Advanced Synthetic Route for Rhizonin A: Enabling Commercial Scale-Up and High Purity
Introduction to the Novel Rhizonin A Synthesis Protocol
The pharmaceutical industry constantly seeks more efficient pathways for complex natural products, and the cyclic heptapeptide Rhizonin A represents a significant target due to its diverse biological activities including antibacterial and antitumor properties. As detailed in patent CN110655558A, a groundbreaking synthetic method has been developed that addresses the historical challenges of low yield and excessive step count associated with this molecule. This new approach utilizes a convergent strategy, starting from readily available precursors like L-N-Boc-valine derivatives, to construct the complex cyclic architecture with remarkable efficiency. By shifting away from linear elongation methods that plagued earlier attempts, this technology offers a robust framework for producing high-purity intermediates essential for drug discovery and development.
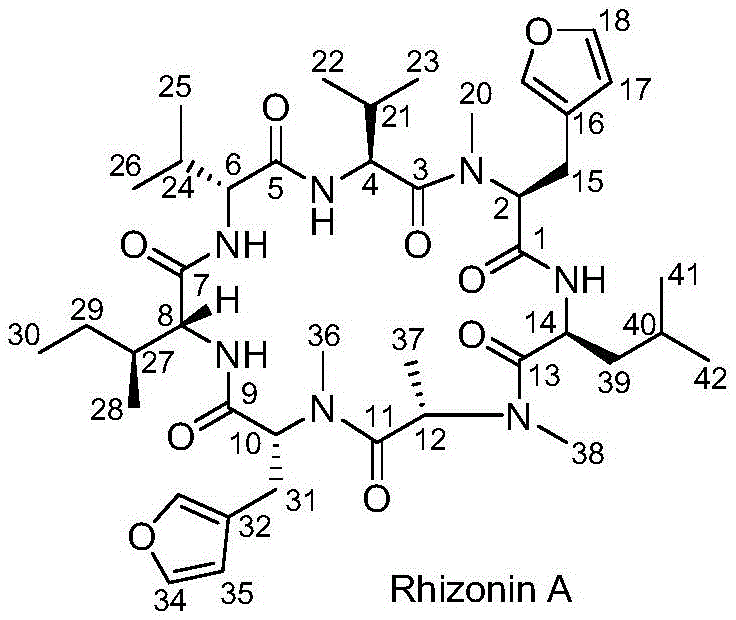
The significance of this patent lies in its ability to provide a reliable supply of Rhizonin A for biological evaluation, which was previously hindered by inefficient synthesis. The method leverages modern peptide coupling reagents such as HATU and HOAt to ensure high fidelity in amide bond formation while minimizing racemization, a critical concern in peptide chemistry. For R&D directors and procurement managers alike, this represents a shift towards more predictable and cost-effective manufacturing processes. The strategic design of the synthesis allows for the isolation of key intermediates, facilitating quality control at multiple stages and ensuring that the final product meets stringent purity specifications required for pharmaceutical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of Rhizonin A was characterized by prohibitive inefficiencies that made large-scale production economically unviable. The existing literature describes a linear synthetic route requiring approximately 17 distinct steps to reach the final target, resulting in a dismal overall yield of less than 1.2%. Such a low throughput not only drives up the cost of goods significantly but also introduces substantial risks regarding supply continuity and material availability for clinical studies. Furthermore, the extended exposure of sensitive peptide intermediates to various reaction conditions over such a long sequence increases the likelihood of side reactions, epimerization, and difficult-to-remove impurities. These factors collectively create a bottleneck for any organization attempting to utilize Rhizonin A in serious therapeutic development programs.
The Novel Approach
In stark contrast, the methodology disclosed in CN110655558A employs a convergent fragment condensation strategy that drastically streamlines the production workflow. By synthesizing two major peptide fragments independently—a tetrapeptide and a tripeptide—and then coupling them, the process reduces the longest linear sequence and improves the management of stereochemistry. This approach allows for the optimization of each fragment synthesis separately, leading to individual step yields that are exceptionally high, such as the 95% yield observed in the initial tripeptide formation. The use of commercially available starting materials further simplifies the supply chain, reducing lead times and dependency on custom-synthesized building blocks. This structural modularity is key to achieving the reported advantages of low cost, simple operation, and mild reaction conditions.
Mechanistic Insights into HATU-Mediated Peptide Coupling
The core chemical transformation driving this synthesis is the amide bond formation mediated by uranium-based coupling reagents, specifically 2-(7-oxidobenzotriazole)-N,N,N',N'-tetramethyluronium hexafluorophosphate (HATU), in the presence of 1-hydroxy-7-azabenzotriazole (HOAt) and N,N-diisopropylethylamine (DIPEA). This reagent system is chosen for its superior ability to activate carboxylic acids rapidly while suppressing racemization, which is paramount when dealing with chiral amino acid residues like valine and alloisoleucine found in Rhizonin A. The mechanism involves the formation of an active ester species that reacts efficiently with the amine component, even in sterically hindered environments typical of peptide chains. The patent specifies precise molar ratios, such as 1:1.1 to 1.5 for the coupling partners, ensuring that the limiting reagent is fully consumed without excessive waste of expensive activated amino acids.
Impurity control is rigorously managed through the selection of protecting groups and hydrolysis conditions. The use of Boc (tert-butyloxycarbonyl) groups provides orthogonal protection that can be cleanly removed under acidic conditions (using trifluoroacetic acid) without affecting the methyl esters or other sensitive functionalities until the appropriate stage. For instance, the selective acid hydrolysis steps allow for the exposure of specific amine termini for subsequent coupling while leaving other parts of the molecule intact. Additionally, the final macrocyclization step is carefully controlled by adjusting pH and concentration to favor intramolecular ring closure over intermolecular polymerization. This level of mechanistic control ensures that the final Rhizonin A product possesses the correct cyclic topology and stereochemical integrity required for its biological activity.
How to Synthesize Rhizonin A Efficiently
The synthesis of Rhizonin A is achieved through a logical sequence of fragment assembly followed by a critical ring-closing step. The process begins with the preparation of a tetrapeptide fragment and a tripeptide fragment using standard solution-phase peptide synthesis techniques. These fragments are then coupled to form a linear heptapeptide precursor, which undergoes deprotection and cyclization to yield the final target. The detailed operational parameters, including solvent choices like dichloromethane and tetrahydrofuran, and specific temperature controls ranging from 0°C to room temperature, are optimized to maximize yield and purity. For a comprehensive understanding of the exact stoichiometry and workup procedures required to replicate this high-efficiency route, please refer to the standardized guide below.
- Synthesize tripeptide and tetrapeptide fragments via acid hydrolysis and HATU-mediated coupling.
- Prepare the second fragment (tripeptide) using similar coupling strategies followed by alkaline hydrolysis.
- Couple the two major fragments to form the linear heptapeptide precursor.
- Perform sequential alkaline and acidic hydrolysis followed by macrocyclization to obtain Rhizonin A.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers transformative benefits regarding cost structure and operational reliability. The primary advantage stems from the drastic reduction in the number of synthetic steps compared to legacy methods, which directly correlates to lower labor costs, reduced solvent consumption, and decreased waste disposal expenses. By eliminating the need for a 17-step linear sequence, the manufacturing timeline is significantly compressed, allowing for faster turnaround times from order to delivery. This efficiency gain is compounded by the use of commodity chemicals and commercially available amino acid derivatives, which mitigates the risk of raw material shortages and price volatility often associated with exotic custom intermediates.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the high yields achieved in the early coupling stages, where yields of 87% to 95% are routinely observed. This high efficiency minimizes the loss of valuable chiral starting materials, which are often the most costly components of peptide synthesis. Furthermore, the simplified purification protocols, primarily relying on silica gel chromatography with standard solvent systems like ethyl acetate and hexane, reduce the need for expensive preparative HPLC at intermediate stages. The overall result is a substantial decrease in the cost of goods sold (COGS), making the commercial production of Rhizonin A financially sustainable for broader market applications.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the modular nature of the synthesis, where the two main fragments can be produced in parallel rather than sequentially. This parallel processing capability effectively halves the production time for the linear precursor, providing a buffer against potential delays in any single part of the workflow. Additionally, the reliance on robust, well-understood chemical transformations ensures that the process is less susceptible to batch-to-batch variability. This predictability allows for more accurate forecasting and inventory planning, ensuring that downstream clients receive their high-purity pharmaceutical intermediates without interruption.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the method utilizes mild reaction conditions that do not require extreme temperatures or pressures, simplifying the engineering requirements for scale-up from laboratory to pilot plant. The solvents used, such as dichloromethane and THF, are standard in the industry and can be efficiently recovered and recycled, aligning with green chemistry principles and reducing the environmental footprint. The avoidance of heavy metal catalysts or toxic reagents further simplifies waste treatment and regulatory compliance, facilitating a smoother path to commercial approval and large-scale manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Rhizonin A using this patented methodology. These answers are derived directly from the experimental data and process descriptions found in the patent documentation, providing clarity on the feasibility and advantages of this route. Understanding these details is crucial for stakeholders evaluating the potential integration of this intermediate into their own development pipelines.
Q: How does this new synthesis method improve upon previous Rhizonin A production techniques?
A: The patented method significantly reduces the synthetic route length and improves total yield compared to the prior art which suffered from less than 1.2% overall yield over 17 steps. By utilizing a convergent fragment condensation strategy with commercially available starting materials, the new process achieves higher efficiency and better suitability for large-scale preparation.
Q: What are the key reaction conditions for the macrocyclization step?
A: The final cyclization involves treating the linear heptapeptide precursor with lithium hydroxide for alkaline hydrolysis, followed by acidification with trifluoroacetic acid. The cyclization itself is mediated by HATU and HOAt in dichloromethane at temperatures ranging from 0°C to room temperature, ensuring controlled ring closure with minimal epimerization.
Q: Is this synthesis route suitable for industrial scale-up?
A: Yes, the method is designed for scalability. It employs mild reaction conditions (0°C to room temperature), common organic solvents like dichloromethane and THF, and avoids exotic catalysts. The high yields in early coupling steps (up to 95%) and the use of robust protecting group strategies make it viable for commercial manufacturing of complex cyclic peptides.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rhizonin A Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a dependable partner for complex peptide intermediates like Rhizonin A. Our technical team has extensively analyzed this patented route and possesses the expertise to implement it with precision, ensuring that every batch meets the rigorous standards demanded by the pharmaceutical industry. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging our state-of-the-art facilities to maintain stringent purity specifications throughout the manufacturing process. Our rigorous QC labs are equipped to perform comprehensive structural characterization, including NMR and HRMS, to guarantee the identity and quality of the final product.
We invite you to collaborate with us to optimize your supply chain for Rhizonin A and related cyclic peptides. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the highest level of confidence and technical support.
