Advanced One-Pot Synthesis of Quinazoline Derivatives for Commercial Scale-Up
Introduction to Next-Generation Quinazoline Synthesis
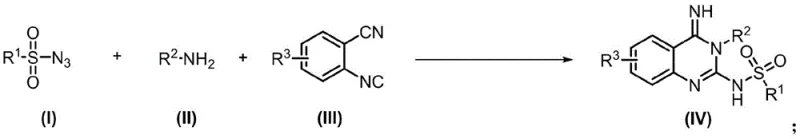
The pharmaceutical industry continuously seeks robust and scalable methodologies for constructing nitrogen-containing heterocycles, particularly the quinazoline scaffold, which serves as the core structure for blockbuster drugs like Gefitinib and Erlotinib. Patent CN113549020B introduces a groundbreaking approach to synthesizing these vital intermediates through a highly efficient, cobalt-catalyzed multicomponent reaction. This technology represents a significant leap forward in process chemistry, utilizing readily available starting materials such as isonitriles, azides, and amine derivatives to construct the quinazoline skeleton in a single operational step. By leveraging a base metal catalyst system, this invention addresses critical pain points in modern drug manufacturing, including the reliance on precious metals and the generation of excessive chemical waste. For R&D directors and procurement specialists, understanding this patented route is essential for securing a reliable quinazoline derivative supplier capable of delivering high-purity materials with improved economic viability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of quinazoline frameworks has relied on synthetic pathways that are often plagued by inefficiencies and environmental burdens. Traditional methods frequently necessitate the use of expensive transition metal catalysts, such as palladium or copper, which not only drive up raw material costs but also introduce challenges in removing trace metal residues to meet stringent pharmaceutical purity standards. Furthermore, many existing protocols require harsh reaction conditions, inert atmospheres, and multiple sequential steps, leading to poor atom economy and complex post-reaction workups. These factors collectively contribute to extended lead times and increased operational expenditures, creating bottlenecks in the supply chain for high-purity API intermediates. The inability to recover catalysts in many conventional processes further exacerbates the cost structure, making large-scale production less sustainable and economically attractive for generic drug manufacturers.
The Novel Approach
In stark contrast, the methodology disclosed in CN113549020B offers a streamlined, one-pot solution that dramatically simplifies the manufacturing landscape. By employing a cobalt-based catalyst system, specifically Co(acac)3, the reaction proceeds efficiently under mild thermal conditions (preferably 80°C) and, notably, in an air atmosphere, eliminating the need for costly inert gas setups. This novel approach utilizes simple, commercially accessible substrates—sulfonyl azides, amines, and o-cyanophenyl isonitriles—which react seamlessly to form the target quinazoline derivatives with excellent yields, reaching up to 98% in optimized examples. The process is characterized by its exceptional step economy and atom efficiency, significantly reducing the volume of solvent and reagents required. This shift towards a greener synthetic paradigm not only lowers the barrier to entry for production but also ensures a more consistent and reliable supply of complex heterocycles for downstream drug development.
Mechanistic Insights into Co(acac)3-Catalyzed Cyclization
Understanding the underlying catalytic cycle is paramount for R&D teams aiming to optimize this process for commercial scale-up. The reaction initiates with a rapid ligand exchange between the cobalt acetylacetonate catalyst and the isonitrile substrate, forming a reactive metal-cobalt complex. This intermediate subsequently interacts with the sulfonyl azide, facilitating the formation of a key metallacycle species. A critical step in the mechanism involves the extrusion of nitrogen gas (N2) from this intermediate, generating a highly reactive carbodiimide species coordinated to the metal center. This electrophilic intermediate is then subjected to nucleophilic attack by the amine component, followed by an intramolecular cyclization where the amino group attacks the nitrile functionality. This cascade of events effectively constructs the quinazoline ring system with high regioselectivity. The robustness of the cobalt catalyst allows it to turnover multiple times without significant degradation, a feature that is essential for maintaining reaction kinetics in large batch reactors.
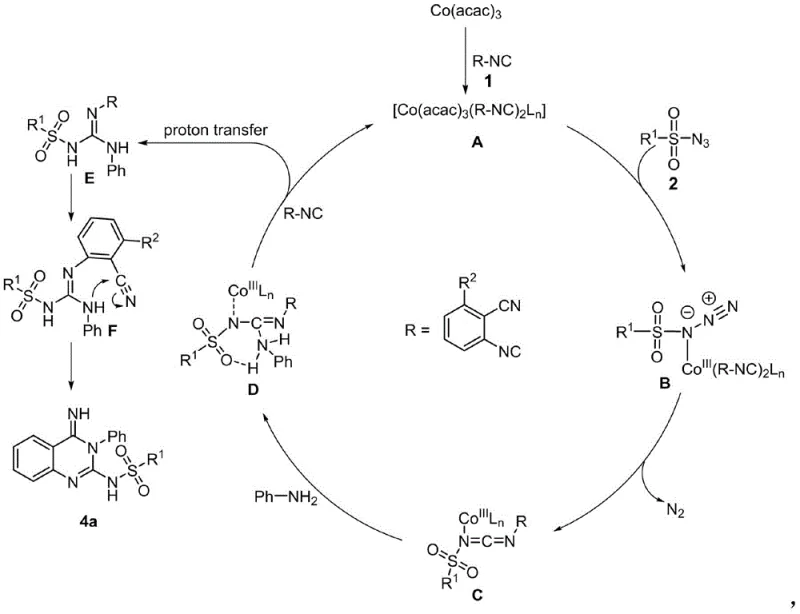
From an impurity control perspective, the mild nature of this catalytic system plays a crucial role in ensuring product quality. Harsh conditions often lead to side reactions such as polymerization of isonitriles or decomposition of sensitive functional groups on the aromatic rings. However, operating at moderate temperatures (40-100°C) minimizes these degradation pathways. The specificity of the cobalt catalyst towards the activation of the azide and isonitrile bonds ensures that the reaction trajectory remains focused on the desired cyclization, thereby reducing the formation of structurally related impurities. This inherent selectivity simplifies the purification process, often allowing for straightforward column chromatography or crystallization to achieve purity levels exceeding 95%. For quality assurance teams, this means a more predictable impurity profile and reduced risk of batch failure due to uncontrolled side reactions, which is a common challenge in heterocyclic chemistry.
How to Synthesize Quinazoline Derivatives Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and reaction parameters to maximize yield and purity. The patent outlines a generalized protocol where the molar ratio of sulfonyl azide, amine, and isonitrile is optimized, typically favoring a slight excess of the azide and isonitrile components to drive the reaction to completion. The choice of solvent is also critical, with toluene, acetonitrile, or DMF proving effective, though toluene is often preferred for its ease of removal and compatibility with the catalyst system. The reaction time generally spans 6 to 12 hours, providing a balance between complete conversion and operational throughput. Detailed standard operating procedures regarding temperature ramping, catalyst loading (typically 2.5-7.5 mol%), and workup strategies are essential for reproducibility. For comprehensive technical details on executing this synthesis, please refer to the standardized guide below.
- Prepare the reaction mixture by combining sulfonyl azide, amine derivative, and o-cyanophenyl isonitrile in an organic solvent such as toluene.
- Add the cobalt catalyst, preferably Co(acac)3, to the mixture under an air atmosphere at a molar ratio optimized for high yield.
- Heat the reaction to 80°C for approximately 12 hours, then purify the crude product via column chromatography to obtain high-purity quinazoline derivatives.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this cobalt-catalyzed technology translates into tangible strategic benefits that extend beyond simple chemistry. The shift from precious metal catalysts to abundant base metals like cobalt fundamentally alters the cost structure of raw materials, insulating the supply chain from the volatile pricing often associated with palladium or rhodium. Furthermore, the ability to recover and reuse the catalyst in large-scale reactions, as demonstrated in the patent data, significantly reduces the effective cost per kilogram of the final product. This economic efficiency is compounded by the simplified post-reaction processing; the elimination of complex purification steps required to remove heavy metal residues reduces both labor costs and waste disposal fees. Consequently, this method offers a pathway to substantial cost savings in pharmaceutical intermediate manufacturing without compromising on the quality or integrity of the final active ingredient.
- Cost Reduction in Manufacturing: The replacement of expensive noble metal catalysts with cost-effective cobalt salts directly lowers the bill of materials. Additionally, the high atom economy of the one-pot reaction minimizes raw material waste, while the mild reaction conditions reduce energy consumption associated with heating and cooling cycles. The potential for catalyst recovery further amplifies these savings, making the overall process financially superior to traditional multi-step syntheses that generate significant waste streams.
- Enhanced Supply Chain Reliability: The starting materials for this reaction—sulfonyl azides, amines, and isonitriles—are commodity chemicals with robust global supply chains, reducing the risk of shortages that can plague specialized reagents. The operational simplicity of running the reaction in air, rather than under strict inert conditions, also reduces the dependency on specialized equipment and gases, thereby enhancing the resilience of the manufacturing infrastructure against logistical disruptions.
- Scalability and Environmental Compliance: The green chemistry principles embedded in this method, such as high atom economy and reduced solvent usage, align perfectly with increasingly stringent environmental regulations. The process generates fewer hazardous byproducts, simplifying waste treatment and lowering the environmental footprint of production. This compliance facilitates smoother regulatory approvals and supports corporate sustainability goals, while the proven scalability of the catalyst system ensures that production can be seamlessly ramped from pilot batches to multi-ton commercial volumes.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this technology, we have compiled answers to common inquiries regarding the practical application and performance of this synthesis method. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation. Understanding these nuances is critical for assessing the feasibility of integrating this route into existing production lines or for initiating new development projects focused on quinazoline-based therapeutics.
Q: What are the primary advantages of this cobalt-catalyzed method over traditional synthesis?
A: This method eliminates the need for expensive noble metal catalysts and complex multi-step procedures. It operates under mild conditions (80°C) in air, offering superior atom economy and simplified post-treatment compared to conventional routes.
Q: Is the catalyst recoverable for large-scale production?
A: Yes, the patent data indicates that the cobalt catalyst can be recovered and reused in large-scale reactions without significant loss of catalytic activity, which is crucial for cost-effective commercial manufacturing.
Q: What is the biological potential of the synthesized compounds?
A: The synthesized quinazoline derivatives exhibit significant antibacterial activity against strains such as Staphylococcus aureus (MSSA/MRSA) and Enterococcus faecalis, making them valuable candidates for antibiotic drug development.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinazoline Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN113549020B for the future of pharmaceutical manufacturing. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are successfully translated into industrial reality. Our state-of-the-art facilities are equipped to handle complex heterocyclic synthesis with rigorous QC labs dedicated to maintaining stringent purity specifications. We understand that consistency and quality are non-negotiable in the pharma industry, and our commitment to process excellence guarantees that every batch of quinazoline derivatives meets the highest international standards.
We invite you to collaborate with us to leverage this cutting-edge technology for your drug development pipeline. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs, demonstrating how this efficient synthesis route can optimize your budget. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the global market through superior chemical manufacturing solutions.
