Advanced Synthesis of Low-Toxicity Cephem Derivatives for Commercial Antibiotic Production
The pharmaceutical landscape is constantly evolving to address the dual challenges of bacterial resistance and drug safety, a theme central to the innovations disclosed in Chinese Patent CN86100852A. This pivotal document introduces a novel class of cephem nuclear derivatives that exhibit exceptional antibacterial activity while overcoming the severe toxicity limitations associated with earlier generations of cephalosporins. Specifically, the patent addresses the critical flaw of prior art compounds, such as those containing 3-(1-quinolinium)methyl groups, which were rendered clinically unusable due to acute toxicity levels around 100 mg/kg. In stark contrast, the new derivatives described herein utilize unique substituents at the 3-position of the cephem skeleton, resulting in a dramatic improvement in safety profiles with LD50 values generally exceeding 3 g/kg. This breakthrough represents a significant leap forward for reliable cephalosporin intermediate suppliers aiming to provide safer, more effective raw materials for the next generation of broad-spectrum antibiotics.
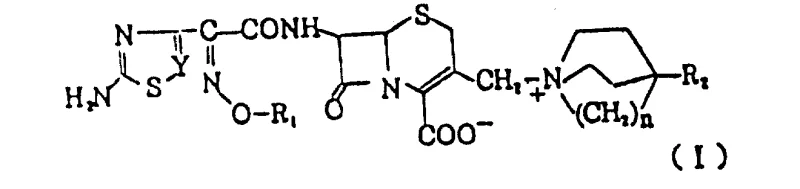
The structural versatility of these compounds allows for extensive optimization of pharmacokinetic properties, making them highly valuable assets in the development of modern antimicrobial therapies. By modifying the side chains at the 7-position and the quaternary ammonium groups at the 3-position, manufacturers can tailor the spectrum of activity against both Gram-positive and Gram-negative bacteria. The patent details a comprehensive range of substituents, including aminothiazole and thiadiazole groups, which are known to enhance beta-lactamase stability. This level of molecular engineering ensures that the resulting active pharmaceutical ingredients (APIs) maintain efficacy even against resistant strains, thereby securing a robust pipeline for high-purity antibiotic intermediates in the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of third-generation cephalosporins faced a significant bottleneck regarding the toxicity of the leaving groups at the 3-position of the cephem nucleus. Conventional methods often relied on introducing pyridinium or quinolinium methyl groups to enhance water solubility and antibacterial potency. However, as highlighted in the background of CN86100852A, these structures proved to be highly malicious in vivo, with acute poisoning values that precluded clinical application. The metabolic breakdown of these conventional intermediates often released toxic byproducts, creating substantial regulatory hurdles and safety liabilities for pharmaceutical developers. Furthermore, the synthesis of these older derivatives frequently involved harsh reaction conditions that compromised the stability of the sensitive beta-lactam ring, leading to lower yields and complex purification challenges that increased cost reduction in antibiotic manufacturing efforts.
The Novel Approach
The innovative strategy presented in this patent circumvents these toxicity issues by employing 3-(4-hydroxy-1-quinuclidinium)methyl or substituted quinoline groups that possess a fundamentally different toxicological profile. This novel approach leverages the steric and electronic properties of bicyclic amine systems to create stable quaternary ammonium salts that retain potent antibacterial activity without the associated lethal side effects. The synthesis pathway is designed to be mild and selective, preserving the integrity of the delicate cephem core while efficiently installing the complex side chains. By shifting the focus to these safer structural motifs, the patent enables the commercial scale-up of complex pharmaceutical intermediates that meet rigorous international safety standards. This transition not only mitigates risk but also opens new avenues for formulating injectable and oral cephalosporin preparations with improved therapeutic indices.
Mechanistic Insights into Nucleophilic Substitution and Acylation
The core chemical transformation driving this invention involves a precise nucleophilic substitution reaction at the 3-position of the cephem nucleus, typically starting from a 3-halomethyl or 3-acyloxymethyl precursor. In the preferred embodiment, a 3-acetoxymethyl cephem carboxylic acid is first activated using silylating agents like N-methyl-N-(trimethylsilyl)trifluoroacetamide, followed by iodination to generate a highly reactive 3-iodomethyl intermediate. This activated species then undergoes a quaternization reaction with a tertiary amine, such as 4-hydroxy-1-quinuclidine or a substituted quinoline, where the lone pair of electrons on the nitrogen atom attacks the electrophilic methylene carbon. This mechanism proceeds efficiently in polar aprotic solvents like acetonitrile or methylene dichloride, facilitating the formation of the stable quaternary ammonium bond essential for the drug's mechanism of action.
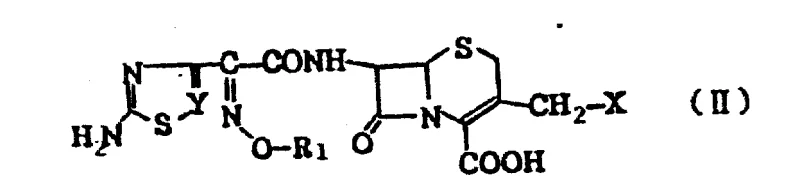
Following the installation of the 3-position substituent, the 7-amino group is acylated with an activated carboxylic acid derivative, typically an aminothiazole glyoxylic acid ester. This acylation step is critical for establishing the antibacterial spectrum and beta-lactamase stability of the final molecule. The reaction is carefully controlled using condensing agents or activated esters to ensure high regioselectivity for the 7-alpha position, minimizing the formation of unwanted isomers. Impurity control is managed through the use of protecting groups on the amino and carboxyl functionalities during intermediate steps, which are subsequently removed under mild hydrolytic or reductive conditions. This meticulous control over the reaction pathway ensures that the final product meets stringent purity specifications required for parenteral administration, thereby reducing lead time for high-purity pharmaceutical intermediates by minimizing downstream purification burdens.
How to Synthesize Cephem Derivatives Efficiently
The synthesis of these advanced cephem derivatives follows a logical sequence of activation, substitution, and acylation steps that are amenable to industrial production. The process begins with the preparation of the key cephem intermediate, which is then subjected to halogenation to create a reactive leaving group at the 3-position. This intermediate is immediately reacted with the chosen amine component in an anhydrous environment to prevent hydrolysis of the beta-lactam ring. The detailed standardized synthesis steps, including specific molar ratios, temperature controls, and workup procedures, are outlined in the comprehensive guide below to assist process chemists in replicating these high-value transformations.
- Prepare the 3-halomethyl cephem intermediate (Formula II) by activating the 3-acetoxymethyl group using silylating agents and iodinating reagents.
- React the activated cephem intermediate with the appropriate amine component (Formula III), such as 4-hydroxy-1-quinuclidine, in an anhydrous organic solvent.
- Purify the resulting quaternary ammonium salt through precipitation, filtration, and silica gel column chromatography to obtain the high-purity final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the synthetic routes described in CN86100852A offers tangible benefits in terms of operational efficiency and risk mitigation. The shift towards less toxic intermediates simplifies waste management protocols and reduces the regulatory burden associated with handling hazardous materials, leading to significant overhead savings. Moreover, the robustness of the reaction conditions allows for flexible manufacturing schedules, ensuring consistent supply continuity even in volatile market environments. By partnering with suppliers who have mastered these specific synthetic pathways, pharmaceutical companies can secure a competitive edge through access to superior quality raw materials that streamline their own API production processes.
- Cost Reduction in Manufacturing: The elimination of highly toxic quinolinium-based intermediates removes the need for expensive specialized containment and disposal systems, directly lowering operational expenditures. Additionally, the high yield and selectivity of the quaternization reaction minimize raw material waste, optimizing the overall cost of goods sold for the final antibiotic product. The ability to use common organic solvents and standard reagents further contributes to a leaner, more cost-effective manufacturing process that does not rely on exotic or prohibitively expensive catalysts.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, including cephem nuclei and various substituted amines, are commercially available from multiple global sources, reducing dependency on single-source suppliers. The synthetic route is resilient to minor fluctuations in reagent quality, ensuring that production batches remain consistent and reliable. This stability is crucial for maintaining uninterrupted supply chains for essential medicines, particularly in the face of global logistical challenges that can impact the availability of specialized chemical building blocks.
- Scalability and Environmental Compliance: The reactions described operate under mild temperature ranges and atmospheric pressure, making them inherently safer and easier to scale from pilot plant to multi-ton commercial production. The reduced toxicity profile of the final compounds aligns with increasingly strict environmental regulations regarding pharmaceutical effluents, facilitating smoother regulatory approvals. This environmental compatibility not only protects the ecosystem but also enhances the corporate social responsibility profile of the manufacturing entity, appealing to eco-conscious stakeholders and investors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these novel cephem derivatives. Understanding these details is essential for R&D teams evaluating the feasibility of incorporating these intermediates into their development pipelines. The answers are derived directly from the experimental data and claims presented in the patent documentation to ensure accuracy and relevance.
Q: What is the primary safety advantage of these new cephem derivatives?
A: Unlike prior art compounds containing 3-(1-quinolinium)methyl groups which exhibited high acute toxicity (LD50 approx. 100 mg/kg), the novel derivatives described in CN86100852A demonstrate significantly reduced toxicity with LD50 values generally exceeding 3 g/kg in mice.
Q: What solvents are suitable for the quaternization reaction?
A: The patent specifies the use of anhydrous organic solvents such as acetonitrile, methylene dichloride, tetrahydrofuran, dimethylformamide, and acetone, typically operating within a temperature range of -10°C to 60°C.
Q: Can these intermediates be scaled for commercial antibiotic production?
A: Yes, the synthesis utilizes standard organic transformation techniques including nucleophilic substitution and acylation, which are well-established for commercial scale-up in pharmaceutical manufacturing facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cephem Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving antibiotics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the demanding volume requirements of global pharmaceutical partners. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of cephem derivative we supply meets the highest international standards for safety and efficacy. Our commitment to excellence makes us the preferred choice for companies seeking to innovate in the field of antimicrobial therapy.
We invite you to contact our technical procurement team to discuss how our advanced manufacturing capabilities can support your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized synthetic routes can reduce your overall production costs. We are ready to provide specific COA data and route feasibility assessments to help you accelerate your development timeline and bring safer, more effective antibiotics to market faster.
