Advanced Synthesis of Large Conjugated Fluorene Diamines for High-Performance Polymers
The landscape of high-performance polymer manufacturing is undergoing a significant transformation, driven by the relentless demand for materials that combine thermal stability with processability. Patent CN102408342B introduces a groundbreaking class of fluorene-containing functional diamine monomers characterized by a large conjugated structure. These novel compounds address the critical bottleneck in polyimide technology: the trade-off between thermal performance and solubility. By integrating a rigid, non-planar fluorene core with extensive conjugated systems, this technology enables the synthesis of polyamides, polyimides, and polyester-imides that possess superior solubility without sacrificing mechanical strength. For R&D directors seeking next-generation electronic materials, this patent offers a robust pathway to creating polymers with tailored photoelectric properties, including notable aggregation-induced luminescence.
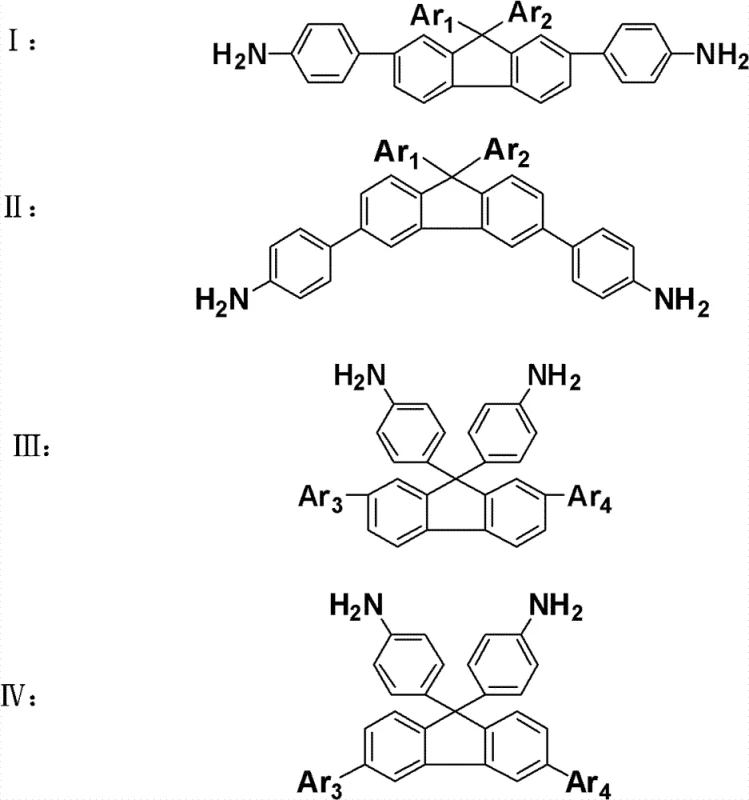
The strategic value of this invention lies in its versatility. The general formulas presented in the patent demonstrate a modular approach where various aryl groups can be introduced at specific positions. This modularity allows for fine-tuning the electronic and physical properties of the final polymer, making it an invaluable asset for developing specialized coatings, films, and microelectronic components. As a reliable electronic chemical supplier, understanding the nuances of such monomer design is crucial for meeting the evolving specifications of the semiconductor and display industries.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional polyimide synthesis has long been plagued by the inherent insolubility and infusibility of the final cured products. Conventional aromatic diamines often result in polymer chains that pack too tightly due to their planar structures, leading to strong intermolecular interactions that render the material impossible to process via standard solution techniques. Historically, manufacturers have relied on precursor polyamic acids, which are moisture-sensitive and suffer from limited storage stability. Furthermore, attempts to improve solubility by introducing flexible aliphatic linkages often come at the expense of thermal stability and glass transition temperature, limiting their application in high-temperature environments such as aerospace or advanced microelectronics. These structural compromises have restricted the broader adoption of polyimides in applications requiring both high performance and ease of fabrication.
The Novel Approach
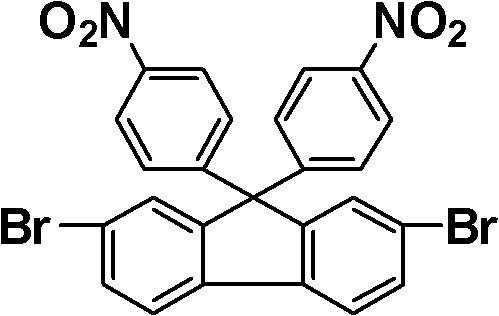
The methodology disclosed in CN102408342B circumvents these limitations through a sophisticated molecular design strategy. Instead of relying on flexible spacers, the invention utilizes the steric hindrance provided by the fluorene skeleton to disrupt chain packing. The synthesis begins with dihalogenated fluorene, which serves as a robust scaffold. By first reacting this scaffold with halonitrobenzene and subsequently linking large conjugated groups via the remaining halogen atoms, the process creates a bulky, three-dimensional architecture. This approach ensures that the resulting polymers maintain high thermal resistance while gaining exceptional solubility. The ability to introduce diverse functional groups through Suzuki coupling further enhances the material's adaptability, allowing for cost reduction in polymer additive manufacturing by enabling solution processing techniques that were previously unfeasible.
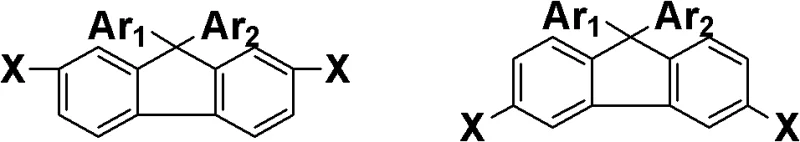
Mechanistic Insights into Suzuki Coupling and Catalytic Reduction
The core of this synthesis relies on two pivotal chemical transformations: palladium-catalyzed Suzuki cross-coupling and catalytic nitro reduction. The Suzuki reaction is employed to construct the large conjugated system by linking arylboronic acids to the halogenated fluorene intermediate. This reaction is highly advantageous due to its mild conditions and tolerance for various functional groups. The mechanism involves the oxidative addition of the aryl halide to the palladium catalyst, followed by transmetallation with the boronic acid species and reductive elimination to form the new carbon-carbon bond. This step is critical for establishing the extended pi-system responsible for the material's photoelectric properties. The precision of this coupling ensures high regioselectivity, minimizing the formation of structural isomers that could act as impurities and degrade the optical clarity of the final polymer.
Following the construction of the conjugated framework, the nitro groups are reduced to primary amines, which are essential for subsequent polymerization. The patent details the use of hydrazine hydrate in the presence of a palladium on carbon (Pd/C) catalyst. This heterogeneous catalytic system offers significant advantages over homogeneous alternatives, particularly regarding product isolation. The mechanism proceeds through the transfer of hydrogen from hydrazine to the nitro group via the metal surface, sequentially reducing the nitrogen oxidation state. This method is exceptionally clean, producing nitrogen gas and water as byproducts, which simplifies the purification process. The rigorous control over this reduction step is vital for ensuring high-purity electronic chemical standards, as residual nitro compounds or over-reduced byproducts can severely impact the dielectric properties of the resulting polyimide films.
How to Synthesize Fluorene Diamine Monomers Efficiently
The synthesis protocol outlined in the patent provides a clear, step-by-step guide for producing these high-value monomers. The process is designed to be robust and reproducible, starting from commercially available dihalofluorenes. The initial alkylation with nitrobenzenes sets the stage for the subsequent functionalization, while the final reduction step unlocks the reactive amine sites necessary for polymerization. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating these results.
- React dihalogenated fluorene with halonitrobenzene to form a dinitro intermediate containing two halogen atoms.
- Perform a Suzuki coupling reaction between the halogenated dinitro intermediate and arylboronic acids to establish the large conjugated system.
- Reduce the nitro groups to amino groups using a palladium catalyst and hydrazine hydrate to obtain the final diamine monomer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers tangible strategic benefits beyond mere technical performance. The process is engineered for efficiency, utilizing widely available raw materials such as dihalofluorenes and substituted boronic acids. This reliance on commodity chemicals mitigates supply risk and stabilizes pricing structures. Furthermore, the purification methods described, which primarily involve crystallization and filtration rather than expensive column chromatography, drastically simplify the downstream processing. This simplicity translates directly into lower operational expenditures and a reduced environmental footprint, aligning with modern green chemistry initiatives.
- Cost Reduction in Manufacturing: The elimination of complex purification steps significantly lowers production costs. By avoiding the need for extensive chromatographic separation, manufacturers can reduce solvent consumption and waste generation. The use of heterogeneous catalysts like Pd/C also facilitates catalyst recovery and reuse, further optimizing the cost structure. Additionally, the high yields reported in the examples indicate a material-efficient process, minimizing the loss of valuable intermediates and maximizing overall throughput.
- Enhanced Supply Chain Reliability: The synthetic route relies on stable, shelf-stable intermediates that can be stockpiled if necessary. The robustness of the Suzuki coupling and reduction reactions means that the process is less susceptible to minor fluctuations in reaction conditions, ensuring consistent batch-to-bquality. This reliability is crucial for maintaining continuous production schedules in high-volume manufacturing environments, reducing the risk of delays caused by failed batches or inconsistent raw material quality.
- Scalability and Environmental Compliance: The reaction conditions, typically involving reflux in common solvents like THF or ethanol, are easily scalable from laboratory to industrial reactors. The workup procedures, which often involve simple precipitation or crystallization upon cooling, are well-suited for large-scale equipment. Moreover, the byproducts of the reduction step are environmentally benign, simplifying waste treatment and ensuring compliance with increasingly stringent environmental regulations regarding volatile organic compounds and heavy metal discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this fluorene diamine technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for stakeholders evaluating this material for integration into their supply chains.
Q: How does this new diamine monomer improve polyimide solubility?
A: The monomer features a rigid non-planar spatial structure due to the sp3 hybridized carbon at the 9-position of the fluorene core. This geometry effectively prevents tight packing of polymer chains, reducing intermolecular forces and significantly enhancing solubility in organic solvents compared to traditional planar aromatic diamines.
Q: What are the key photoelectric properties of these monomers?
A: These monomers exhibit distinct aggregation-induced emission (AIE) characteristics. The large conjugated structure combined with the fluorene functional group endows the resulting polymers with specific photoelectric properties, making them highly suitable for applications in microelectronics and optoelectronic devices.
Q: Is the synthesis process scalable for industrial production?
A: Yes, the synthesis method is designed for industrial feasibility. It utilizes standard reactions like Suzuki coupling and catalytic reduction, followed by simple purification steps such as crystallization and filtration, which avoids complex chromatographic separations and facilitates large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluorene Diamine Monomer Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the fluorene-containing functional diamines described in CN102408342B. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped to handle the specific requirements of Suzuki couplings and catalytic reductions with the utmost precision. We adhere to stringent purity specifications and operate rigorous QC labs to ensure that every batch of monomer meets the exacting standards required for high-performance polyimide applications in the electronics sector.
We invite you to collaborate with us to unlock the full potential of these advanced materials. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes. Please contact our technical procurement team to request specific COA data and comprehensive route feasibility assessments. Let us help you secure a stable supply of high-quality fluorene diamines that will drive innovation in your next-generation polymer products.
