Advanced Synthesis of Substituted Quinazoline Derivatives for Commercial Anticancer Drug Manufacturing
Advanced Synthesis of Substituted Quinazoline Derivatives for Commercial Anticancer Drug Manufacturing
The pharmaceutical industry continuously seeks robust synthetic routes for potent anticancer agents, particularly protein tyrosine kinase (PTK) inhibitors. Patent CN1126743C introduces a groundbreaking methodology for the preparation of substituted quinazoline derivatives, specifically targeting the synthesis of compounds like 4-(3-bromoaniline)-6-(vinylamide)-quinazoline. This technology addresses a critical limitation in prior art, specifically the unwanted polymerization of vinyl groups during synthesis, which often compromises yield and purity. By employing a novel elimination strategy under mild conditions, this process ensures the structural integrity of the final active pharmaceutical ingredient (API) intermediate. For global procurement teams and R&D directors, understanding this patented pathway is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering high-quality materials for oncology drug development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of vinyl-substituted quinazolines has been plagued by significant chemical instability issues. Prior art, such as the methods disclosed in US Patent 5,760,041, often resulted in the polymerization of the sensitive vinyl group during the final stages of synthesis. This side reaction not only drastically reduces the overall yield of the desired product but also introduces complex impurity profiles that are difficult and costly to remove during downstream purification. For manufacturing facilities, dealing with polymeric byproducts means increased solvent consumption, longer processing times, and potential failures in meeting stringent regulatory purity specifications. Furthermore, harsh reaction conditions typically required to drive these transformations can degrade other sensitive functional groups on the quinazoline core, limiting the scope of substituents that can be introduced. These technical bottlenecks have traditionally hindered the cost reduction in API manufacturing for this class of anticancer drugs.
The Novel Approach
The methodology described in CN1126743C offers a transformative solution by decoupling the formation of the quinazoline ring from the introduction of the vinyl functionality. Instead of attempting to install the vinyl group under aggressive conditions, the process utilizes a precursor containing a removable leaving group (such as methoxy, methylthio, or chloro) at the relevant position. The key innovation lies in the final elimination step, which employs mild basic conditions to remove this leaving group and generate the vinyl double bond. 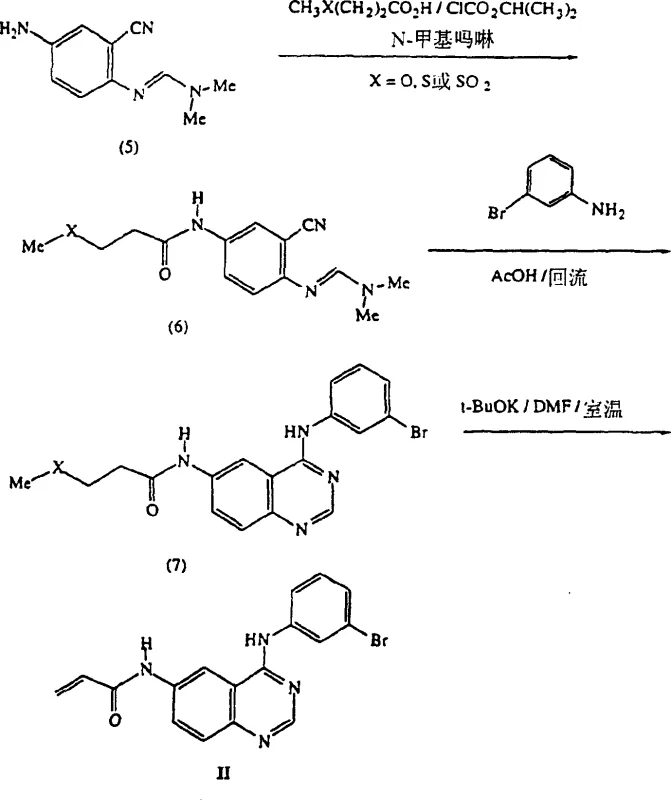 As illustrated in the reaction scheme, this approach allows the quinazoline core to be fully assembled and purified before the sensitive vinyl group is exposed. This strategic sequencing effectively prevents the polymerization issues observed in conventional methods, leading to superior product quality and process reliability. The ability to perform the initial acylation and rearrangement steps in a telescoped manner further streamlines the operation, making it highly attractive for commercial scale-up of complex pharmaceutical intermediates.
As illustrated in the reaction scheme, this approach allows the quinazoline core to be fully assembled and purified before the sensitive vinyl group is exposed. This strategic sequencing effectively prevents the polymerization issues observed in conventional methods, leading to superior product quality and process reliability. The ability to perform the initial acylation and rearrangement steps in a telescoped manner further streamlines the operation, making it highly attractive for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Dimroth Rearrangement and Base-Catalyzed Elimination
The core of this synthetic strategy relies on a sophisticated sequence of heterocyclic transformations, beginning with the acylation of an iminoaniline derivative. The starting material, typically a 3-cyano-4-(dimethylformylamino)aniline, reacts with an activated acylating agent, such as a mixed anhydride generated in situ from an acid and isobutyl chloroformate. This acylation step is critical for setting up the subsequent cyclization. Following acylation, the intermediate undergoes a Dimroth-type rearrangement in the presence of an aniline derivative and a protic solvent like acetic acid under reflux conditions. This rearrangement is the mechanistic engine that constructs the quinazoline ring system, converting the open-chain acylated precursor into the fused bicyclic heterocycle. The choice of acetic acid as the solvent is particularly advantageous, as it facilitates the proton transfer events necessary for the rearrangement while maintaining solubility of the intermediates.
The final and most crucial mechanistic step is the elimination of the leaving group (HZ) to form the vinylamide moiety. The patent demonstrates that treating the saturated side-chain intermediate with a weak base, such as potassium tert-butoxide in dimethylformamide (DMF) at room temperature, efficiently drives this elimination.  This mild condition is key to avoiding the thermal or radical initiation that leads to vinyl polymerization. The mechanism likely proceeds via an E2 or E1cb pathway, where the base abstracts a proton alpha to the carbonyl, leading to the expulsion of the leaving group and the formation of the carbon-carbon double bond. By carefully controlling the basicity and temperature, the process maximizes the formation of the desired alkene while minimizing side reactions. This precise control over the reaction mechanism is what enables the production of high-purity quinazoline derivatives suitable for clinical applications.
This mild condition is key to avoiding the thermal or radical initiation that leads to vinyl polymerization. The mechanism likely proceeds via an E2 or E1cb pathway, where the base abstracts a proton alpha to the carbonyl, leading to the expulsion of the leaving group and the formation of the carbon-carbon double bond. By carefully controlling the basicity and temperature, the process maximizes the formation of the desired alkene while minimizing side reactions. This precise control over the reaction mechanism is what enables the production of high-purity quinazoline derivatives suitable for clinical applications.
How to Synthesize 4-(3-bromoaniline)-6-(vinylamide)-quinazoline Efficiently
Implementing this synthesis requires careful attention to reaction parameters, particularly temperature control during the acylation and the choice of base for the elimination. The process begins with the activation of the carboxylic acid side chain, followed by coupling to the aniline core. Once the quinazoline ring is formed via the thermal rearrangement in acetic acid, the crude product can often be carried forward without extensive purification, thanks to the robustness of the intermediates. The final elimination step is performed at ambient temperature, which significantly reduces energy consumption compared to high-temperature dehydration methods.
- Acylation of 3-cyano-4-(dimethylformylamino)aniline with acid halides or mixed anhydrides in THF at 0-5°C using N-methylmorpholine.
- Reaction of the acylated product with an aniline derivative followed by Dimroth rearrangement in refluxing acetic acid to form the quinazoline core.
- Elimination of the leaving group (HZ) using potassium tert-butoxide in DMF at room temperature to generate the final vinylamide functionality without polymerization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers tangible benefits in terms of cost efficiency and supply security. The primary advantage lies in the significant simplification of the purification process. By eliminating the formation of polymeric byproducts, the need for complex chromatographic separations or multiple recrystallizations is drastically reduced. This directly translates to lower manufacturing costs and shorter cycle times, allowing for faster response to market demand. Additionally, the use of common, commercially available reagents such as THF, acetic acid, and potassium tert-butoxide ensures that the supply chain is not dependent on exotic or scarce catalysts. This accessibility of raw materials enhances supply chain reliability and mitigates the risk of production delays due to material shortages.
- Cost Reduction in Manufacturing: The streamlined nature of this process, particularly the potential for one-pot synthesis in the early stages, reduces the number of unit operations required. Fewer isolation and purification steps mean less solvent usage, lower waste disposal costs, and reduced labor hours. Furthermore, the high yields reported in the patent examples, such as the 89.1% yield for the quinazoline intermediate and 83.4% for the final vinyl product, indicate a highly efficient atom economy. This efficiency is crucial for achieving cost reduction in API manufacturing, especially for high-value oncology drugs where margin pressure is significant. The avoidance of expensive transition metal catalysts also removes the cost and regulatory burden associated with heavy metal removal and testing.
- Enhanced Supply Chain Reliability: The robustness of the chemical route ensures consistent batch-to-batch quality, which is vital for maintaining long-term supply contracts with pharmaceutical partners. Since the process avoids unstable intermediates and harsh conditions that could lead to batch failures, manufacturers can guarantee higher on-time delivery rates. The use of standard equipment and solvents means that production can be easily transferred between different manufacturing sites if necessary, providing flexibility in the global supply network. This reliability is a key differentiator for a reliable pharmaceutical intermediate supplier operating in the competitive generic and branded drug markets.
- Scalability and Environmental Compliance: From an environmental perspective, the process generates less hazardous waste compared to traditional methods that might require stoichiometric amounts of toxic reagents. The mild conditions of the final elimination step reduce energy consumption, contributing to a lower carbon footprint for the manufacturing process. Scalability is further supported by the fact that the exothermic events are manageable; for instance, the acylation is conducted at low temperatures (0-5°C) which is easily controlled in large reactors, and the elimination is at room temperature. These factors make the commercial scale-up of complex anticancer intermediates feasible and compliant with increasingly strict environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of these quinazoline derivatives. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing supply chains. The answers are derived directly from the experimental data and claims within the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: How does this patent method prevent vinyl group polymerization?
A: Unlike previous methods (e.g., US 5,760,041) which caused polymerization, this invention utilizes mild elimination conditions using weak bases like potassium tert-butoxide in DMF at room temperature, preserving the sensitive vinyl functionality.
Q: What are the preferred leaving groups for the elimination step?
A: The patent identifies methoxy, methylthio, and chloro groups as effective leaving groups. Specifically, converting methyl sulfides to sulfoxides or sulfones prior to elimination can further facilitate the reaction under mild conditions.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process uses commercially available starting materials and standard solvents like THF and acetic acid. The ability to perform the first two steps as a one-pot synthesis significantly simplifies the workflow and enhances scalability for industrial applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-(3-bromoaniline)-6-(vinylamide)-quinazoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving cancer therapies. Our technical team has extensively analyzed the pathways described in CN1126743C and possesses the expertise to optimize this chemistry for industrial production. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from clinical trials to full-scale market launch. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of quinazoline derivative meets the highest international standards for pharmaceutical use.
We invite you to collaborate with us to leverage this advanced synthetic technology for your drug development pipeline. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a stable, cost-effective supply of these critical anticancer intermediates, driving your projects forward with confidence and speed.
