Advanced Aqueous Acylation Process for High-Purity Cefepime Dihydrochloride Manufacturing
The pharmaceutical industry continuously seeks robust synthetic pathways for fourth-generation cephalosporins, particularly cefepime, due to its broad-spectrum efficacy against resistant bacterial strains. Patent CN1140714A introduces a transformative aqueous acylation process that addresses long-standing challenges in stereochemical control and process efficiency. This innovation centers on the utilization of a stable crystalline cis-isomer of 2-(2-aminothiazol-4-yl)-2-methoxyiminoacetyl chloride hydrochloride, which serves as a pivotal intermediate for producing cefepime dihydrochloride hydrate. The significance of this technology lies in its ability to yield the final antibiotic substantially free of undesirable trans-isomers and Delta-2 isomers, thereby enhancing the therapeutic profile and safety of the active pharmaceutical ingredient. By bypassing complex protection and deprotection sequences, this method offers a streamlined route that aligns with modern green chemistry principles and cost-effective manufacturing strategies.
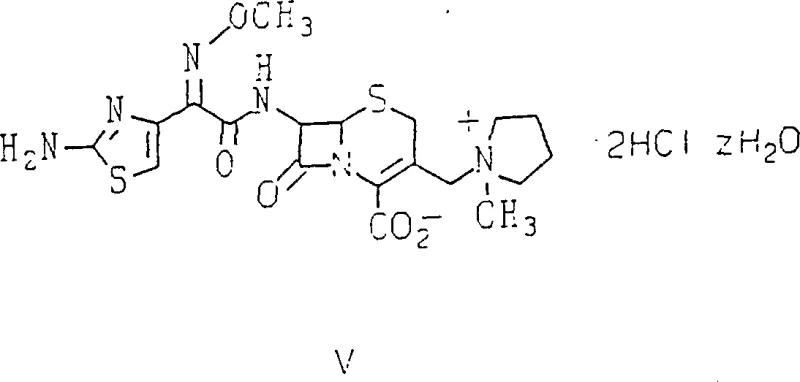
For R&D directors and process chemists, the structural integrity of the final product is paramount. The patent explicitly defines the target molecule, cefepime dihydrochloride hydrate, represented by Formula V, which demonstrates the precise arrangement of the methoxyimino side chain attached to the cephalosporin nucleus. Achieving this specific configuration without significant isomeric impurities has historically required rigorous purification steps. The disclosed process ensures that the geometric (Z) configuration of the methoxyimino group is maintained throughout the synthesis, directly impacting the biological activity and stability of the drug substance. This level of control is critical for meeting stringent regulatory specifications regarding impurity profiles in injectable antibiotics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cefepime and related cephalosporins has been plagued by inefficiencies associated with the handling of the aminothiazole side chain. Conventional methodologies, such as those described in earlier patents like US 4,406,899, typically necessitated the use of amino protecting groups to prevent side reactions during the activation of the carboxylic acid. These protecting groups, while chemically necessary in older routes, introduce additional synthetic steps for installation and subsequent removal, which inherently lowers overall yield and increases material costs. Furthermore, traditional activation methods often relied on complex organic leaving groups or active esters that required chromatographic purification to separate the desired cis-isomer from the pharmacologically inactive or less active trans-isomer. The reliance on column chromatography is particularly detrimental in a commercial setting, as it limits batch sizes, increases solvent consumption, and complicates waste management, making the process less scalable for industrial production.
The Novel Approach
In stark contrast to prior art, the novel approach detailed in CN1140714A leverages a specifically prepared stable crystalline cis-isomer of the acyl chloride hydrochloride, depicted as Formula III. This intermediate allows for direct N-acylation of the cephalosporin nucleus in an aqueous environment without the need for amino protection. The use of this unprotected cis-isomer eliminates the chemical steps associated with blocking and deblocking the amine functionality, thereby drastically simplifying the synthetic sequence. Moreover, the process utilizes a simple chloride ion as the leaving group, which avoids the toxicity and environmental concerns associated with leaving groups like 2-mercaptobenzothiazole. The ability to isolate this intermediate as a stable crystalline salt ensures that the stereochemical integrity is preserved prior to the coupling reaction, providing a reliable starting material for high-purity API synthesis.
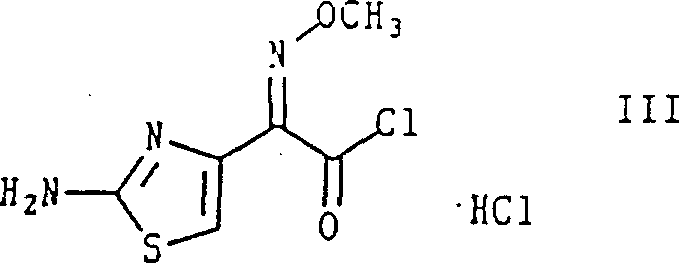
The strategic advantage of employing Formula III extends beyond mere step reduction. By establishing a method to prepare this acid chloride hydrochloride substantially free of the trans-isomer, the subsequent acylation reaction naturally favors the formation of the desired cefepime stereoisomer. This intrinsic stereocontrol reduces the burden on downstream purification processes, allowing manufacturers to achieve high-purity standards through crystallization rather than chromatography. For procurement and supply chain teams, this translates to a more predictable manufacturing timeline and reduced dependency on specialized purification resins or columns. The robustness of this intermediate also facilitates better inventory management, as the stable crystalline form can be stored and transported with greater confidence compared to unstable oily intermediates often encountered in traditional cephalosporin synthesis.
Mechanistic Insights into Stereocontrolled Aqueous Acylation
The core of this technological advancement lies in the precise control of reaction conditions during the formation and utilization of the acyl chloride intermediate. The conversion of the cis-acid to the corresponding acid chloride hydrochloride is achieved using oxalyl chloride in combination with dimethylformamide (DMF) within an inert organic solvent. Critical to this transformation is the maintenance of low temperatures, specifically ranging from approximately -10°C to -40°C, preferably in dichloromethane. The molar ratio of DMF to oxalyl chloride is meticulously managed; the patent reveals that using a molar excess of DMF relative to oxalyl chloride, or conversely insufficient amounts, can lead to isomerization or instability. This delicate balance prevents the formation of the unwanted trans-isomer and avoids chlorination at the 5-position of the thiazole ring, which would otherwise generate difficult-to-remove impurities. The resulting intermediate retains the cis-configuration essential for the biological efficacy of the final antibiotic.
Following the preparation of the activated intermediate, the acylation of the cephalosporin nucleus occurs in a mixed aqueous-organic solvent system. The reaction pH is tightly controlled between 5.0 and 7.5, typically using bases such as triethylamine or N-methylmorpholine, to facilitate the nucleophilic attack of the 7-amino group on the acyl chloride while minimizing hydrolysis of the beta-lactam ring. This aqueous acylation step is performed at temperatures between -10°C and -40°C to further suppress side reactions. A key mechanistic benefit of this process is the direct crystallization of the cefepime dihydrochloride hydrate from the reaction mixture upon acidification. Unlike previous methods that required isolation of zwitterionic forms or sulfate salts followed by conversion, this route allows for the direct precipitation of the stable dihydrochloride hydrate. This direct crystallization not only simplifies the workflow but also ensures that the Delta-3 double bond of the cephalosporin nucleus is preserved, preventing the formation of the Delta-2 isomer which is a common degradation product in cephalosporin chemistry.
How to Synthesize Cefepime Dihydrochloride Efficiently
The synthesis of cefepime dihydrochloride via this patented route involves a sequence of highly controlled chemical transformations designed to maximize yield and stereochemical purity. The process begins with the conversion of the cis-2-(2-aminothiazol-4-yl)-2-methoxyiminoacetic acid into its corresponding acid chloride hydrochloride salt under strictly anhydrous and low-temperature conditions. This activated intermediate is then immediately employed in an aqueous acylation reaction with 7-amino-3-[(1-methyl-1-pyrrolidinyl)methyl]ceph-3-ene-4-carboxylate. The reaction environment is carefully buffered to maintain optimal pH levels that favor amide bond formation while protecting the sensitive beta-lactam core. Detailed standard operating procedures for scaling this synthesis from laboratory to commercial production require precise adherence to temperature profiles and reagent addition rates to ensure consistent quality.
- Preparation of the stable crystalline cis-isomer acid chloride hydrochloride by reacting the cis-acid with oxalyl chloride and dimethylformamide at controlled low temperatures.
- N-acylation of 7-amino-3-[(1-methyl-1-pyrrolidinyl)methyl]ceph-3-ene-4-carboxylate in an aqueous organic solvent system while maintaining pH between 5.0 and 7.5.
- Acidification and crystallization of the final cefepime dihydrochloride hydrate directly from the reaction mixture without intermediate isolation of zwitterionic forms.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis route offers substantial benefits for organizations focused on cost optimization and supply chain resilience. The elimination of amino protecting groups and the associated deprotection steps results in a significant reduction in raw material consumption and waste generation. By removing the necessity for chromatographic purification, manufacturers can avoid the high costs associated with silica gel, resins, and large volumes of organic solvents, leading to a more economically viable production model. This streamlined process enhances the overall throughput of the manufacturing facility, allowing for larger batch sizes and more efficient use of reactor capacity. For procurement managers, this translates into a more competitive cost structure for the final API, enabling better pricing strategies in the global market.
- Cost Reduction in Manufacturing: The removal of protecting group chemistry and chromatographic purification steps fundamentally alters the cost equation for cefepime production. Traditional methods incur significant expenses related to the purchase of protecting reagents, the solvents required for their removal, and the disposal of hazardous waste generated during these extra steps. By utilizing the stable cis-isomer acid chloride directly, the process bypasses these cost centers entirely. Furthermore, the use of simple chloride as a leaving group avoids the procurement of expensive and potentially regulated activated esters or toxic leaving groups. This simplification of the bill of materials directly contributes to lower variable costs per kilogram of produced antibiotic, enhancing profit margins without compromising on quality standards.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the complexity of multi-step syntheses involving unstable intermediates. The ability to isolate the key acyl chloride intermediate as a stable crystalline salt provides a strategic buffer in the supply chain. This stability allows for the intermediate to be manufactured in advance, quality tested, and stored, decoupling the production of the side chain from the final coupling reaction. This flexibility reduces the risk of batch failures due to immediate intermediate degradation and allows for more robust production scheduling. Additionally, the avoidance of specialized chromatography columns reduces dependency on specific consumables that may face supply shortages, ensuring a more reliable and uninterrupted manufacturing flow for critical antibiotic supplies.
- Scalability and Environmental Compliance: Scaling chemical processes often exposes limitations in heat transfer and mixing that are not apparent at the laboratory scale. The aqueous acylation method described is inherently more scalable than non-aqueous alternatives because water acts as an excellent heat sink, facilitating temperature control during exothermic reactions. The reduction in organic solvent usage, particularly the elimination of solvents required for chromatography, significantly lowers the environmental footprint of the manufacturing process. This aligns with increasingly stringent environmental regulations and corporate sustainability goals. The process generates less hazardous waste, simplifying disposal and reducing the regulatory burden on the manufacturing site, which is a crucial factor for long-term operational viability in the pharmaceutical sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology. Understanding these aspects is vital for stakeholders evaluating the feasibility of adopting this route for commercial production. The answers are derived directly from the technical disclosures within the patent documentation, ensuring accuracy and relevance to the specific chemical challenges associated with cephalosporin manufacturing.
Q: What is the primary advantage of using the cis-isomer intermediate described in CN1140714A?
A: The primary advantage is the ability to produce cefepime dihydrochloride hydrate substantially free of trans-isomers and Delta-2 isomers without requiring chromatographic purification or amino protecting groups, significantly simplifying the manufacturing workflow.
Q: How does this process improve supply chain reliability for cephalosporin antibiotics?
A: By utilizing a stable crystalline salt of the acyl chloride intermediate, the process allows for safer storage and transport of the reactive species, reducing the risk of degradation and ensuring consistent quality for downstream acylation steps.
Q: Does this method eliminate the need for toxic leaving groups?
A: Yes, the method utilizes a simple chloride ion as the leaving group, avoiding the use of potentially toxic organic leaving groups such as 2-mercaptobenzothiazole often found in conventional activated ester methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefepime Dihydrochloride Supplier
The technical innovations outlined in CN1140714A represent a significant leap forward in the manufacturing of fourth-generation cephalosporins, offering a pathway to higher purity and greater efficiency. NINGBO INNO PHARMCHEM stands at the forefront of translating such complex chemical methodologies into commercial reality. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in large-scale operations. Our facilities are equipped with rigorous QC labs and advanced process control systems capable of maintaining the stringent purity specifications required for injectable antibiotics, guaranteeing that every batch meets the highest international standards.
We invite pharmaceutical companies and procurement leaders to engage with our technical procurement team to explore how this optimized synthesis route can benefit your supply chain. By leveraging our expertise, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to request specific COA data and route feasibility assessments to validate the potential of this technology for your portfolio. Partnering with us ensures not only access to high-quality cefepime dihydrochloride but also a collaborative relationship focused on continuous improvement and supply security in the dynamic global pharmaceutical market.
