Advanced Sildenafil Synthesis: Technical Breakthroughs and Commercial Scalability for Global Pharma
The pharmaceutical industry continuously seeks robust synthetic pathways that balance high purity with economic efficiency, particularly for high-volume therapeutic agents. Patent CN1092660C, published in October 2002, presents a significant advancement in the preparation of Sildenafil, widely known by its brand name Viagra. This intellectual property outlines a refined methodology focusing on two critical transformation stages: the cyclization of a specific pyrazole precursor and a subsequent N-methylation reaction. Unlike earlier methodologies that often relied on harsh conditions or expensive catalytic systems, this approach emphasizes the use of accessible alkaline reagents and straightforward solvent systems. For R&D Directors and technical decision-makers, understanding the nuances of this patent is crucial, as it offers a viable alternative for producing high-purity Active Pharmaceutical Ingredients (APIs). The process not only addresses the structural integrity of the final heterocyclic compound but also implicitly targets the reduction of process complexity, which is a key driver for modern pharmaceutical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for Sildenafil and related pyrazolo-pyrimidinone derivatives have historically faced several bottlenecks that hinder efficient commercial production. Many legacy processes involve multi-step sequences that require stringent anhydrous conditions or the use of hazardous reagents, which complicate safety protocols and waste management. Furthermore, conventional cyclization methods often suffer from moderate yields due to side reactions or incomplete conversions, necessitating extensive purification steps that drive up operational costs. The reliance on specific transition metal catalysts in some older patents introduces the risk of heavy metal contamination, requiring additional downstream processing to meet stringent regulatory limits for residual metals in APIs. These factors collectively contribute to longer lead times and higher production costs, creating friction in the supply chain for reliable agrochemical intermediate supplier networks and pharmaceutical manufacturers alike who demand consistency and speed.
The Novel Approach
In contrast, the methodology disclosed in CN1092660C introduces a streamlined pathway that mitigates many of these historical challenges by leveraging alkaline-mediated cyclization. This novel approach utilizes readily available bases such as sodium hydroxide, potassium hydroxide, or potassium tert-butoxide, which are significantly more cost-effective and easier to handle than specialized organometallic catalysts. The reaction conditions are flexible, accommodating a temperature range from 50°C to 170°C, allowing process engineers to optimize energy consumption based on available infrastructure. Additionally, the compatibility with common organic solvents like ethanol, methanol, and tetrahydrofuran simplifies solvent recovery and recycling processes. By shifting the paradigm towards simpler reagents and robust conditions, this method enhances the overall feasibility of cost reduction in electronic chemical manufacturing and pharmaceutical sectors, ensuring a more stable and predictable production workflow.
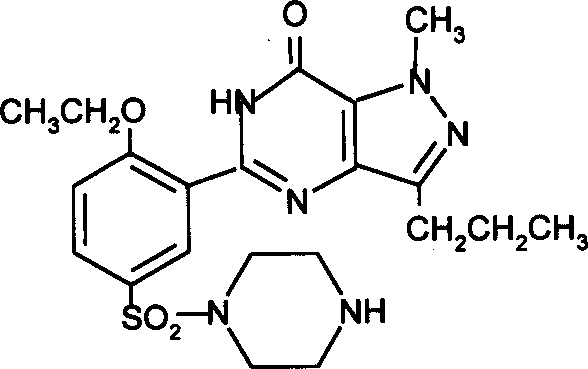
Mechanistic Insights into Alkaline Cyclization and N-Methylation
The core of this technical breakthrough lies in the precise execution of the cyclization reaction, where the open-chain precursor undergoes ring closure to form the pyrazolo-pyrimidinone core. Under alkaline conditions, the deprotonation of active hydrogen atoms facilitates nucleophilic attack, driving the formation of the heterocyclic ring system with high regioselectivity. The patent highlights the potential use of oxidants like hydrogen peroxide in conjunction with the base, which can assist in oxidative cyclization steps, ensuring complete conversion of the starting material. This mechanistic pathway is critical for R&D teams focused on impurity control, as the choice of base and solvent directly influences the profile of by-products. For instance, the use of tert-butanol with potassium tert-butoxide provides a non-nucleophilic environment that minimizes unwanted side reactions, thereby preserving the integrity of the sensitive functional groups present in the molecule.
Following the cyclization, the N-methylation step serves as the final functionalization to generate the bioactive Sildenafil molecule. This transformation can be achieved through multiple reagent systems, including the classic Eschweiler-Clarke conditions using formaldehyde and formic acid, or via alkylation with methyl iodide or methyl sulfate in the presence of a base. The flexibility in reagent selection allows manufacturers to choose the most economically viable option without compromising yield. Crucially, the reaction temperature for this step is maintained between 0°C and 120°C, providing a wide operational window that supports both batch and continuous flow processing. Understanding these mechanistic details is essential for achieving commercial scale-up of complex polymer additives and pharmaceutical intermediates, as it ensures that the critical quality attributes of the API remain consistent across different production batches.
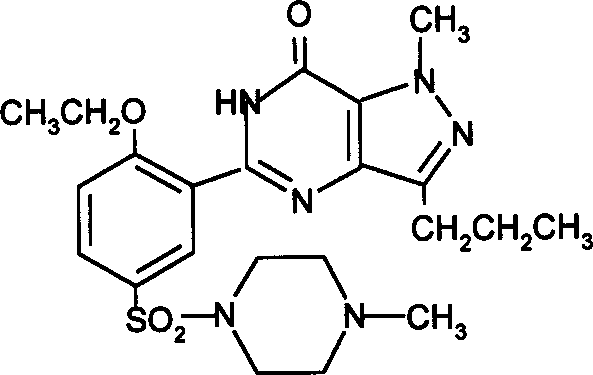
How to Synthesize Sildenafil Efficiently
Implementing this synthesis route requires a disciplined approach to reaction parameters and workup procedures to maximize yield and purity. The process begins with the preparation of the cyclization mixture, where the precursor is dissolved in a suitable solvent and treated with the selected base under reflux conditions. Monitoring the reaction progress via HPLC or TLC is recommended to determine the optimal endpoint, typically ranging from 2 to 72 hours depending on the specific reagents used. Once the cyclization is complete, the mixture is neutralized, and the intermediate is extracted and purified, often through recrystallization, to prepare it for the subsequent methylation step. The detailed standardized synthesis steps see the guide below for specific molar ratios and safety precautions.
- Perform alkaline cyclization of the pyrazole carboxamide precursor using bases like potassium tert-butoxide or sodium hydroxide in solvents such as tert-butanol or ethanol at 50-170°C.
- Isolate the intermediate pyrazolo-pyrimidinone compound through neutralization, extraction, and purification to ensure high structural integrity before methylation.
- Execute N-methylation using formaldehyde/formic acid or methyl iodide/methyl sulfate under controlled temperatures (0-120°C) to yield the final Sildenafil active ingredient.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this patented process offers substantial strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the simplification of the raw material basket; by utilizing common alkaline reagents and standard organic solvents, manufacturers can reduce dependency on specialized or single-source suppliers. This diversification of the supply base enhances supply chain reliability, mitigating the risks associated with geopolitical disruptions or market volatility for niche chemicals. Furthermore, the elimination of expensive transition metal catalysts removes the need for costly scavenging steps and rigorous metal testing, directly contributing to cost reduction in manufacturing. These efficiencies translate into a more competitive pricing structure for the final API, allowing companies to offer better value to their downstream partners while maintaining healthy margins.
- Cost Reduction in Manufacturing: The process significantly lowers operational expenditures by replacing precious metal catalysts with inexpensive inorganic bases like sodium hydroxide or potassium carbonate. This substitution not only reduces the direct cost of goods sold but also minimizes the environmental burden associated with heavy metal waste disposal. Additionally, the high yields reported in the patent embodiments suggest a more efficient use of starting materials, reducing the overall material intensity of the production process. Such improvements in atom economy and reagent cost are vital for maintaining profitability in a competitive generic pharmaceutical market.
- Enhanced Supply Chain Reliability: By relying on commoditized chemicals such as ethanol, methanol, and formaldehyde, the production process becomes less susceptible to supply shocks that often affect specialized reagents. The robustness of the reaction conditions, which tolerate a wide range of temperatures and solvent mixtures, ensures that production can continue even if specific grades of solvents are temporarily unavailable. This flexibility is crucial for reducing lead time for high-purity pharmaceutical intermediates, enabling manufacturers to respond quickly to fluctuating market demands and urgent customer orders without compromising on quality standards.
- Scalability and Environmental Compliance: The synthetic route is inherently scalable, as demonstrated by the use of standard unit operations like reflux, extraction, and crystallization which are easily transferred from pilot plant to commercial scale. The absence of hazardous reagents and the use of recyclable solvents align with modern green chemistry principles, facilitating easier regulatory approval and environmental compliance. This alignment reduces the administrative burden on EHS teams and lowers the risk of production stoppages due to regulatory non-compliance, ensuring a continuous and sustainable supply of the active ingredient for global distribution networks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis pathway. They are derived from the specific experimental data and claims found within the patent documentation, providing clarity on reaction conditions, impurity profiles, and scalability potential. Understanding these details helps stakeholders make informed decisions about integrating this technology into their existing manufacturing portfolios.
Q: What are the key advantages of the alkaline cyclization method described in CN1092660C?
A: The alkaline cyclization method allows for the use of cleaner and more cost-effective raw materials compared to traditional routes. It operates under manageable temperature conditions (50-170°C) and utilizes common bases like sodium hydroxide or potassium tert-butoxide, significantly simplifying the reaction environment and reducing the need for exotic catalysts.
Q: How does the N-methylation step impact the overall purity of the Sildenafil product?
A: The N-methylation step is critical for converting the intermediate into the bioactive Sildenafil structure. By optimizing reagents such as methyl sulfate or formaldehyde/formic acid combinations, the process minimizes side reactions and impurities. The patent demonstrates that careful control of reaction time and temperature during this phase leads to high yields and facilitates easier downstream purification.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the route is designed with scalability in mind. The use of standard solvents like ethanol, methylene dichloride, and tetrahydrofuran, along with robust reaction conditions, makes it highly adaptable for industrial scale-up. The elimination of complex transition metal catalysts further enhances its viability for consistent, high-volume manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sildenafil Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating innovative patent technologies into reliable commercial realities. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Sildenafil produced meets the highest international standards. We understand that the transition from lab-scale synthesis to full-scale manufacturing requires deep technical expertise, and our team is dedicated to navigating these complexities to deliver superior results for our global partners.
We invite you to collaborate with us to unlock the full potential of this efficient synthesis route for your business. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can enhance your supply chain efficiency. Let us be your trusted partner in delivering high-quality pharmaceutical intermediates that drive your success in the global marketplace.
