Advanced Anhydrous Acylation Process for High-Purity Cefepime Antibiotic Manufacturing
Introduction to Next-Generation Cephalosporin Synthesis
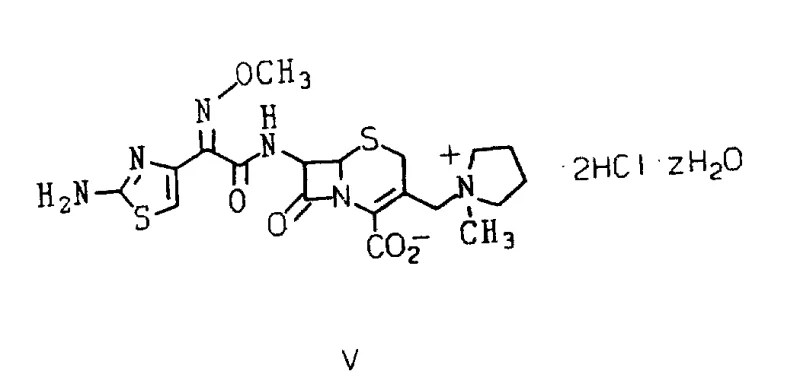
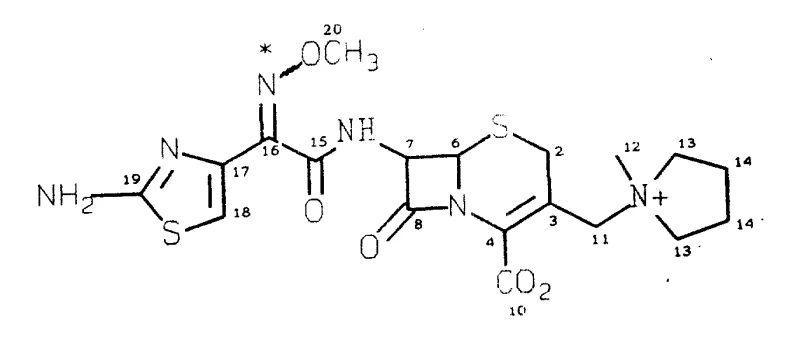
The pharmaceutical landscape constantly demands more efficient pathways for producing broad-spectrum antibiotics, particularly fourth-generation cephalosporins like Cefepime. Patent CN1070398A introduces a transformative anhydrous acylation process that addresses long-standing challenges in stereochemical control and product stability. This technology enables the preparation of Cefepime dihydrochloride hydrate that is substantially free of undesirable trans-isomers and delta-2 isomers, which are critical quality attributes for therapeutic efficacy. By bypassing traditional aqueous acylation methods that require complex protecting group strategies, this innovation offers a direct route to stable crystalline salts. For global supply chains, this represents a significant opportunity to enhance the reliability of antibiotic intermediate sourcing while reducing the environmental footprint associated with multi-step purification processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Cefepime and related cephalosporins has relied heavily on aqueous acylation techniques that introduce significant inefficiencies into the manufacturing workflow. Traditional methods often necessitate the use of amino protecting groups on the thiazole ring to prevent side reactions, followed by rigorous deblocking steps that add time and cost. Furthermore, aqueous processes frequently yield zwitterionic forms of the antibiotic that are thermally unstable at room temperature, requiring immediate conversion or specialized storage conditions. A major bottleneck in these legacy routes is the reliance on chromatography to separate the desired cis-isomer from the trans-isomer and delta-2 isomers, a technique that is notoriously difficult to scale for commercial production. The use of harsh activating agents like phosphorus pentachloride in older protocols often exacerbates isomerization issues, leading to lower overall yields and increased waste generation.
The Novel Approach
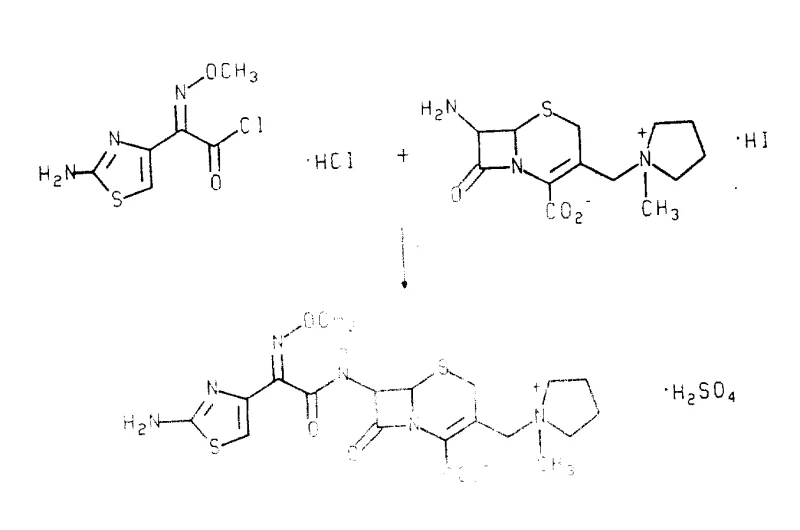
The innovative methodology described in the patent data circumvents these obstacles through a meticulously controlled anhydrous acylation strategy. By utilizing a specific cis-isomer of 2-(2-aminothiazol-4-yl)-2-methoxyiminoacetyl chloride hydrochloride, the process achieves high stereochemical fidelity without the need for protecting groups. The reaction conditions are optimized to maintain the integrity of the methoxyimino configuration, employing oxalyl chloride with a precise molar ratio of dimethyl formamide as a catalyst at sub-zero temperatures. This approach allows for the direct formation of the stable Cefepime dihydrochloride hydrate from the reaction mixture, eliminating the need for chromatographic purification. The result is a streamlined workflow that produces a temperature-stable crystalline product suitable for long-term storage and distribution.
Mechanistic Insights into Stereochemical Control and Isomer Suppression
The core technical breakthrough of this process lies in the precise management of reaction parameters to suppress the formation of trans-isomers and delta-2 isomers. The selection of the chlorinating agent is paramount; while phosphorus pentachloride is common in the industry, it promotes isomerization of the methoxyimino group. In contrast, the use of oxalyl chloride combined with a slight molar excess of dimethyl formamide generates the active Vilsmeier-Haack reagent in situ under mild conditions. This system activates the carboxylic acid without inducing the geometric isomerization that plagues other methods. Additionally, the silylation of the cephalosporin nucleus is conducted under strictly non-alkaline conditions using reagents like trimethylchlorosilane and hexamethyldisilazane. This prevents the base-catalyzed migration of the double bond from the delta-3 position to the delta-2 position, preserving the biological activity of the beta-lactam ring.
Impurity control is further enhanced by the physical properties of the intermediates and final products. The key acid chloride intermediate can be isolated as a stable crystalline solid, allowing for quality verification before the acylation step. During the final coupling, the reaction mixture is quenched with water to form a biphasic system, where the antibiotic partitions into the aqueous phase. Subsequent acidification and the addition of water-miscible organic solvents like acetone induce the crystallization of the dihydrochloride salt. This crystallization step acts as a final purification barrier, excluding residual trans-isomers and unreacted starting materials from the crystal lattice. The distinct nuclear magnetic resonance signatures of the cis and trans isomers, particularly the chemical shift of the thiazole ring proton, confirm the high purity of the final material.
How to Synthesize Cefepime Dihydrochloride Hydrate Efficiently
Implementing this synthesis route requires careful attention to temperature control and reagent stoichiometry to maximize yield and purity. The process begins with the activation of the side chain acid in an inert solvent such as methylene dichloride or acetonitrile, maintaining temperatures below -10°C to prevent degradation. Following the preparation of the silylated cephalosporin derivative, the coupling reaction is performed with slow addition of the acid chloride to manage exotherms. The detailed standardized synthesis steps, including specific molar equivalents and workup procedures, are outlined in the structured guide below.
- Prepare the cis-isomer of 2-(2-aminothiazol-4-yl)-2-methoxyiminoacetyl chloride hydrochloride using oxalyl chloride and controlled DMF catalysis in methylene dichloride at low temperatures.
- Generate the silylated derivative of 7-amino-3-[(1-methyl-1-pyrrolidinyl)methyl]ceph-3-em-4-carboxylate hydriodide using trimethylchlorosilane and hexamethyldisilazane under non-alkaline conditions.
- Couple the acid chloride with the silylated cephalosporin in an inert solvent, followed by aqueous workup and crystallization using acetone and hydrochloric acid to isolate the stable dihydrochloride hydrate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this anhydrous acylation technology translates into tangible operational improvements and risk mitigation. The elimination of chromatography steps significantly reduces the consumption of silica gel and organic solvents, leading to a drastic simplification of the waste treatment process. By removing the need for amino protecting groups, the total number of synthetic steps is reduced, which inherently lowers the cost of goods sold and shortens the production cycle time. The ability to isolate stable crystalline intermediates allows for flexible inventory management, decoupling the production of the side chain from the final coupling step. This flexibility enhances supply chain resilience, enabling manufacturers to respond more rapidly to fluctuations in market demand for broad-spectrum antibiotics.
- Cost Reduction in Manufacturing: The process eliminates expensive and toxic reagents such as phosphorus pentachloride and removes the need for complex protection-deprotection sequences. This reduction in material complexity directly lowers raw material costs and minimizes the labor hours required for process monitoring. Furthermore, the avoidance of chromatographic purification removes a major bottleneck in batch processing, allowing for larger batch sizes and improved equipment utilization rates. The overall efficiency gain results in substantial cost savings per kilogram of active pharmaceutical ingredient produced.
- Enhanced Supply Chain Reliability: The stability of the crystalline Cefepime dihydrochloride dihydrate ensures that the product can withstand varying transportation and storage conditions without degradation. Unlike zwitterionic forms that require strict temperature control, this salt form maintains its potency and crystal structure even under elevated temperatures. This robustness reduces the risk of spoilage during logistics and extends the shelf life of the inventory. Suppliers can therefore offer more reliable delivery schedules and reduce the safety stock levels required to buffer against quality failures.
- Scalability and Environmental Compliance: The use of common industrial solvents like methylene dichloride and acetone facilitates easy scale-up from pilot plant to commercial manufacturing volumes. The process generates fewer hazardous by-products compared to traditional methods, simplifying compliance with increasingly stringent environmental regulations. The straightforward workup procedure, involving simple phase separation and crystallization, is highly amenable to continuous processing technologies. This scalability ensures that the supply of high-purity antibiotic intermediates can be expanded rapidly to meet global health needs without compromising on quality standards.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this patented synthesis method. These answers are derived directly from the experimental data and comparative examples provided in the patent documentation. They highlight the specific advantages of the anhydrous route over prior art methods in terms of isomer control and product stability.
Q: How does this process control cis/trans isomerism in the side chain?
A: The process utilizes a specific chlorination method involving oxalyl chloride with a controlled molar excess of dimethyl formamide (DMF) at temperatures below -10°C. This prevents the isomerization often caused by phosphorus pentachloride, ensuring the formation of the biologically active cis-isomer substantially free of the trans-isomer.
Q: Why is the anhydrous acylation method superior to aqueous methods?
A: Anhydrous conditions eliminate the need for amino protecting groups and subsequent deblocking steps. This simplifies the workflow, avoids the instability associated with zwitterionic forms found in aqueous processes, and allows for direct crystallization of the stable dihydrochloride hydrate without chromatography.
Q: What are the storage stability benefits of the resulting Cefepime salt?
A: The resulting crystalline Cefepime dihydrochloride dihydrate demonstrates high thermal stability, retaining its crystal morphology and water content even after prolonged exposure to 70°C or vacuum drying. This contrasts with previous forms that were prone to losing water of crystallization or degrading at room temperature.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefepime Intermediate Supplier
At NINGBO INNO PHARMCHEM, we leverage advanced proprietary technologies like the anhydrous acylation process to deliver superior pharmaceutical intermediates to the global market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that every batch meets stringent purity specifications. We operate rigorous QC labs equipped with state-of-the-art analytical instruments to verify the absence of trans-isomers and delta-2 impurities in our Cefepime derivatives. Our commitment to quality assurance guarantees that your downstream manufacturing processes proceed without interruption or yield loss due to raw material variability.
We invite you to collaborate with us to optimize your antibiotic supply chain and achieve significant operational efficiencies. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production volumes. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our high-purity intermediates can enhance your final product quality. Let us be your partner in delivering life-saving medications to patients worldwide through superior chemical manufacturing excellence.
