Next-Generation Cephem Derivatives: Scalable Synthesis and Commercial Viability
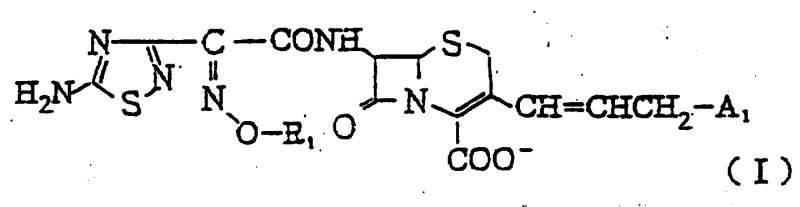
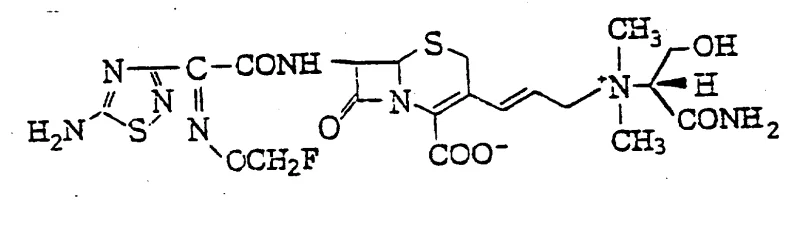
The pharmaceutical landscape is constantly evolving, driven by the urgent need for more effective antibiotics to combat resistant bacterial strains. Patent CN1022837C introduces a groundbreaking class of cephem derivatives characterized by a unique structural motif: a fluorine-substituted lower alkyl group, specifically a fluoromethoxyimino moiety, on the 7-side chain, coupled with a cyclic or acyclic ammonio group at the 3-position. This dual modification represents a significant leap forward in cephalosporin chemistry, offering enhanced antimicrobial spectra and improved pharmacokinetic properties. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediates supplier partnerships, understanding the depth of this technology is crucial. The invention provides not just a single compound, but a versatile platform for generating a wide array of potent antibacterial agents, addressing critical gaps in current therapeutic options.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional cephalosporin synthesis often struggles with balancing broad-spectrum activity against metabolic stability. Conventional 7-side chains, such as simple aminothiazolyl methoxyimino groups, while effective, can be susceptible to hydrolysis by extended-spectrum beta-lactamases produced by resistant Gram-negative pathogens. Furthermore, modifications at the 3-position have historically been limited to simple leaving groups or neutral substituents, which do not actively contribute to solubility or cellular uptake. The reliance on older synthetic routes often involves harsh conditions that can degrade the sensitive beta-lactam ring, leading to lower yields and complex purification challenges. These limitations result in higher manufacturing costs and restricted supply chain flexibility, making it difficult to meet the growing global demand for advanced antibiotics without compromising on quality or purity standards.
The Novel Approach
The methodology outlined in CN1022837C overcomes these hurdles through a strategic integration of fluorine chemistry and quaternary ammonium functionality. By introducing a fluoromethoxyimino group, the electron-withdrawing nature of the fluorine atom stabilizes the oxime ether linkage against enzymatic degradation, thereby extending the half-life of the drug in vivo. Simultaneously, the installation of a cationic ammonio group at the 3-position creates a zwitterionic or cationic character that drastically improves aqueous solubility without the need for external salt formation. This novel approach allows for the synthesis of compounds that exhibit fabulous anti-microbial activity, as demonstrated by MIC values as low as 0.025 μg/ml against key pathogens like E. coli and Klebsiella. The process utilizes mild reaction conditions, such as acylation at temperatures between -50°C to 50°C, preserving the integrity of the cephem nucleus while ensuring high stereochemical purity of the Z-isomer, which is critical for biological activity.
Mechanistic Insights into Fluoromethoxyimino Acylation and Quaternization
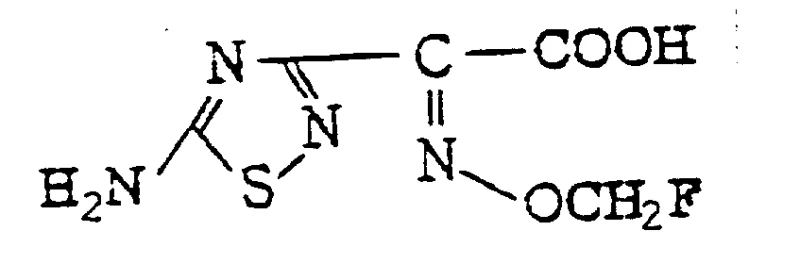
The core of this technology lies in the precise construction of the side chain and its subsequent attachment to the cephem nucleus. The synthesis of the key intermediate, 2-(5-amino-1,2,4-thiadiazol-3-yl)-(Z)-2-fluoromethoxyiminoacetic acid, involves a delicate fluoromethylation step. As shown in the experimental data, reacting the oximino precursor with fluorobromomethane in the presence of a base like potassium carbonate in DMSO allows for the selective introduction of the fluoromethyl group. This step is pivotal; the fluorine atom's high electronegativity modifies the electronic distribution of the side chain, enhancing its ability to penetrate the outer membrane of Gram-negative bacteria. The resulting acid is then activated, typically as an acid chloride or a reactive ester, to facilitate coupling with the 7-amino cephem scaffold.
Parallel to the side chain synthesis, the modification of the cephem core at the 3-position is achieved through a multi-step sequence involving phosphonium salt formation and nucleophilic displacement. The process begins with converting a 3-halomethyl cephem into a phosphonium salt, which serves as a stabilized ylide precursor. Subsequent reaction with an aldehyde generates a 3-allyl cephem intermediate. This allyl group is then functionalized with a halogen, creating a highly reactive electrophile capable of undergoing quaternization with various amines. This mechanism allows for the introduction of diverse ammonio groups, such as the 1-formamyl-2-hydroxyethyl dimethylammonio moiety, which adds chirality and specific hydrogen-bonding capabilities to the molecule. The final acylation step links these two sophisticated fragments, resulting in the target cephem derivative with exceptional structural complexity and therapeutic potential.
How to Synthesize High-Purity Cephem Derivatives Efficiently
Executing the synthesis of these advanced cephem derivatives requires strict adherence to the patented protocols to ensure the correct stereochemistry and purity profiles demanded by regulatory bodies. The process involves handling sensitive intermediates like fluoromethoxylamine and activated acid chlorides, which necessitates controlled environments and precise stoichiometry. The patent details specific solvent systems, such as mixtures of methylene chloride and isopropyl ether for crystallization, which are critical for isolating the product in high yield and purity. For manufacturers aiming for commercial scale-up of complex pharmaceutical intermediates, understanding these nuances is vital to avoid batch failures and ensure consistent quality. The detailed standardized synthesis steps provided in the patent serve as a robust foundation for process development teams to optimize reaction parameters for larger vessel sizes.
- Preparation of the 7-side chain acid involving fluoromethylation of oximino acetate precursors using fluorobromomethane under basic conditions.
- Modification of the cephem nucleus at the 3-position via phosphonium salt formation followed by nucleophilic substitution with allyl halides.
- Final quaternization of the 3-allyl cephem with specific amines and subsequent acylation at the 7-position with the activated side chain acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology offers significant strategic advantages beyond mere technical superiority. The synthetic route described eliminates the need for exotic transition metal catalysts often found in cross-coupling reactions, relying instead on widely available organic reagents and standard unit operations. This simplification of the chemical bill of materials translates directly into cost reduction in antibiotic manufacturing. By avoiding expensive noble metals, the process reduces both raw material costs and the downstream burden of heavy metal removal, which is a costly and time-consuming step in API production. Furthermore, the robustness of the fluoromethylation and quaternization steps ensures high reproducibility, minimizing batch-to-batch variability and reducing waste generation.
- Cost Reduction in Manufacturing: The process design inherently lowers production expenses by utilizing cost-effective reagents like fluorobromomethane and common solvents such as acetone and ethyl acetate. The elimination of complex catalytic cycles means that capital expenditure on specialized reactor linings or filtration systems for catalyst recovery is unnecessary. Additionally, the high selectivity of the reactions minimizes the formation of difficult-to-remove impurities, streamlining the purification workflow and reducing solvent consumption. These factors collectively contribute to a leaner manufacturing model that can withstand market price fluctuations while maintaining healthy margins.
- Enhanced Supply Chain Reliability: Sourcing raw materials for this synthesis is straightforward, as the key building blocks are derived from commodity chemicals or established pharmaceutical intermediates. This reduces the risk of supply disruptions associated with niche or single-source reagents. The synthetic pathway is modular, allowing for the decoupled production of the side chain acid and the cephem nucleus, which can then be converged in the final step. This flexibility enables manufacturers to build inventory buffers for critical intermediates, ensuring reducing lead time for high-purity pharmaceutical intermediates and guaranteeing continuity of supply even during periods of high demand or logistical constraints.
- Scalability and Environmental Compliance: The reaction conditions specified, such as temperatures ranging from -20°C to 50°C, are easily achievable in standard stainless steel reactors found in most multipurpose API plants. The absence of pyrophoric reagents or extreme pressures simplifies safety protocols and facilitates rapid scale-up from pilot to commercial scale. Moreover, the process generates waste streams that are manageable through conventional treatment methods, aligning with increasingly stringent environmental regulations. This compliance reduces the risk of regulatory shutdowns and enhances the long-term sustainability of the manufacturing operation, making it an attractive option for green chemistry initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these cephem derivatives. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these details helps in evaluating the feasibility of integrating this technology into existing production portfolios and assessing its potential impact on product pipelines.
Q: What is the primary advantage of the fluoromethoxyimino group in these cephem derivatives?
A: The introduction of the fluoromethoxyimino group significantly enhances antimicrobial activity, particularly against Gram-negative bacteria, by improving beta-lactamase stability and binding affinity.
Q: How does the 3-position quaternary ammonium group affect the drug's properties?
A: The cationic group at the 3-position improves water solubility and pharmacokinetic profiles, facilitating better distribution and efficacy in treating severe infections.
Q: Are these intermediates suitable for large-scale commercial production?
A: Yes, the patent outlines robust synthetic routes using standard reagents and conditions that are amenable to scale-up from kilogram to multi-ton annual production capacities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cephem Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the chemistry described in CN1022837C for the next generation of cephalosporin antibiotics. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering stringent purity specifications that meet or exceed global pharmacopoeia standards, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis of the fluoromethoxyimino side chain or the fully assembled cephem derivative, our infrastructure is designed to support your specific volume and quality requirements with precision and reliability.
We invite you to collaborate with us to unlock the full commercial value of this patented technology. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs, highlighting opportunities for efficiency gains and expense reduction. Please contact us to request specific COA data for our available intermediates and to discuss route feasibility assessments for your upcoming projects. Together, we can accelerate the development of life-saving antibiotics and secure a robust supply chain for the future of infectious disease treatment.
