Scalable Nickel-Catalyzed Synthesis of 2,3,3',4'-BPDA for Advanced Polyimide Applications
The development of high-performance polyimide (PI) materials relies heavily on the availability of specialized monomers, particularly asymmetric dianhydrides like 2,3,3',4'-biphenyltetracarboxylic dianhydride (a-BPDA). Patent CN102020622A introduces a transformative methodology for synthesizing this critical compound, addressing long-standing inefficiencies in the production of unsymmetric polyimide precursors. This innovation is particularly relevant for manufacturers seeking to optimize the thermal and mechanical properties of PI films used in flexible electronics and advanced composites. By shifting away from precious metal catalysis and complex protection strategies, the disclosed method offers a streamlined pathway that enhances both economic viability and operational simplicity. For R&D teams and procurement strategists, understanding this shift is crucial for securing a reliable polyimide monomer supplier capable of meeting the rigorous demands of next-generation electronic materials.
The significance of this patent lies in its ability to bypass the traditional bottlenecks associated with biphenyl dianhydride synthesis. Historically, the preparation of asymmetric isomers has been plagued by low yields and difficult purification processes due to the formation of symmetric byproducts. The technical breakthrough presented here utilizes a direct coupling strategy of readily available halo-o-xylene derivatives, followed by a robust oxidation sequence. This approach not only improves the overall atom economy but also aligns with modern green chemistry principles by reducing the number of synthetic steps. As the demand for high-purity electronic chemical intermediates grows, adopting such efficient manufacturing protocols becomes a strategic imperative for maintaining competitiveness in the global supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for 2,3,3',4'-biphenyltetracarboxylic dianhydride have historically relied on palladium-catalyzed cross-coupling reactions of phthalic esters, a method fraught with significant technical and economic drawbacks. The primary issue with palladium catalysis in this context is the poor regioselectivity, which often results in a complex mixture of isomers where the desired asymmetric product constitutes less than 10% of the total yield. This low selectivity necessitates extensive and costly downstream purification processes, such as repeated recrystallization or chromatography, to isolate the target molecule. Furthermore, alternative methods utilizing rhodium catalysts for oxidative coupling, while offering better selectivity, are economically prohibitive due to the exorbitant cost of rhodium metal, rendering them unsuitable for large-scale industrial application. Previous nickel-catalyzed attempts often required the introduction of protecting groups, such as esters or amides, to prevent side reactions during the coupling phase. This addition of protection and deprotection steps significantly lengthens the production cycle, increases solvent consumption, and generates substantial chemical waste, thereby inflating the cost reduction in electronic chemical manufacturing efforts.
The Novel Approach
The methodology disclosed in the patent represents a paradigm shift by eliminating the need for protecting groups and precious metal catalysts entirely. Instead of starting with functionalized phthalic esters, the process initiates with the direct coupling of 3-halo-o-xylene and 4-halo-o-xylene derivatives. This strategic choice allows for the construction of the biphenyl backbone using inexpensive nickel catalysts and zinc reducing agents, which are far more cost-effective than palladium or rhodium systems. By deferring the oxidation of the methyl groups to carboxylic acids until after the coupling step is complete, the synthesis avoids the complications associated with reactive carboxyl groups during the bond-forming event. This sequential logic—coupling first, oxidizing second—simplifies the reaction profile and dramatically improves the isolation of the desired 2,3,3',4'-tetramethyl biphenyl intermediate. The subsequent oxidation and dehydration steps are performed under standard conditions, ensuring that the overall process remains robust and scalable for commercial scale-up of complex polymer additives.
Mechanistic Insights into Nickel-Catalyzed Cross-Coupling
The core of this synthesis lies in the nickel-catalyzed reductive coupling mechanism, which facilitates the formation of the carbon-carbon bond between the two aromatic rings. In this catalytic cycle, a nickel(II) halide precursor, such as nickel bromide, is reduced in situ by zinc powder to generate the active nickel(0) species. This low-valent nickel center undergoes oxidative addition with the carbon-halogen bonds of the 3-chloro-o-xylene and 4-chloro-o-xylene substrates. The presence of phosphine ligands, specifically triphenylphosphine or bipyridine derivatives, is critical for stabilizing the nickel intermediate and modulating its electronic properties to favor the cross-coupling over homocoupling. The ligand environment helps to discriminate between the different halo-xylene isomers, promoting the formation of the asymmetric 2,3,3',4'-substitution pattern. Following the oxidative addition, a transmetallation or radical recombination step occurs, facilitated by the zinc reductant, which regenerates the nickel catalyst and releases the coupled 2,3,3',4'-tetramethyl biphenyl product. This mechanistic pathway is highly efficient and tolerant of the methyl substituents, which remain inert under the coupling conditions.
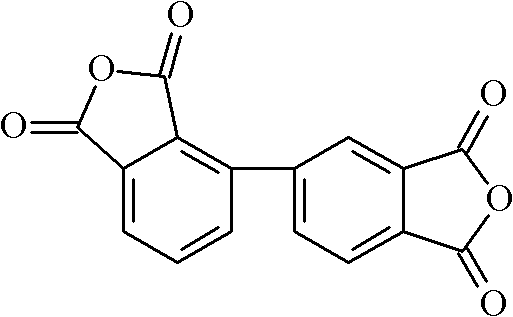
Following the coupling stage, the transformation of the tetramethyl biphenyl intermediate into the final dianhydride involves a vigorous oxidation process. The patent specifies the use of strong oxidizing agents such as potassium permanganate (KMnO4) or potassium dichromate in a mixed solvent system of pyridine and water. This step converts all four methyl groups into carboxylic acid functionalities, yielding 2,3,3',4'-biphenyltetracarboxylic acid. The choice of solvent is crucial here; pyridine acts as a phase transfer catalyst and solubilizer for the organic intermediate, while water serves as the medium for the inorganic oxidant. The reaction is typically conducted at elevated temperatures between 80°C and 120°C to ensure complete conversion. Finally, the tetracarboxylic acid undergoes dehydration, either through thermal fusion at high temperatures (210-380°C) or azeotropic distillation with acetic anhydride, to close the anhydride rings. This final cyclization step is thermodynamically driven and results in the highly stable 2,3,3',4'-biphenyltetracarboxylic dianhydride structure, ready for polymerization.
How to Synthesize 2,3,3',4'-Biphenyltetracarboxylic Dianhydride Efficiently
The practical execution of this synthesis requires careful control of stoichiometry and reaction parameters to maximize yield and purity. The process begins with the precise mixing of 3-halo-o-xylene and 4-halo-o-xylene in a molar ratio ranging from 1:1 to 3:3, depending on the desired optimization of isomer distribution. These substrates are combined with the nickel catalyst, phosphine ligand, and zinc powder in a polar aprotic solvent such as DMF or DMAc. The coupling reaction proceeds at moderate temperatures around 70°C, after which the crude tetramethyl biphenyl is isolated. Detailed standardized synthesis steps for optimizing each stage of this process are provided in the guide below.
- Perform nickel-catalyzed cross-coupling of 3-halo-o-xylene and 4-halo-o-xylene using zinc powder and triphenylphosphine to form 2,3,3',4'-tetramethyl biphenyl.
- Oxidize the tetramethyl biphenyl intermediate using potassium permanganate in a pyridine-water solvent system to generate the tetracarboxylic acid.
- Execute thermal dehydration of the tetracarboxylic acid via fusion or azeotropic distillation to yield the final 2,3,3',4'-biphenyltetracarboxylic dianhydride.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this nickel-catalyzed protocol offers substantial strategic benefits beyond mere technical feasibility. The most immediate impact is seen in the raw material cost structure, as the process replaces expensive noble metals with abundant base metals and utilizes commodity chemical feedstocks like chloro-xylenes. This shift significantly reduces the dependency on volatile precious metal markets, leading to more stable pricing models for the final monomer. Additionally, the elimination of protection and deprotection steps shortens the manufacturing lead time and reduces the volume of solvents and reagents required per kilogram of product. This efficiency translates directly into lower operational expenditures and a smaller environmental footprint, aligning with increasingly stringent regulatory requirements for chemical manufacturing facilities.
- Cost Reduction in Manufacturing: The economic advantage of this method is primarily driven by the substitution of palladium and rhodium catalysts with nickel, which is orders of magnitude cheaper. Furthermore, by avoiding the esterification and hydrolysis steps required in previous methods, the process saves on reagents like alcohols, acids, and bases, as well as the energy costs associated with additional heating and cooling cycles. The simplified workflow means fewer unit operations are required, reducing labor costs and equipment occupancy time. These cumulative savings allow for a more competitive pricing structure for high-purity electronic chemical intermediates without compromising on quality.
- Enhanced Supply Chain Reliability: Sourcing reliability is greatly improved because the key raw materials, such as 3-chloro-o-xylene and 4-chloro-o-xylene, are produced on a large industrial scale for other applications. Unlike specialized organometallic reagents that may have limited suppliers and long lead times, these halo-xylenes are readily available from multiple global chemical producers. The robustness of the nickel catalyst system also means that the process is less sensitive to minor variations in raw material quality, reducing the risk of batch failures. This resilience ensures a consistent supply of the dianhydride monomer, which is critical for maintaining continuous production lines in the downstream polyimide industry.
- Scalability and Environmental Compliance: The process conditions are inherently scalable, utilizing standard reactor types and common solvents that are easily managed in large-scale facilities. The absence of air-sensitive reagents simplifies the engineering controls required for safety, making the technology easier to transfer from pilot plants to full commercial production. From an environmental perspective, the reduction in synthetic steps leads to a lower E-factor (mass of waste per mass of product), as fewer byproducts and spent reagents are generated. This aligns with global sustainability goals and helps manufacturers meet strict waste discharge regulations, facilitating smoother permitting and operation of production sites.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the method's capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is the nickel-catalyzed route superior to palladium methods for a-BPDA?
A: Traditional palladium-catalyzed methods often suffer from poor selectivity (<10% yield) and require complex separation of isomers. The nickel-catalyzed approach described in patent CN102020622A utilizes abundant 3-halo and 4-halo o-xylene feedstocks directly, avoiding precious metal costs and achieving significantly higher selectivity through controlled stoichiometry and ligand optimization.
Q: Does this process require protection group chemistry?
A: No, a major advantage of this specific invention is the elimination of protection and deprotection steps. Unlike previous methods that required esterification or amidation to protect carboxylic groups before coupling, this route couples the methyl-substituted halides first and performs oxidation as a subsequent step, drastically simplifying the workflow and reducing waste.
Q: What are the scalability advantages of this synthesis method?
A: The process operates under relatively mild conditions (70°C for coupling, 80-120°C for oxidation) using common industrial solvents like DMF and toluene. The avoidance of air-sensitive organometallic reagents and the use of robust nickel catalysts make the transition from laboratory bench scale to multi-ton commercial production much more feasible and safer.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3,3',4'-Biphenyltetracarboxylic Dianhydride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance monomers play in the advancement of polyimide technology. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric demands of global electronics and aerospace industries. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify structural integrity and minimize impurity profiles. Our capability to implement the nickel-catalyzed synthesis route described in CN102020622A allows us to offer a cost-effective and reliable source of a-BPDA that outperforms traditional market offerings.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific application requirements. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the economic advantages of switching to our supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project needs, ensuring a seamless integration of our high-quality monomers into your polymerization processes.
