Advanced Synthesis of Aromatic Diether Dianhydride for Commercial Polyetherimide Production
The landscape of high-performance polymer manufacturing is undergoing a significant transformation driven by the need for more efficient monomer synthesis. Patent CN110483454B introduces a groundbreaking preparation method for aromatic diether dianhydride, a critical precursor for polyetherimide (PEI) and advanced thermosetting resins. This technology addresses long-standing inefficiencies in traditional synthesis routes by integrating a streamlined three-step process that eliminates cumbersome intermediate isolations. For R&D directors and procurement specialists, this represents a pivotal shift towards higher yield and reduced operational complexity. The method leverages a unique combination of nucleophilic substitution and hydrolysis conditions that allow for direct progression between stages without the need for extensive purification of intermediates. By optimizing solvent systems and reaction parameters, this approach ensures that the final product meets stringent purity specifications required for aerospace and electronic applications. The implications for industrial scale-up are profound, offering a pathway to reduce both production time and environmental footprint while maintaining exceptional product quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aromatic diether dianhydrides has been plagued by inefficient multi-step protocols that hinder commercial viability. Traditional methods, such as those disclosed in US3957826, typically require the preparation of diphenol alkali metal salts under strictly anhydrous conditions, which demands significant energy and specialized equipment. These processes often rely on toxic solvents like dichloromethane, necessitating complex containment systems to prevent environmental contamination and ensuring worker safety. Furthermore, the need to isolate and purify intermediate products after every reaction step introduces substantial material loss and extends the overall production cycle. For instance, older techniques have reported total yields as low as 57.6%, primarily due to the accumulation of losses during repeated filtration, extraction, and evaporation stages. The reliance on phase transfer catalysts and the difficulty in removing high-boiling solvents further complicate the downstream processing, making cost reduction in electronic chemical manufacturing a challenging endeavor for many producers.
The Novel Approach
In stark contrast, the novel approach detailed in the patent data simplifies the workflow into a highly efficient sequence that maximizes atom economy and operational throughput. The process begins with a substitution reaction in a mixed solvent system comprising a polar organic solvent and a water-carrying agent, which facilitates the removal of byproduct water and drives the reaction to completion. A key innovation is the ability to proceed directly to hydrolysis without isolating the N-substituted dietherimide intermediate, thereby saving considerable time and resources. The subsequent cyclization step utilizes a mixture of acetic acid and acetic anhydride, which not only acts as the dehydrating agent but also allows for easy recovery and reuse of solvents. This methodology drastically reduces the number of unit operations required, leading to substantial cost savings and a more robust supply chain. By avoiding the use of highly toxic chlorinated solvents and minimizing waste generation, this route aligns perfectly with modern environmental compliance standards while delivering superior yields.
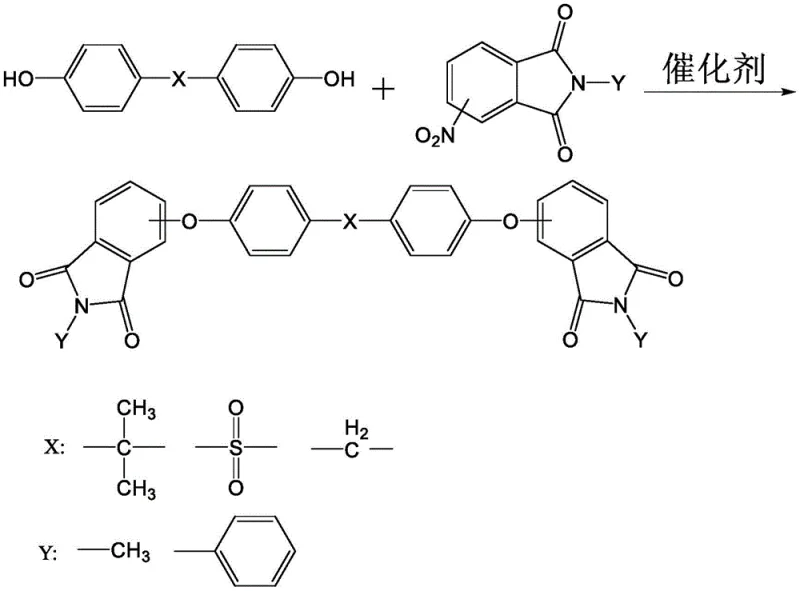
Mechanistic Insights into Nucleophilic Substitution and Cyclization
The core of this synthesis lies in the precise control of nucleophilic substitution and subsequent hydrolysis mechanisms. In the first stage, the bisphenol compound reacts with N-substituted phthalimide in the presence of an alkali metal carbonate catalyst. The use of a water-carrying agent like toluene in conjunction with polar solvents such as DMF or DMAC is critical for shifting the equilibrium towards the formation of the N-substituted dietherimide. This step is designed to be exhaustive, ensuring that the reaction mixture contains minimal unreacted starting materials that could interfere with downstream processing. The choice of catalyst, specifically alkali metal carbonates or bicarbonates, provides a mild yet effective basic environment that promotes the nucleophilic attack of the phenoxide ion on the phthalimide ring without causing excessive degradation. This careful balance of reactivity ensures that the intermediate formed is of sufficient quality to proceed directly to the next stage without purification.
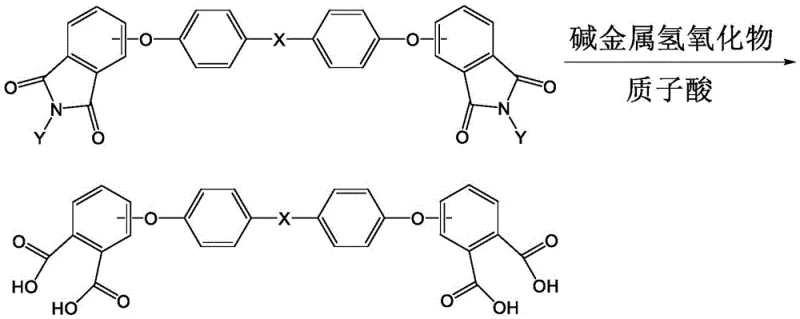
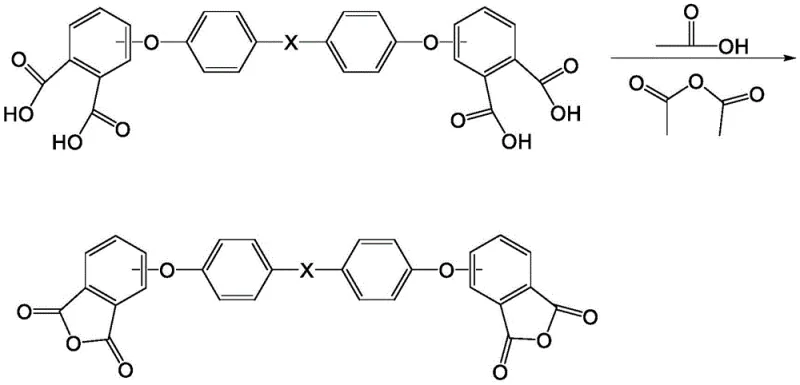
Following the substitution, the hydrolysis step converts the imide groups into carboxylic acid groups using an excess of alkali metal hydroxide at elevated temperatures ranging from 140°C to 200°C. A remarkable feature of this mechanism is the behavior of the inorganic salts generated during acidification. Instead of requiring separation, the diether tetraacid precipitates in a form that coats the inorganic salts, effectively isolating itself from impurities that could hinder the final cyclization. This phenomenon eliminates the need for a dedicated purification step for the tetraacid, which is a major bottleneck in conventional methods. The final cyclization involves heating the dried solid in acetic acid and acetic anhydride, where the carboxylic acid groups undergo dehydration to form the anhydride rings. The recrystallization from acetonitrile and toluene further refines the product, removing any remaining soluble impurities and ensuring a final purity greater than 99.7%.
How to Synthesize Aromatic Diether Dianhydride Efficiently
Implementing this synthesis route requires careful attention to solvent ratios and temperature controls to maximize efficiency and yield. The process is designed to be scalable, moving seamlessly from laboratory validation to commercial production with minimal adjustment. Detailed standard operating procedures are essential to maintain the precise molar ratios of bisphenol to phthalimide and catalyst, typically optimized around 1:2:2.4 for best results. The following guide outlines the critical operational parameters necessary to replicate the high success rates observed in the patent examples.
- Perform nucleophilic substitution between bisphenol and N-substituted phthalimide in a polar solvent with a water-carrying agent to form N-substituted dietherimide.
- Hydrolyze the intermediate using alkali metal hydroxide and water, followed by acidification to precipitate the diether tetraacid without intermediate purification.
- Dehydrate and cyclize the solid in acetic acid and acetic anhydride, followed by recrystallization to achieve purity greater than 99.7%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis method offers tangible benefits that extend beyond simple chemical yield. The elimination of intermediate isolation steps translates directly into reduced labor costs and lower energy consumption, as fewer heating and cooling cycles are required. The ability to recover and reuse solvents like DMF and toluene significantly lowers the raw material expenditure per kilogram of product, contributing to substantial cost savings over the lifecycle of the production campaign. Furthermore, the avoidance of toxic chlorinated solvents reduces the regulatory burden and waste disposal costs associated with hazardous material handling. This streamlined process enhances supply chain reliability by shortening the manufacturing lead time, allowing for faster response to market demands and reducing the risk of production bottlenecks.
- Cost Reduction in Manufacturing: The simplified process flow eliminates the need for multiple filtration and drying stages, which are often resource-intensive and time-consuming. By removing the requirement for intermediate purification, the consumption of auxiliary materials and solvents is drastically reduced, leading to a more economical production model. The high yield achieved through this method ensures that raw material utilization is optimized, minimizing waste and maximizing the output from each batch. Additionally, the recovery of solvents allows for a closed-loop system that further drives down operational expenses without compromising product quality.
- Enhanced Supply Chain Reliability: The robustness of this synthesis route means that production schedules are less susceptible to delays caused by complex purification failures or equipment downtime. The use of commercially available and stable reagents ensures a consistent supply of inputs, reducing the risk of shortages that can plague more exotic chemical processes. Shorter cycle times enable manufacturers to maintain lower inventory levels while still meeting delivery commitments, improving cash flow and operational agility. This reliability is crucial for maintaining long-term contracts with downstream polymer producers who depend on consistent monomer supply.
- Scalability and Environmental Compliance: Scaling this process from pilot to commercial volumes is straightforward due to the absence of sensitive anhydrous conditions and the use of standard reactor equipment. The reduction in hazardous waste generation aligns with increasingly strict environmental regulations, making it easier to obtain and maintain operating permits. The ability to operate with lower toxicity solvents improves workplace safety and reduces the need for specialized containment infrastructure. This environmental compatibility not only mitigates risk but also enhances the brand reputation of the manufacturer as a sustainable partner in the global supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this aromatic diether dianhydride synthesis method. These answers are derived directly from the patent data and are intended to provide clarity for technical teams evaluating the feasibility of this route for their specific applications. Understanding these details is essential for making informed decisions about process adoption and supply chain integration.
Q: How does this method improve yield compared to conventional processes?
A: Conventional methods often suffer from low yields around 57.6% due to complex isolation steps and toxic solvents. This novel approach achieves yields exceeding 90% by eliminating intermediate purification and utilizing efficient solvent systems.
Q: Is intermediate purification required between reaction steps?
A: No, the process is designed to minimize handling. The N-substituted dietherimide does not require separation before hydrolysis, and the resulting inorganic salts do not interfere with the final cyclization, significantly streamlining production.
Q: What purity levels can be achieved with this synthesis route?
A: Through optimized recrystallization using acetonitrile and toluene, the final aromatic diether dianhydride product consistently achieves HPLC purity levels greater than 99.7%, suitable for high-performance polymer applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aromatic Diether Dianhydride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity monomers play in the performance of advanced engineering plastics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory success to industrial reality is seamless. We are committed to delivering aromatic diether dianhydride that meets stringent purity specifications, supported by our rigorous QC labs that verify every batch against the highest industry standards. Our expertise in process optimization allows us to adapt this patented methodology to meet specific customer requirements while maintaining the efficiency and cost-effectiveness inherent in the design.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your production volumes. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can support your long-term strategic goals. Let us be your partner in achieving superior material performance and operational excellence.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
