Advanced Synthesis of Aromatic Diether Dianhydride for High-Performance Polymer Manufacturing
Advanced Synthesis of Aromatic Diether Dianhydride for High-Performance Polymer Manufacturing
The chemical industry is constantly seeking more efficient pathways to produce high-performance polymer monomers, and the recent disclosure in patent CN112321550B presents a transformative approach to synthesizing aromatic diether dianhydride. This compound serves as a critical monomer for polyetherimide (PEI) production and acts as a vital curing agent for thermosetting resins and high-temperature adhesives. The disclosed methodology fundamentally shifts the synthetic paradigm by employing isobutene or tertiary butanol as a carboxyl protecting group, thereby circumventing the notoriously difficult hydrolysis steps associated with traditional phthalimide routes. By integrating a mild acid deprotection strategy followed by direct thermal cyclization, the entire reaction sequence is compressed into a remarkably short cycle time of approximately 21 hours. This innovation not only addresses the longstanding issue of prolonged reaction times but also significantly enhances the safety profile and overall production efficiency for manufacturers aiming to scale up complex electronic and polymer materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of aromatic diether dianhydride has been plagued by inefficient multi-step sequences that rely heavily on harsh reaction conditions and excessive reagent consumption. The traditional route typically involves reacting nitrobenzene anhydride or chlorophthalic anhydride with amines to form phthalimides, followed by a substitution reaction with bisphenol disodium salt. The most critical bottleneck occurs during the hydrolysis of the resulting diether diphthalimide, which requires strong alkali treatment under high pressure at temperatures around 120°C for over 30 hours. Due to significant steric hindrance, this hydrolysis demands more than ten equivalents of sodium hydroxide, leading to massive consumption of strong acids in the subsequent acidification step to recover the tetra-carboxylic acid. Furthermore, the presence of sensitive functional groups like thioethers or ketones often leads to undesirable side reactions under these aggressive alkaline conditions, resulting in reduced yields and complex purification challenges that hinder commercial viability.
The Novel Approach
In stark contrast to the legacy methods, the novel preparation technique described in the patent introduces a sophisticated protection-deprotection strategy that elegantly bypasses the need for harsh hydrolysis. By initially protecting the carboxyl groups of chloro- or nitrophthalic acid with tert-butyl esters using isobutene or tertiary butanol, the synthesis creates a robust intermediate that withstands the subsequent etherification conditions. Following the coupling with bisphenol disodium salt, the tert-butyl groups are removed using only a small amount of acid treatment rather than strong alkali, yielding the diether tetracarboxylic acid under much milder conditions. The final step involves directly heating the solid tetracarboxylic acid to form the anhydride without the need for acetic anhydride dehydration agents. This streamlined process eliminates the high-pressure hydrolysis step entirely, reduces the total reaction time to about 21 hours, and simplifies the workup procedure, making it highly attractive for large-scale manufacturing environments.
Mechanistic Insights into Tert-Butyl Protection and Thermal Cyclization
The core of this technological breakthrough lies in the strategic use of the tert-butyl group as a temporary mask for the carboxylic acid functionalities, which fundamentally alters the reaction landscape. As illustrated in the reaction mechanism, the process begins with the acid-catalyzed esterification of phthalic acid derivatives, creating a sterically bulky yet chemically stable di-tert-butyl ester. This protected intermediate then undergoes nucleophilic aromatic substitution with the bisphenol disodium salt in a dipolar aprotic solvent, forming the carbon-oxygen bonds necessary for the diether backbone. Crucially, the tert-butyl ester is labile to acid but stable to base, allowing for selective deprotection using mild acids like acetic acid or dilute hydrochloric acid at moderate temperatures. This selectivity prevents the degradation of the newly formed ether linkages and avoids the side reactions common in strong alkaline environments, ensuring a cleaner reaction profile and higher purity of the intermediate tetra-acid before the final cyclization step.
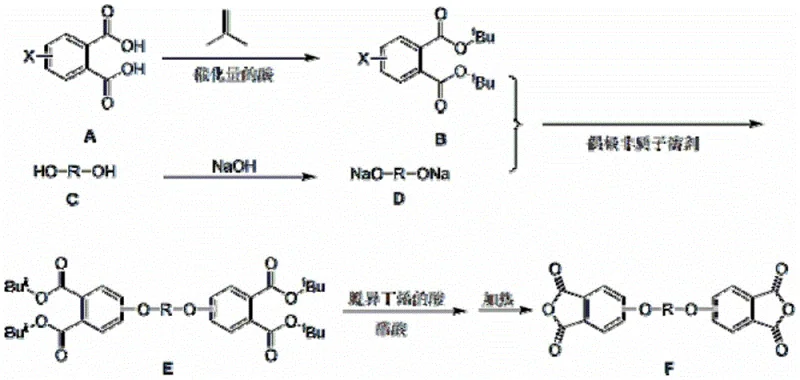
Following the successful removal of the protecting groups, the resulting aromatic diether tetracarboxylic acid undergoes a thermal dehydration process to close the anhydride rings. This solid-state or melt-phase cyclization is driven by heating the pure acid to temperatures between 180°C and 260°C, depending on the specific substrate structure. The absence of water or dehydration agents like acetic anhydride in this final step is a significant advantage, as it eliminates the need for solvent removal and reduces the generation of acetic acid waste. The mechanism ensures that the rigid aromatic backbone is preserved while forming the cyclic anhydride moieties essential for polymerization. By avoiding the formation of phenolic ester byproducts that are difficult to purify, as seen in some direct etherification attempts, this method guarantees a high-purity final product suitable for demanding applications in aerospace and electronics where material consistency is paramount.
How to Synthesize Aromatic Diether Dianhydride Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing high-quality aromatic diether dianhydride with minimal operational complexity. The process is designed to be robust, utilizing readily available starting materials such as chlorophthalic acid or nitrophthalic acid and common bisphenols. The initial protection step is conducted in solvents like toluene or xylene with catalytic acid, followed by a straightforward workup to prepare the reactive intermediate. Subsequent etherification is performed in polar aprotic solvents like dimethylacetamide or DMSO, ensuring high conversion rates. The deprotection and cyclization steps are equally manageable, requiring standard heating and filtration equipment found in most fine chemical facilities. For detailed operational parameters and specific stoichiometric ratios, please refer to the standardized synthesis guide below.
- Protect chloro- or nitrophthalic acid using isobutene or tertiary butanol with catalytic acid to form the di-tert-butyl ester intermediate.
- React the protected intermediate with bisphenol disodium salt in a dipolar aprotic solvent at 140-170°C to form the diether tetra-ester.
- Remove the tert-butyl protecting groups using a small amount of acid (e.g., acetic acid/HCl) without strong alkali treatment to obtain the tetra-acid.
- Recrystallize the crude tetra-acid and heat the pure product to 180-260°C to induce thermal dehydration and cyclization into the final dianhydride.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this novel synthesis route offers substantial strategic benefits that directly impact the bottom line and operational reliability. The elimination of the high-pressure, high-temperature hydrolysis step removes a major bottleneck in production scheduling, allowing for faster batch turnover and increased facility throughput. By replacing the requirement for massive quantities of sodium hydroxide and subsequent neutralization acids with a mild acid deprotection step, the process significantly reduces the consumption of bulk chemicals and lowers the cost associated with raw material procurement. Furthermore, the simplified purification process, which avoids difficult extractions and high-boiling solvent removals, translates into reduced utility costs and shorter processing times. These efficiencies collectively contribute to a more resilient supply chain capable of meeting tight delivery schedules without compromising on product quality or safety standards.
- Cost Reduction in Manufacturing: The economic implications of this process are profound, primarily driven by the drastic reduction in reagent usage and waste treatment costs. Traditional methods consume over ten equivalents of caustic soda and equivalent amounts of mineral acids, generating large volumes of saline wastewater that require expensive treatment. By avoiding strong alkali treatment entirely and eliminating the need for acetic anhydride in the dehydration step, the new method achieves significant cost savings through reduced chemical spend and lower waste disposal fees. Additionally, the shorter reaction cycle of approximately 21 hours compared to over 30 hours for conventional routes means better asset utilization, allowing manufacturers to produce more batches per year with the same equipment infrastructure, thereby lowering the fixed cost per kilogram of the final product.
- Enhanced Supply Chain Reliability: Supply chain stability is often threatened by complex processes that are prone to failure or yield fluctuations, but this streamlined approach mitigates those risks effectively. The use of stable intermediates and mild reaction conditions reduces the likelihood of batch failures due to runaway reactions or incomplete hydrolysis, which are common pitfalls in the traditional high-pressure processes. The ability to use a wide range of bisphenol substrates, including those with sensitive linkages like sulfones or ketones, ensures that suppliers can flexibly source raw materials without being constrained by the limitations of harsh hydrolysis conditions. This flexibility, combined with the robust nature of the tert-butyl protection chemistry, ensures a consistent and reliable flow of high-purity dianhydride to downstream polymer manufacturers, securing the continuity of production for critical applications.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to industrial production often exposes hidden inefficiencies, yet this method is inherently designed for scalability. The avoidance of high-pressure reactors for hydrolysis simplifies the engineering requirements for large-scale vessels, reducing capital expenditure for new capacity expansion. Moreover, the environmental footprint is considerably smaller due to the reduced generation of hazardous waste and the elimination of volatile organic solvents in the final dehydration step. The process aligns well with increasingly stringent environmental regulations regarding wastewater discharge and solvent emissions, making it a future-proof choice for manufacturers aiming to maintain compliance while expanding their production capabilities for high-performance polymer additives and electronic materials.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced preparation method. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this technology outperforms legacy synthesis routes. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this process for their specific manufacturing needs and for procurement specialists assessing the long-term value proposition.
Q: How does the new preparation method reduce reaction time compared to traditional routes?
A: The traditional method involves a multi-step sequence including imide formation, high-pressure strong alkali hydrolysis (>30 hours), and acidification. The novel method utilizes a tert-butyl protection strategy that allows for mild acid deprotection and direct thermal cyclization, reducing the total process time to approximately 21 hours while eliminating the need for harsh hydrolysis conditions.
Q: What are the advantages regarding reagent consumption and waste generation?
A: Conventional processes require excessive amounts of strong alkali (over ten equivalents of sodium hydroxide) and strong acid for hydrolysis and neutralization, generating significant saline waste. This patented approach avoids strong alkali treatment entirely during deprotection, using only catalytic amounts of acid and mild conditions, thereby drastically reducing chemical consumption and simplifying wastewater treatment protocols.
Q: Can this method be applied to various bisphenol structures?
A: Yes, the method demonstrates broad substrate scope. It is compatible with various diphenol structures including bisphenol A, hydroquinone, and others containing linkages such as -O-, -S-, -SO2-, or -CO-. The etherification step effectively accommodates these variations without the side reactions often seen in strong alkaline hydrolysis of sensitive functional groups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aromatic Diether Dianhydride Supplier
As the demand for high-performance polymers continues to surge in sectors ranging from aerospace to consumer electronics, securing a dependable source of critical monomers like aromatic diether dianhydride is more important than ever. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging deep technical expertise to deliver superior chemical solutions that meet the rigorous standards of global manufacturers. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can support your growth from pilot trials to full-scale industrial deployment. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of material performs consistently in your downstream polymerization processes.
We invite you to explore how our advanced manufacturing capabilities can optimize your supply chain and reduce your overall production costs. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing us to demonstrate precisely how our optimized synthesis of aromatic diether dianhydride can drive value and efficiency for your organization.
