Advanced Fluorene-Based Diamine Monomers for High-Performance Flexible Display Substrates
Advanced Fluorene-Based Diamine Monomers for High-Performance Flexible Display Substrates
Introduction to Next-Generation Barrier Materials
The rapid evolution of flexible electronics, particularly Organic Light Emitting Diode (OLED) and Organic Liquid Crystal Display (OLCD) technologies, has placed unprecedented demands on substrate materials. As detailed in patent CN109456215B, the industry is shifting towards diamine monomers that incorporate fluorene or fluorenone structures to achieve exceptional planarity. This structural innovation is not merely a chemical curiosity but a critical engineering solution to the permeation issues that plague conventional polymer films. By leveraging the rigid fused-ring systems of fluorene derivatives, manufacturers can now access polyimides and polyamides with significantly reduced free volume, directly addressing the stringent water vapor and oxygen transmission requirements necessary for device lifespans exceeding 10,000 hours.
For R&D directors and procurement specialists, understanding the provenance of these materials is vital for supply chain security. The technology described represents a third-party validated approach to overcoming the limitations of traditional aromatic diamines, which often suffer from non-planar conformations that create voids in the polymer matrix. This patent provides a robust framework for synthesizing monomers that maintain high thermal stability while offering superior barrier performance. As a reliable electronic chemical supplier, recognizing the potential of these high-planarity monomers allows enterprises to future-proof their material sourcing strategies against the escalating performance standards of next-generation flexible devices.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional diamine monomers used in the synthesis of polyimide substrates often rely on flexible linkages or bulky substituents that disrupt the linearity of the polymer chain. This structural irregularity results in a higher fractional free volume within the cured film, creating micro-channels that facilitate the diffusion of moisture and oxygen. In the context of flexible display manufacturing, even minute permeation rates can lead to cathode oxidation and rapid device failure, rendering the end product commercially unviable. Furthermore, many conventional synthesis routes involve harsh conditions or expensive catalysts that complicate purification and increase the overall cost of goods sold, making them less attractive for high-volume consumer electronics applications.
Additionally, the thermal dimensional stability of standard polymer films often degrades at the elevated processing temperatures required for thin-film transistor fabrication. Conventional materials may exhibit significant coefficient of thermal expansion (CTE) mismatches with inorganic layers, leading to delamination or cracking during device operation. The reliance on non-planar monomers limits the ability of the polymer chains to pack efficiently, thereby compromising both the mechanical integrity and the barrier properties of the final substrate. These inherent deficiencies necessitate a fundamental redesign of the monomer architecture to meet the rigorous demands of modern optoelectronic applications without sacrificing processability.
The Novel Approach
The innovative strategy outlined in the patent data utilizes the intrinsic rigidity of fluorene and fluorenone cores to enforce a high degree of planarity in the resulting diamine monomers. By strategically positioning functional groups at the 2 and 7 positions of the fluorene ring, the synthesis ensures that the resulting polymer chains adopt a linear, rod-like conformation that promotes dense stacking. This molecular design effectively minimizes the interstitial spaces between chains, creating a tortuous path that drastically impedes the permeation of small gas molecules. The result is a polymer film that exhibits exceptional barrier properties while maintaining the flexibility required for roll-to-roll processing and flexible device integration.
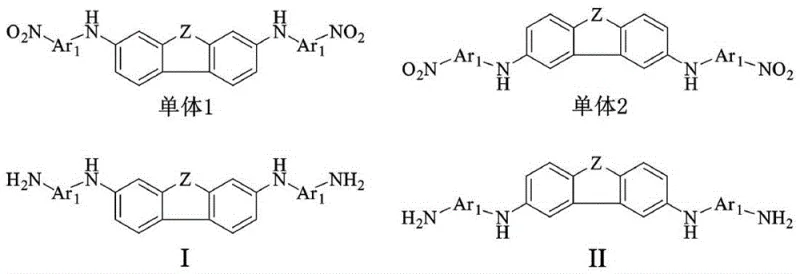
Furthermore, the versatility of the synthetic routes allows for the incorporation of various linkages such as amide, urea, and ether bonds without compromising the core planarity. This modularity enables chemists to fine-tune the solubility and glass transition temperature of the final polymer to match specific manufacturing requirements. The ability to synthesize these monomers through established reactions like Buchwald-Hartwig coupling and catalytic reduction ensures that the technology is not just theoretically sound but practically viable for cost reduction in electronic chemical manufacturing. This approach represents a significant leap forward in material science, bridging the gap between laboratory-scale innovation and industrial-scale production of high-performance substrates.
Mechanistic Insights into Fluorene-Based Monomer Design
The core mechanism driving the performance enhancement lies in the stereoelectronic properties of the fluorene and fluorenone units. Unlike biphenyl or other flexible aromatic systems, the fluorene backbone is locked into a coplanar configuration due to the bridging carbon atom, which prevents rotation around the aryl-aryl bonds. When this rigid unit is incorporated into a diamine monomer, it imparts a persistent linearity to the growing polymer chain during polycondensation. This structural rigidity is further enhanced by the presence of polar functional groups like carbonyls in fluorenone, which can engage in intermolecular interactions that further tighten the chain packing and reduce free volume.
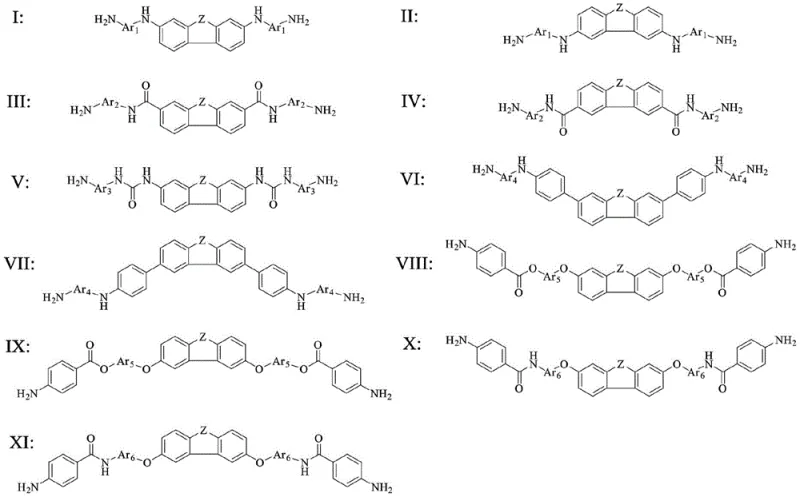
From an impurity control perspective, the synthesis pathways described allow for the formation of crystalline intermediates that can be easily purified via recrystallization. This is a critical advantage over amorphous oligomers that often trap solvent residues or catalyst byproducts, which can act as degradation initiation sites in the final film. The reduction of dinitro precursors to diamines using hydrazine hydrate or catalytic hydrogenation is highly selective, minimizing the formation of azo or azoxy byproducts that could discolor the transparent substrate. Such high purity is essential for optical applications where light transmission and color neutrality are paramount, ensuring that the material meets the stringent specifications required by top-tier display manufacturers.
How to Synthesize High Planarity Fluorene Diamine Monomers Efficiently
The synthesis of these advanced monomers typically begins with the functionalization of dihalogenated fluorene or fluorenone precursors. Depending on the desired linkage, chemists may employ palladium-catalyzed cross-coupling reactions to attach aromatic amines or utilize nucleophilic substitution to introduce ether or ester bonds. The subsequent reduction of nitro groups to amines is performed under controlled conditions to ensure complete conversion while preserving the integrity of the sensitive fluorene core. Detailed standardized synthesis steps see the guide below.
- Initiate the synthesis by reacting dihalogenated fluorene or fluorenone with amino groups via Buchwald-Hartwig coupling to form dinitro intermediates.
- Alternatively, convert dihalogenated precursors into diacyl chloride monomers through cyanidation and hydrolysis before amidation.
- Complete the process by reducing the dinitro-containing intermediates using catalytic hydrogenation or hydrazine hydrate to yield the final diamine monomer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers tangible benefits beyond mere performance metrics. The synthetic routes described rely on commodity chemicals and widely available catalysts, which mitigates the risk of raw material shortages and price volatility. By eliminating the need for exotic reagents or complex multi-step protections, the overall process complexity is significantly reduced, leading to substantial cost savings in the manufacturing overhead. This efficiency translates directly into a more competitive pricing structure for the final polyimide resin, allowing downstream device manufacturers to maintain healthy margins in a cost-sensitive market.
- Cost Reduction in Manufacturing: The streamlined synthesis pathways avoid the use of expensive transition metal catalysts that require rigorous removal steps, thereby simplifying the purification process. By utilizing crystallization as the primary purification method instead of column chromatography, the production process becomes far more amenable to large-scale batch operations. This operational efficiency eliminates the need for costly silica gel and large volumes of organic solvents, resulting in significant waste reduction and lower disposal costs. Consequently, the overall cost of goods is optimized, making high-performance barrier materials accessible for mass-market consumer electronics.
- Enhanced Supply Chain Reliability: The robustness of the chemical transformations, such as the Buchwald-Hartwig reaction and catalytic reduction, ensures high reproducibility across different production batches. This consistency is crucial for maintaining a stable supply of monomers that meet strict quality control standards without frequent process adjustments. Furthermore, the starting materials are derived from established petrochemical feedstocks, ensuring a secure and continuous supply chain that is less susceptible to geopolitical disruptions. This reliability allows supply chain planners to forecast inventory needs with greater confidence and reduce safety stock levels.
- Scalability and Environmental Compliance: The processes described are inherently scalable, moving seamlessly from gram-scale laboratory synthesis to ton-scale industrial production without fundamental changes to the reaction engineering. The use of aqueous workups and recyclable solvents aligns with modern green chemistry principles, reducing the environmental footprint of the manufacturing facility. This compliance with environmental regulations minimizes the risk of production shutdowns due to regulatory non-compliance and enhances the corporate sustainability profile. Ultimately, this facilitates the commercial scale-up of complex electronic chemicals with minimal environmental impact.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of these high-planarity diamine monomers in industrial settings. The answers are derived directly from the mechanistic data and synthesis examples provided in the patent literature, ensuring accuracy and relevance for technical decision-makers. Understanding these details is essential for evaluating the feasibility of integrating this material into existing production lines.
Q: How does the high planarity of this diamine monomer improve polymer barrier properties?
A: The high planarity of the fluorene or fluorenone structure allows polymer molecular chains to stack more compactly, significantly reducing free volume within the material matrix. This dense packing creates a more tortuous path for gas and water vapor permeation, thereby enhancing the water vapor transmission rate (WVTR) and oxygen transmission rate (O2TR) critical for flexible display longevity.
Q: What are the primary synthetic routes available for producing these monomers?
A: The patent outlines several versatile pathways, including Buchwald-Hartwig amination of dihalogenated fluorenes, Suzuki coupling reactions, and nucleophilic substitution involving acyl chlorides. These methods allow for the introduction of various functional linkages such as amide, urea, and ether bonds, providing flexibility in tuning the final polymer's thermal and mechanical properties.
Q: Are these monomers suitable for large-scale industrial production of polyimides?
A: Yes, the synthesis methods described utilize standard organic transformations like cyanidation, hydrolysis, and catalytic reduction which are well-established in industrial chemistry. The intermediates can be purified through crystallization and filtration, avoiding complex chromatographic separations, which supports scalable manufacturing for commercial polyimide and polyamide applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluorene Diamine Monomer Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced monomers play in the development of next-generation flexible displays. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from R&D to mass manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of fluorene diamine monomer meets the exacting standards required for high-barrier polyimide synthesis. Our commitment to quality ensures that your final polymer products deliver consistent performance in the most demanding optoelectronic applications.
We invite you to collaborate with our technical procurement team to explore how these innovative monomers can optimize your material costs and enhance product performance. Please contact us to request a Customized Cost-Saving Analysis tailored to your specific production volumes and requirements. Our experts are ready to provide specific COA data and route feasibility assessments to support your engineering decisions. By partnering with us, you gain access to a supply chain partner dedicated to driving innovation and efficiency in the electronic materials sector.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
