Advanced Synthesis of Rhizonin A: Scalable Cyclic Heptapeptide Production for Pharmaceutical R&D
The pharmaceutical and agrochemical industries are constantly seeking efficient pathways to access complex bioactive natural products, and the cyclic heptapeptide Rhizonin A represents a prime target due to its unique biological properties including weight loss efficacy and seed germination promotion. Patent CN110655558B discloses a groundbreaking synthetic method that overcomes the historical limitations associated with producing this intricate molecule. By leveraging a strategic fragment condensation approach, this technology enables the construction of the heptapeptide backbone with remarkable precision and efficiency. The core structure of Rhizonin A, characterized by its specific amide linkages and furan-containing side chains, is depicted below, highlighting the stereochemical complexity that demands a robust synthetic strategy. 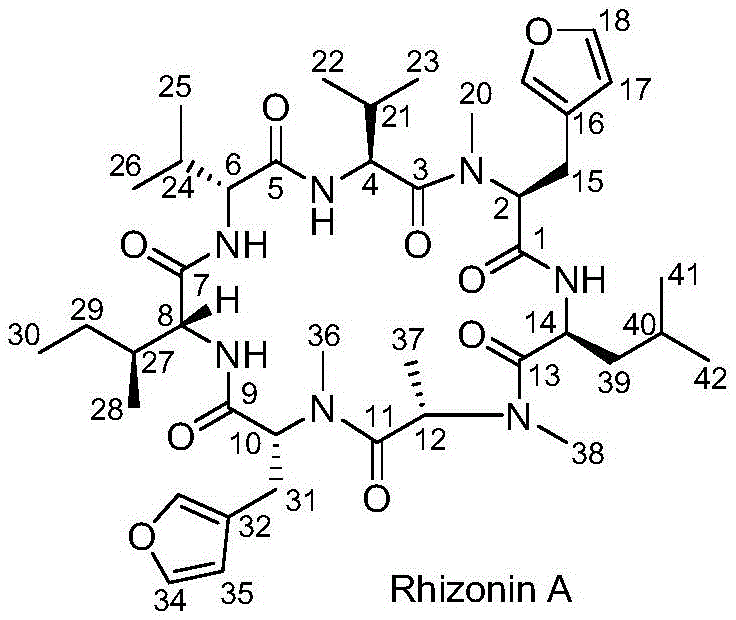 . For R&D directors and procurement specialists, understanding this patented route is critical for securing a reliable pharmaceutical intermediates supplier capable of delivering high-purity materials for biological evaluation and potential therapeutic development.
. For R&D directors and procurement specialists, understanding this patented route is critical for securing a reliable pharmaceutical intermediates supplier capable of delivering high-purity materials for biological evaluation and potential therapeutic development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovation detailed in CN110655558B, the synthesis of Rhizonin A was plagued by inefficiencies that rendered large-scale production economically unviable. The previously reported method, as documented in Tetrahedron Letters, relied on a linear assembly strategy that necessitated a staggering 17 synthetic steps from initial raw materials. This excessive step count not only accumulated significant material losses at each stage but also resulted in a dismal overall yield of less than 1.2%. Such low efficiency creates a bottleneck for supply chain heads who require consistent volumes for preclinical and clinical studies. Furthermore, the lengthy route involves multiple protection and deprotection cycles that increase the consumption of reagents and solvents, thereby inflating the cost of goods sold. The complexity of the linear synthesis also heightens the risk of impurity accumulation, complicating the purification process and potentially compromising the quality of the final active pharmaceutical ingredient. These factors collectively make the conventional method unsuitable for the commercial scale-up of complex pharmaceutical intermediates required by modern drug discovery pipelines.
The Novel Approach
In stark contrast, the novel approach introduced in the patent utilizes a convergent synthesis strategy that dramatically streamlines the production process. By synthesizing smaller peptide fragments independently and then coupling them, the method reduces the total number of operational steps and minimizes the handling of intermediates. This fragment-based logic allows for better control over stereochemistry and purity at each stage, ensuring that the final heptapeptide meets stringent quality specifications. The use of commercially available starting materials, such as protected valine and allo-isoleucine derivatives, further simplifies the supply chain and reduces lead time for high-purity pharmaceutical intermediates. The reaction conditions are notably mild, typically ranging from 0°C to room temperature, which enhances safety and reduces energy consumption during manufacturing.  . This strategic shift from linear to convergent synthesis represents a significant technological leap, offering a viable pathway for cost reduction in pharmaceutical intermediates manufacturing while maintaining the structural integrity of the target molecule.
. This strategic shift from linear to convergent synthesis represents a significant technological leap, offering a viable pathway for cost reduction in pharmaceutical intermediates manufacturing while maintaining the structural integrity of the target molecule.
Mechanistic Insights into Peptide Condensation and Macrolactamization
The success of this synthesis hinges on the precise execution of peptide coupling reactions and the final macrocyclization step. The process employs potent coupling reagents such as 2-(7-azabenzotriazol-1-yl)-N,N,N',N'-tetramethyluronium hexafluorophosphate (HATU) alongside 1-hydroxy-7-azabenzotriazole (HOAt) to facilitate amide bond formation with minimal racemization. These reagents activate the carboxyl groups of the amino acid fragments, allowing them to react efficiently with the amino groups of the partnering peptides in solvents like dichloromethane. The careful selection of protecting groups, such as Boc (tert-butyloxycarbonyl) and methyl esters, ensures that reactivity is directed solely to the desired sites. Acid hydrolysis using trifluoroacetic acid is utilized to remove Boc groups selectively, while alkaline hydrolysis with lithium hydroxide cleaves methyl esters to reveal carboxylic acids for subsequent coupling. This orthogonal protection strategy is vital for maintaining the sequence fidelity of the heptapeptide chain. The final cyclization step involves the intramolecular condensation of the linear heptapeptide precursor, a challenging transformation that requires high dilution conditions to favor ring closure over polymerization.  . Understanding these mechanistic details allows R&D teams to troubleshoot potential issues and optimize the process for even greater efficiency and yield in a production environment.
. Understanding these mechanistic details allows R&D teams to troubleshoot potential issues and optimize the process for even greater efficiency and yield in a production environment.
Impurity control is another critical aspect of this mechanistic framework, particularly given the presence of multiple chiral centers in Rhizonin A. The use of high-quality chiral starting materials and mild coupling conditions helps preserve the stereochemical integrity of the amino acid residues throughout the synthesis. Any epimerization during the activation step could lead to diastereomeric impurities that are difficult to separate and could alter the biological activity of the final product. The patent specifies rigorous purification protocols, including silica gel column chromatography with specific eluent systems like ethyl acetate and n-hexane, to isolate the desired intermediates and the final product with high purity. The analytical data provided, including NMR and HRMS, confirms the structural identity and purity of the synthesized compounds. For procurement managers, this level of detail assures that the supplier has a deep understanding of the critical quality attributes (CQAs) and can consistently deliver material that meets the rigorous standards required for biological activity evaluation. The robust nature of the chemistry ensures that the process is reproducible, a key factor for long-term supply chain reliability.
How to Synthesize Rhizonin A Efficiently
The synthesis of Rhizonin A is executed through a series of well-defined steps that prioritize yield and operational simplicity. The process begins with the preparation of key peptide fragments, such as the tetrapeptide and tripeptide units, using standard coupling protocols. These fragments are then deprotected and coupled to form the linear heptapeptide precursor. The final and most critical step is the macrocyclization, which forms the characteristic cyclic structure of Rhizonin A. Detailed standard operating procedures for each reaction, including specific molar ratios, solvent volumes, and reaction times, are essential for replicating the high yields reported in the patent. The following guide outlines the critical phases of this synthesis, providing a roadmap for technical teams looking to implement this route. For a comprehensive breakdown of the standardized synthesis steps, please refer to the detailed guide below.
- Prepare tetrapeptide and tripeptide fragments by condensing protected amino acids like L-N-Boc-valine and D-N-Boc-allo-isoleucine using HATU coupling reagents.
- Couple the tetrapeptide and tripeptide fragments to form the linear heptapeptide precursor through careful deprotection and activation steps.
- Perform macrocyclization of the linear heptapeptide via alkaline and acidic hydrolysis followed by self-condensation to yield Rhizonin A.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthesis method offers substantial benefits for procurement and supply chain teams managing the sourcing of complex peptide intermediates. The reduction in synthetic steps directly translates to a more streamlined manufacturing process, which inherently lowers the risk of production delays and batch failures. By utilizing commercially available starting materials, the dependency on custom-synthesized precursors is minimized, thereby enhancing supply chain resilience and reducing lead times. The mild reaction conditions also mean that the process can be executed in standard chemical manufacturing facilities without the need for specialized high-pressure or cryogenic equipment, further lowering capital expenditure requirements. These factors combine to create a more agile and cost-effective supply chain capable of responding quickly to the fluctuating demands of the pharmaceutical market. The ability to produce Rhizonin A efficiently positions suppliers to offer more competitive pricing structures without compromising on quality or delivery performance.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the significant reduction in the number of synthetic steps compared to conventional methods. Fewer steps mean less consumption of solvents, reagents, and labor hours, which are the major components of manufacturing costs. Additionally, the higher overall yield ensures that more product is obtained from the same amount of starting material, effectively lowering the cost per gram of the final API intermediate. The elimination of complex purification stages required for linear synthesis byproducts further contributes to cost savings. By avoiding the use of expensive transition metal catalysts that require rigorous removal, the process simplifies the downstream processing workflow. These cumulative efficiencies result in substantial cost savings that can be passed down the supply chain, making the final therapeutic candidate more economically viable for development.
- Enhanced Supply Chain Reliability: Supply chain reliability is bolstered by the use of robust and readily available raw materials. The reliance on commercial amino acid derivatives reduces the risk of supply disruptions associated with custom synthesis of exotic building blocks. The simplicity of the operation also means that the process is less prone to technical failures, ensuring consistent batch-to-batch quality and on-time delivery. The scalability of the method allows suppliers to ramp up production quickly in response to increased demand from clinical trials or commercial launch. This flexibility is crucial for pharmaceutical companies that need to secure a steady supply of critical intermediates to maintain their development timelines. Partnering with a supplier who utilizes this advanced route ensures a stable and predictable flow of materials, mitigating the risks associated with single-source dependencies.
- Scalability and Environmental Compliance: The environmental profile of this synthesis is significantly improved due to the reduced waste generation associated with fewer synthetic steps. The use of mild conditions and common solvents facilitates easier waste treatment and disposal, aligning with increasingly stringent environmental regulations. The process is designed for scalability, meaning it can be transitioned from laboratory scale to multi-ton production without significant re-engineering. This scalability is essential for meeting the growing demand for Rhizonin A as its biological applications are further explored. The efficient use of resources and the minimization of hazardous waste contribute to a more sustainable manufacturing footprint. For supply chain heads, this means partnering with a supplier who is not only cost-effective but also compliant with global environmental standards, reducing the risk of regulatory penalties and reputational damage.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and supply of Rhizonin A. These answers are derived directly from the technical specifications and beneficial effects outlined in the patent data. Understanding these details helps stakeholders make informed decisions about integrating this intermediate into their development pipelines. The responses cover aspects of yield improvement, raw material sourcing, and scalability, providing a comprehensive overview of the technology's value proposition. For more specific technical data or custom synthesis inquiries, please consult with our technical team.
Q: How does the new synthesis method improve yield compared to conventional routes?
A: The conventional route reported in Tetrahedron Letters requires 17 steps with an overall yield of less than 1.2%. The novel method described in patent CN110655558B significantly shortens the reaction route and utilizes high-yielding fragment condensation, resulting in a substantially higher total yield suitable for industrial production.
Q: What are the key starting materials for this Rhizonin A synthesis?
A: The process utilizes commercially available amino acid derivatives such as L-N-Boc-valine-D-C-OMe-valine, D-N-Boc-allo-isoleucine, and L-N-Me-3-(3-furyl) alanine. These readily available starting materials contribute to the cost-effectiveness and supply chain reliability of the method.
Q: Is this synthesis method scalable for commercial manufacturing?
A: Yes, the method is designed for large-scale synthesis. It features mild reaction conditions (0°C to room temperature), simple operation procedures, and avoids extremely harsh reagents, making it environmentally friendly and highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rhizonin A Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthesis routes in accelerating drug discovery and development. Our expertise as a CDMO partner allows us to leverage advanced technologies like the one described in CN110655558B to deliver high-quality cyclic heptapeptides. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are equipped with stringent purity specifications and rigorous QC labs to guarantee that every batch of Rhizonin A meets the highest industry standards. By choosing us as your reliable Rhizonin A supplier, you gain access to a partner committed to technical excellence and supply chain stability. We understand the complexities of peptide synthesis and are dedicated to providing solutions that enhance your R&D productivity.
We invite you to collaborate with us to optimize your supply chain for this valuable intermediate. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality expectations. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your projects. Let us help you navigate the challenges of complex peptide synthesis and secure a reliable source of high-purity materials for your next breakthrough. Together, we can drive innovation and efficiency in the development of novel therapeutic agents based on the Rhizonin A scaffold.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
