to eco-conscious clients and investors. The process is inherently designed for large-scale preparation, making it suitable for meeting the high-volume demands of the global polymer industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this patented ionic liquid catalysis technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation to ensure accuracy and reliability. Understanding these details is essential for evaluating the feasibility of integrating this process into existing manufacturing workflows.
Q: What are the primary advantages of this ionic liquid process over traditional oleum methods?
A: This process eliminates the need for large quantities of oleum and subsequent ammonia neutralization, thereby removing the generation of low-value ammonium sulfate waste and significantly reducing equipment corrosion risks.
Q: Is the ionic liquid catalyst reusable in this synthesis method?
A: Yes, the N-protonated caprolactam ionic liquid catalyst demonstrates excellent reusability as the product does not strongly bind to the catalyst, allowing for efficient separation and recycling of the catalytic medium.
Q: What are the typical reaction conditions for this Beckmann rearrangement?
A: The reaction typically proceeds under mild conditions with temperatures controlled between 50°C and 120°C and reaction times ranging from 1 to 6 hours, achieving high conversion and selectivity rates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Epsilon-Caprolactam Supplier
NINGBO INNO PHARMCHEM stands at the forefront of adopting advanced catalytic technologies to deliver superior chemical intermediates to the global market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes like this ionic liquid method are successfully translated into robust industrial operations. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of epsilon-caprolactam meets the exacting standards required for high-performance polymer applications. Our commitment to technical excellence allows us to offer products that not only meet but exceed the performance expectations of our international clientele.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through the adoption of this cutting-edge synthesis route. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production volumes and requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can support your strategic sourcing goals. Let us help you engineer a more efficient and sustainable future for your chemical manufacturing operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
