Advanced 4-Step Synthesis of Levetiracetam for Commercial API Production
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of Levetiracetam has been hindered by inefficient synthetic routes that rely on cumbersome multi-step sequences. Traditional methodologies, as documented in prior art such as EP0165919B1, typically involve a six-step process starting from propionic aldehyde or butyric acid. These legacy routes are characterized by low overall yields, often stagnating around 4.7%, which drastically inflates the cost of goods sold (COGS) due to significant material attrition at each stage. Furthermore, conventional methods frequently necessitate the use of hazardous hydrogen cyanide (HCN) gas for the introduction of the nitrile group, posing severe safety risks and requiring expensive, corrosion-resistant reactor infrastructure to manage toxic emissions and ensure operator safety. The complexity of these older pathways also introduces multiple purification challenges, increasing the likelihood of impurity carryover and complicating regulatory compliance for API manufacturers.
The Novel Approach
In stark contrast, the technology disclosed in patent CN1288133C introduces a streamlined four-step synthetic strategy that fundamentally reengineers the production workflow for superior efficiency. By optimizing the reaction sequence to include a Strecker reaction, hydrolysis, chiral resolution, and cyclization, this novel approach eliminates two entire unit operations found in previous literature. The substitution of gaseous HCN with solid sodium cyanide (NaCN) not only mitigates safety hazards but also simplifies the handling and dosing of reagents in a standard batch reactor environment. This methodological shift facilitates a dramatic improvement in overall yield, elevating production efficiency to approximately 10%, which represents a more than twofold increase over traditional benchmarks. The integration of a robust diastereomer resolution step ensures high stereochemical purity early in the process, reducing the burden on downstream purification.
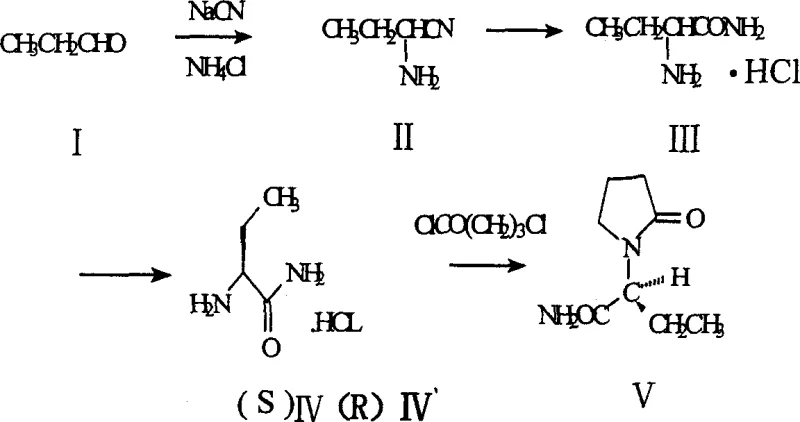
Mechanistic Insights into Strecker Reaction and Diastereomer Resolution
The core of this advanced synthesis lies in the precise execution of the Strecker reaction followed by a highly selective chiral resolution. Initially, propionaldehyde reacts with sodium cyanide and ammonium chloride to form alpha-aminobutyronitrile. This intermediate is subsequently hydrolyzed under acidic conditions, typically using hydrogen chloride in an alcohol solvent, to generate the racemic DL-alpha-aminobutanamide hydrochloride. The critical innovation occurs in the resolution phase, where the racemic amide is treated with a chiral resolving agent such as L-(+)-tartaric acid, (+)-mandelic acid, or (+)-oxysuccinic acid in an organic solvent like methanol or ethanol. Under alkaline conditions facilitated by bases such as triethylamine or sodium hydroxide, diastereomeric salts are formed. Due to the distinct differences in solubility between the (S)- and (R)-diastereomeric salts in the chosen solvent system, the desired (S)-enantiomer precipitates selectively while the (R)-isomer remains in the mother liquor. This physical separation mechanism is far more scalable and cost-effective than enzymatic kinetic resolution or preparative chiral chromatography.
Following the isolation of the optically pure (S)-alpha-aminobutanamide hydrochloride, the final cyclization step constructs the pyrrolidone ring essential for biological activity. This is achieved by reacting the chiral amine with 4-chlorobutyryl chloride in the presence of a base like potassium hydroxide and a phase transfer catalyst such as benzyltriethylammonium chloride. The reaction proceeds through a nucleophilic acyl substitution followed by intramolecular cyclization. Rigorous control of reaction temperature, typically maintained between 10-15°C during reagent addition and allowed to warm to room temperature, is crucial to minimize side reactions such as polymerization or over-acylation. The resulting crude product is purified via recrystallization from ethyl acetate, yielding Levetiracetam with high chemical and optical purity, consistent with the physicochemical standards required for antiepileptic medications.
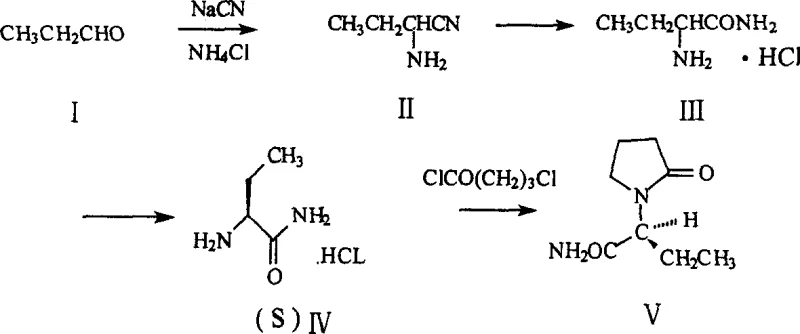
How to Synthesize (S)-alpha-ethyl-2-oxo-1-pyrrolidine acetamide Efficiently
Implementing this synthesis requires careful attention to solvent selection and stoichiometry to maximize the efficiency of the chiral resolution. The process begins with the formation of the nitrile intermediate, followed by hydrolysis to the amide salt. The pivotal step involves dissolving the DL-amide in a lower alcohol solvent and adding the chiral acid resolving agent to induce crystallization of the target diastereomer. After filtration and washing, the salt is liberated with base and converted back to the hydrochloride salt before the final cyclization. For detailed operational parameters, stoichiometry ratios, and specific workup procedures validated by experimental data, please refer to the standardized synthesis guide below.
- Perform Strecker reaction using propionaldehyde, sodium cyanide, and ammonium chloride to form alpha-aminobutyronitrile.
- Hydrolyze the nitrile intermediate using hydrogen chloride in alcohol to obtain DL-alpha-aminobutanamide hydrochloride.
- Resolve the racemic mixture using chiral acids like L-(+)-tartaric acid or (+)-mandelic acid to isolate the (S)-enantiomer.
- Execute cyclization with 4-chlorobutyryl chloride under alkaline conditions to form the final pyrrolidone ring structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers compelling economic and logistical benefits that extend beyond simple yield improvements. The reduction in synthetic steps from six to four inherently lowers the consumption of raw materials, solvents, and utilities, leading to a significant reduction in variable manufacturing costs. By eliminating the need for handling toxic HCN gas, facilities can avoid the capital expenditure associated with specialized scrubbing systems and corrosion-resistant alloys, thereby lowering the barrier to entry for production and reducing maintenance overheads. The use of commodity chemicals like propionaldehyde and sodium cyanide ensures a stable and reliable supply chain, minimizing the risk of raw material shortages that can plague more exotic synthetic pathways.
- Cost Reduction in Manufacturing: The streamlined four-step process drastically reduces the number of isolation and purification stages, which are typically the most labor and time-intensive parts of API manufacturing. By cutting out two full reaction steps, the process minimizes solvent usage for extractions and washes, directly lowering waste disposal costs and environmental compliance burdens. The improved overall yield means that less starting material is required to produce the same amount of final API, effectively amplifying the purchasing power of raw material budgets and enhancing the gross margin profile of the finished product without compromising quality standards.
- Enhanced Supply Chain Reliability: Relying on widely available bulk chemicals such as propionaldehyde and sodium cyanide mitigates the supply risks associated with specialized or custom-synthesized intermediates. The robustness of the diastereomer resolution technique allows for flexible scaling; if demand surges, the crystallization steps can be easily expanded in larger vessels without the need for complex chromatographic columns or enzymatic reactors that often bottleneck production. This flexibility ensures consistent lead times and enables manufacturers to respond rapidly to market fluctuations, securing a steady flow of high-purity Levetiracetam for downstream formulation.
- Scalability and Environmental Compliance: The replacement of hazardous HCN gas with solid NaCN significantly simplifies the environmental health and safety (EHS) profile of the manufacturing site. This change reduces the regulatory scrutiny and permitting complexity often associated with toxic gas storage and usage, facilitating faster approval for capacity expansions. Furthermore, the simplified workflow generates less chemical waste per kilogram of product, aligning with green chemistry principles and reducing the carbon footprint of the manufacturing operation, which is increasingly a key criterion for global pharmaceutical supply chain partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on how this method outperforms legacy routes in terms of safety, yield, and operational simplicity. Understanding these nuances is critical for technical teams evaluating process transfer feasibility.
Q: How does this new synthesis route improve safety compared to traditional methods?
A: The patented process replaces hazardous hydrogen cyanide (HCN) gas with solid sodium cyanide (NaCN) in the initial Strecker reaction. This substitution significantly reduces operational risks associated with toxic gas handling and eliminates the need for specialized corrosion-resistant equipment required for HCN, thereby enhancing overall plant safety and environmental compliance.
Q: What is the impact of the diastereomer resolution method on product purity?
A: By employing diastereomeric salt formation with resolving agents such as L-(+)-tartaric acid or (+)-mandelic acid, the process effectively separates the desired (S)-enantiomer from the (R)-isomer. This chemical resolution step, followed by recrystallization, ensures high optical purity (ee value) and consistent physicochemical properties, meeting stringent pharmaceutical specifications without requiring complex chromatographic separation.
Q: Why is the reduction from six steps to four steps significant for manufacturing?
A: Reducing the synthetic sequence from six steps to four directly correlates to a substantial increase in overall yield, rising from approximately 4.7% to 10%. Fewer unit operations mean reduced solvent consumption, lower energy requirements for heating and cooling, and minimized material loss during transfers and isolations, resulting in a more cost-effective and scalable manufacturing process suitable for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Levetiracetam Supplier
At NINGBO INNO PHARMCHEM, we leverage deep technical expertise to transform innovative patent strategies like CN1288133C into commercial reality. Our R&D team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this 4-step synthesis are fully realized in large-scale manufacturing. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to monitor chiral purity and impurity profiles at every stage, guaranteeing that our Levetiracetam meets the exacting standards required by global regulatory bodies.
We invite potential partners to engage with our technical procurement team to discuss how this optimized route can enhance your supply chain resilience. By requesting a Customized Cost-Saving Analysis, you can quantify the specific economic benefits of switching to this more efficient methodology for your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project timelines.
