Advanced Nickel-Catalyzed Synthesis of 2,3,3',4'-Biphenyltetracarboxylic Dianhydride for High-Performance Polyimides
Advanced Nickel-Catalyzed Synthesis of 2,3,3',4'-Biphenyltetracarboxylic Dianhydride for High-Performance Polyimides
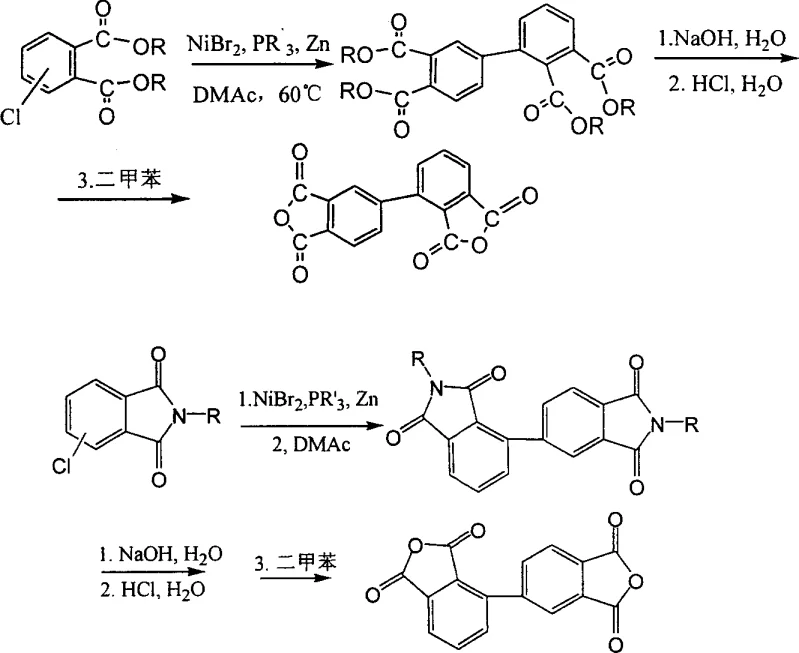
The development of high-performance polyimide materials relies heavily on the availability of specific monomeric building blocks, particularly isomers of biphenyltetracarboxylic dianhydride (BPDA). Patent CN1436780A introduces a transformative methodology for the preparation of 2,3,3',4'-biphenyltetracarboxylic dianhydride and its derivatives, addressing a critical gap in the electronic chemicals sector. Traditionally, the synthesis of biphenyl tetracarboxylic acids has been dominated by routes favoring the 3,3',4,4'-isomer, often relegating the 2,3,3',4'-isomer to the status of an undesirable byproduct with low yields. This patent delineates a robust nickel-catalyzed reductive coupling strategy that utilizes readily available 3- and 4-chlorophthalic acid diesters or chlorophthalimides as fundamental starting materials. By leveraging a catalytic system composed of nickel halides, trialkylphosphine ligands, and zinc powder in aprotic polar solvents, the process achieves direct and selective construction of the biphenyl backbone. This technological breakthrough not only enhances the structural diversity available to polymer chemists but also establishes a new benchmark for the reliable polyimide monomer supplier seeking to optimize their feedstock portfolios for advanced flexible circuit applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of biphenyl tetracarboxylic dianhydrides has faced significant challenges regarding isomer selectivity and process efficiency. Conventional synthetic pathways, such as the oxidative coupling of phthalic acid tetraalkyl esters or dehalogenation linked reactions, frequently suffer from poor regioselectivity, predominantly yielding the 3,3',4,4'-isomer which is thermodynamically more stable. When the 2,3,3',4'-isomer is produced via these traditional methods, it typically appears as a minor byproduct with very low yields, necessitating complex and costly purification steps to isolate the desired material. Furthermore, many older protocols rely on stoichiometric amounts of expensive transition metals or harsh reaction conditions that generate substantial quantities of hazardous waste, complicating environmental compliance and driving up the overall cost reduction in electronic chemical manufacturing. The inability to directly access the 2,3,3',4'-isomer efficiently has limited the exploration of polyimides with unique thermal and mechanical properties derived from this specific asymmetric structure, creating a bottleneck for innovation in high-end dielectric films.
The Novel Approach
The methodology described in CN1436780A represents a paradigm shift by employing a nickel-catalyzed cross-coupling mechanism that allows for precise control over the molecular architecture of the final product. Instead of relying on non-selective oxidative processes, this novel approach utilizes a reductive coupling of specific chloro-substituted precursors, namely 3-chlorophthalic acid dialkyl esters and 4-chlorophthalic acid dialkyl esters, in defined molar ratios. The use of a nickel halide catalyst activated by trialkylphosphine ligands and zinc powder facilitates the formation of the carbon-carbon bond between the aromatic rings under relatively mild conditions, typically ranging from 60°C to 120°C. This strategy effectively bypasses the thermodynamic preferences that plague oxidative methods, enabling the direct synthesis of the target 2,3,3',4'-biphenyltetracarboxylic tetraester or bisimide intermediates with significantly improved yields. The subsequent hydrolysis and cyclodehydration steps are streamlined, involving simple acidification and azeotropic dehydration in solvents like toluene or xylene, which simplifies the downstream processing and enhances the overall viability for the commercial scale-up of complex polyimide monomers.
Mechanistic Insights into Nickel-Catalyzed Reductive Coupling
The core of this synthetic innovation lies in the intricate catalytic cycle mediated by the nickel-phosphine complex, which operates through a classic oxidative addition and reductive elimination manifold tailored for aryl halides. In the initial phase, the nickel(II) halide precursor is reduced in situ by the zinc powder to generate the active nickel(0) species coordinated by the trialkylphosphine ligands. This electron-rich metal center then undergoes oxidative addition into the carbon-chlorine bond of the chlorophthalic acid derivatives, forming an organonickel(II) intermediate. The presence of the phosphine ligand is crucial here, as it stabilizes the low-valent nickel species and modulates its steric and electronic properties to favor the coupling of the specific 3- and 4-substituted aryl rings. Following the oxidative addition, a transmetallation or radical-mediated pathway facilitated by the zinc surface likely occurs, leading to a diarylnickel species that subsequently undergoes reductive elimination to forge the biaryl linkage. This mechanism is highly efficient in polar aprotic solvents such as DMAc or NMP, which solvate the ionic intermediates and facilitate the electron transfer processes required for the regeneration of the active catalyst.
Beyond the primary coupling event, the process incorporates sophisticated impurity control mechanisms inherent to the choice of starting materials and reaction conditions. By strictly controlling the ratio of 3-chloro to 4-chloro isomers in the feedstock, the reaction kinetics can be tuned to minimize the formation of homocoupled byproducts such as 3,3',4,4'-BPDA or 2,2',3,3'-BPDA. The use of zinc powder as a stoichiometric reductant ensures that the nickel catalyst remains in the active low-valent state throughout the reaction duration, preventing catalyst deactivation which often leads to incomplete conversion and difficult-to-remove impurities. Furthermore, the hydrolysis step using sodium hydroxide followed by acidification with concentrated hydrochloric acid serves as a dual-purpose purification stage; it not only converts the ester or imide intermediates into the free tetracarboxylic acid but also precipitates inorganic salts and residual metal species, leaving the organic acid in solution or as a clean precipitate depending on pH adjustments. This rigorous control over the chemical environment ensures that the final dianhydride product meets the stringent purity specifications required for electronic grade applications, where trace metal contaminants can severely degrade the dielectric performance of the resulting polyimide films.
How to Synthesize 2,3,3',4'-Biphenyltetracarboxylic Dianhydride Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing the importance of anhydrous conditions and precise temperature control to maximize yield. The process begins with the dissolution of the chlorophthalic precursors and the nickel catalyst system in a dry polar solvent under an inert nitrogen atmosphere to prevent oxidation of the sensitive intermediates.
- Prepare the reaction mixture by dissolving chlorophthalic acid diesters or chlorophthalimides with nickel halide catalyst and phosphine ligand in a polar aprotic solvent like DMAc under nitrogen protection.
- Heat the mixture to 60-120°C and add zinc powder as a reducing agent to initiate the coupling reaction, maintaining stirring for 0.5 to 6 hours until completion.
- Quench the reaction in water, hydrolyze the ester or imide intermediate with NaOH, acidify with HCl to precipitate the acid, and finally dehydrate via reflux in xylene to obtain the dianhydride.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this nickel-catalyzed route offers compelling economic and logistical benefits over traditional precious metal-catalyzed processes. The substitution of expensive palladium catalysts with abundant and cost-effective nickel halides drastically reduces the raw material expenditure associated with the catalytic system, directly impacting the bottom line of the manufacturing budget. Additionally, the use of zinc powder as a reductant is far more economical than alternative reducing agents, and the solvents employed, such as DMAc and NMP, are commodity chemicals with stable global supply chains, ensuring consistent availability and reducing the risk of production delays due to material shortages. The simplified workup procedure, which avoids complex chromatographic separations in favor of crystallization and filtration, further lowers the operational costs by reducing solvent consumption and waste disposal fees, aligning perfectly with the goals of cost reduction in electronic chemical manufacturing.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts like palladium represents a significant structural cost saving, as nickel is orders of magnitude cheaper and more abundant globally. This shift not only lowers the direct cost of goods sold but also reduces the financial burden associated with recovering and recycling expensive metals from waste streams. Furthermore, the high atom economy of the coupling reaction minimizes the generation of organic byproducts, leading to less waste treatment overhead and more efficient use of reactor volume per batch. The ability to operate at moderate temperatures also translates to lower energy consumption for heating and cooling cycles, contributing to a leaner and more cost-effective production profile that enhances competitiveness in the global market.
- Enhanced Supply Chain Reliability: The reliance on widely available commodity chemicals such as chlorophthalic esters, zinc powder, and standard industrial solvents mitigates the risk of supply chain disruptions often associated with specialized reagents. Since the raw materials are produced on a massive scale for various industries, procurement teams can leverage existing vendor relationships to secure favorable pricing and guaranteed delivery schedules. The robustness of the reaction conditions, which tolerate slight variations in temperature and stoichiometry without catastrophic failure, ensures high batch-to-batch consistency, reducing the likelihood of production stoppages due to quality deviations. This reliability is crucial for maintaining continuous supply to downstream polyimide manufacturers who operate on tight just-in-time delivery schedules for the electronics sector.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing standard stirred-tank reactors and common unit operations like filtration and distillation that are easily replicated from pilot plant to full commercial production. The absence of highly toxic reagents or extreme pressure requirements simplifies the safety engineering controls needed for large-scale implementation, facilitating faster regulatory approvals and site acceptance. Moreover, the aqueous workup and the potential for solvent recovery align with modern green chemistry principles, reducing the environmental footprint of the manufacturing process. This compliance with environmental standards not only avoids potential regulatory fines but also enhances the corporate sustainability profile, which is increasingly important for suppliers serving major multinational technology corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route, providing clarity for stakeholders evaluating its adoption. These insights are derived directly from the experimental data and process descriptions found within the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: What is the primary advantage of using nickel catalysis over palladium for BPDA synthesis?
A: Nickel catalysts significantly reduce raw material costs compared to precious metal palladium systems while maintaining high efficiency for reductive coupling of aryl halides, making the process more economically viable for large-scale electronic chemical manufacturing.
Q: How does this patent address the isomer purity issues common in biphenyl tetracarboxylic acid synthesis?
A: By specifically utilizing a controlled ratio of 3-chloro and 4-chloro phthalic acid derivatives as starting materials, the process directs the coupling towards the 2,3,3',4'-isomer, minimizing the formation of the thermodynamically favored 3,3',4,4'-isomer often seen in oxidative coupling methods.
Q: Can this synthesis method be scaled for industrial polyimide monomer production?
A: Yes, the use of common industrial solvents like DMAc and NMP, combined with moderate reaction temperatures between 60°C and 120°C, ensures that the process is highly adaptable for commercial scale-up without requiring exotic high-pressure or cryogenic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3,3',4'-Biphenyltetracarboxylic Dianhydride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity monomers play in the performance of next-generation polyimide materials, and we are committed to delivering excellence in every batch. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the identity and quality of our products, guaranteeing that every shipment meets the exacting demands of the semiconductor and flexible display industries. Our commitment to quality assurance means that customers can rely on us for consistent material performance, which is essential for maintaining the integrity of their own downstream manufacturing processes.
We invite potential partners to engage with our technical procurement team to discuss how this advanced nickel-catalyzed technology can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your production volume and requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive innovation and profitability in your organization. Let us be your trusted partner in navigating the complexities of fine chemical synthesis and securing a competitive edge in the global market.
