Advanced Asymmetric Synthesis of (R)-5'-Methoxylaudanosine for Commercial Mivacurium Production
Advanced Asymmetric Synthesis of (R)-5'-Methoxylaudanosine for Commercial Mivacurium Production
The pharmaceutical landscape for neuromuscular blocking agents continues to evolve, driven by the demand for ultra-high purity intermediates that ensure patient safety and regulatory compliance. Patent CN113480480A introduces a groundbreaking synthesis method for (R)-5'-methoxylaudanosine, a critical chiral building block for Mivacurium Chloride, a short-acting non-depolarizing muscle relaxant. This technical disclosure outlines a robust three-step pathway that leverages asymmetric hydrogenation to achieve exceptional stereocontrol, bypassing the limitations of traditional resolution techniques. By integrating a non-genotoxic Ruthenium-BINAP catalyst system, this methodology not only secures optical purity exceeding 99.98% after refinement but also aligns with modern green chemistry principles essential for sustainable API manufacturing. For global procurement and R&D teams, this represents a significant leap forward in securing reliable supply chains for complex benzylisoquinoline derivatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of (R)-5'-methoxylaudanosine has been plagued by inefficiencies inherent in classical resolution strategies and earlier catalytic attempts. Traditional methods, such as those described in U.S. Pat. No. 5,476,418A, rely on the resolution of racemic mixtures using D-(-)-dibenzoyl tartaric acid. This approach is fundamentally flawed for modern high-volume production because the theoretical maximum yield is capped at 50%, leading to substantial material waste and inflated costs. Furthermore, incomplete resolution often leaves behind the unwanted (S)-enantiomer, which poses severe challenges in subsequent synthetic steps, particularly when constructing the specific diastereomers required for Mivacurium Chloride. Even more concerning are the asymmetric hydrogenation methods reported in literature (e.g., Tetrahedron: Asymmetry, 2013), which, while improving yield, often utilize catalysts containing p-toluenesulfonyl groups. These catalysts carry a significant risk of generating genotoxic impurities that are notoriously difficult to remove to the stringent parts-per-million levels required by international pharmacopoeias, thereby creating a hidden liability in the supply chain.
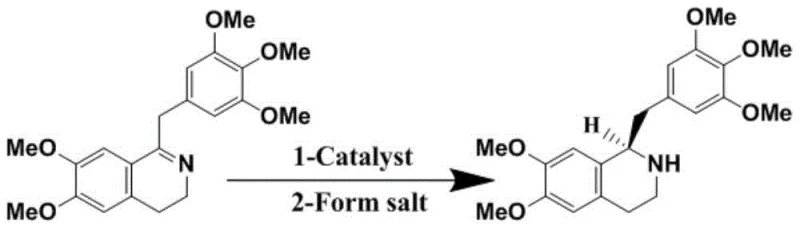
The Novel Approach
The innovative strategy disclosed in CN113480480A fundamentally reengineers the synthetic pathway to eliminate these bottlenecks through a precise three-step sequence: N-formylation, asymmetric hydrogenation, and formyl reduction. Instead of struggling with racemic separation, this method constructs the chiral center directly with high fidelity. The process begins with the conversion of 6,7-dimethoxy-1-(3,4,5-trimethoxybenzyl)-3,4-dihydroisoquinoline into a Z-type imine intermediate via N-formylation, a crucial step that pre-organizes the molecule for selective reduction. The core breakthrough lies in the subsequent asymmetric hydrogenation, which utilizes a specialized Ruthenium catalyst that avoids toxic functional groups entirely. This novel route achieves a total yield of up to 81.9%, effectively doubling the efficiency of traditional resolution methods while simultaneously elevating optical purity to levels that simplify downstream purification. The result is a streamlined, cost-effective process that delivers pharmaceutical-grade intermediates with minimal environmental footprint.
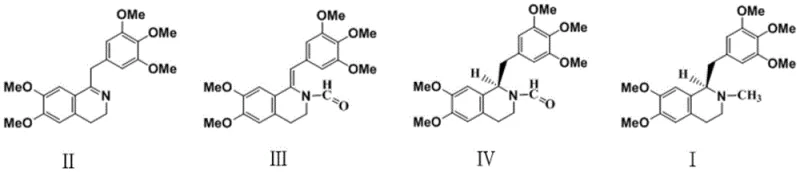
Mechanistic Insights into Ru-BINAP Catalyzed Asymmetric Hydrogenation
The heart of this technological advancement is the deployment of the chiral selective catalyst Ru(CH3COO)2((R)-BINAP). Unlike the bulky and potentially toxic sulfonamide ligands used in previous generations of catalysts, this Ruthenium-BINAP complex offers a sterically defined environment that strictly controls the facial selectivity of hydrogen addition to the imine double bond. The mechanism involves the coordination of the Z-type imine substrate to the ruthenium center, where the chiral BINAP ligand creates a specific pocket that favors the formation of the (R)-configuration. This precise molecular recognition ensures that the hydrogen atoms are delivered exclusively to one face of the planar imine, resulting in an initial optical purity of over 99.79% ee directly from the reactor. The use of acetate ligands on the ruthenium center further enhances the catalyst's stability and activity under mild conditions, allowing the reaction to proceed efficiently at temperatures between 23°C and 35°C and low hydrogen pressures of 1 to 4.2 atm.
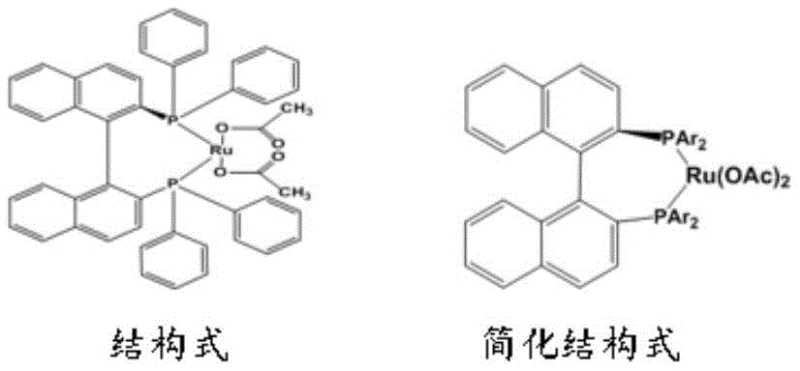
Beyond stereocontrol, the choice of this catalyst system is pivotal for impurity management and regulatory compliance. The absence of genotoxic structural alerts in the catalyst ligand means that the risk of carrying over mutagenic residues into the final API is virtually eliminated. This is a critical advantage for R&D directors managing regulatory filings, as it removes the need for extensive and expensive purge studies typically required for sulfonamide-based processes. Furthermore, the reaction solvent system, typically a mixture of methanol or ethanol with dichloromethane in a 5:1 ratio, is optimized to maintain catalyst solubility while facilitating easy product isolation. The subsequent formyl reduction step, achievable with either lithium aluminum hydride or a rhodium/silane system, cleanly removes the protecting group without racemization, preserving the hard-won chiral integrity throughout the synthesis.
How to Synthesize (R)-5'-Methoxylaudanosine Efficiently
Implementing this synthesis requires careful attention to the preparation of the Z-type imine intermediate, as its geometric purity directly influences the enantioselectivity of the hydrogenation step. The process initiates with the N-formylation of the dihydroisoquinoline starting material, which can be achieved using various reagents such as formic acid with zinc powder or anhydrides, often aided by light irradiation to promote the Z-isomer formation. Once the imine is secured and recrystallized, it undergoes the critical asymmetric hydrogenation in a pressurized reactor equipped for safe hydrogen handling. The final deprotection step is straightforward, utilizing standard reducing agents to yield the target amine. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized synthesis guide below.
- Perform N-formylation on 6,7-dimethoxy-1-(3,4,5-trimethoxybenzyl)-3,4-dihydroisoquinoline using formic acid derivatives to obtain the Z-type imine intermediate.
- Conduct asymmetric hydrogenation using Ru(CH3COO)2((R)-BINAP) catalyst in a methanol/dichloromethane mixed solvent under mild hydrogen pressure.
- Execute formyl reduction using lithium aluminum hydride or rhodium chloride/diphenylsilane, followed by recrystallization to achieve >99.98% optical purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the transition to this patented synthesis method offers profound strategic advantages that extend beyond simple unit cost calculations. The primary value driver is the drastic simplification of the purification train. By eliminating the need for chiral resolution columns or the removal of genotoxic catalyst residues, manufacturers can significantly reduce the number of processing steps, solvent consumption, and quality control testing burdens. This streamlining translates directly into lower operational expenditures and a reduced carbon footprint, aligning with the sustainability goals of major multinational pharmaceutical corporations. Moreover, the high overall yield of nearly 82% means that less raw material is required to produce the same amount of active intermediate, providing a natural hedge against volatility in the pricing of specialty chemical starting materials.
- Cost Reduction in Manufacturing: The elimination of genotoxic catalysts removes the necessity for expensive scavenging resins and rigorous heavy metal testing, which are significant cost centers in traditional API manufacturing. Additionally, the high atom economy of the asymmetric hydrogenation step, compared to the 50% theoretical limit of resolution, effectively doubles the output per batch of starting material. This efficiency gain allows for substantial cost savings in raw material procurement and waste disposal, as the process generates significantly less chemical waste per kilogram of product. The use of industrially mature catalysts also ensures that the cost of goods sold remains stable and predictable, avoiding the premiums associated with exotic or custom-synthesized ligands.
- Enhanced Supply Chain Reliability: The reliance on commercially available and robust reagents, such as Ru-BINAP and common organic solvents, mitigates the risk of supply disruptions often caused by single-source dependencies on niche reagents. The mild reaction conditions (near ambient temperature and low pressure) reduce the engineering complexity required for production, meaning that the process can be easily transferred between different manufacturing sites or scaled up without requiring specialized high-pressure reactors. This flexibility ensures a continuous and reliable supply of high-purity intermediates, safeguarding the production schedules of downstream drug products against technical bottlenecks or equipment failures.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, with reaction parameters that are easily controlled in large-scale vessels. The avoidance of hazardous genotoxic substances simplifies the environmental, health, and safety (EHS) profile of the manufacturing site, reducing the regulatory burden and insurance costs associated with handling toxic materials. Furthermore, the high purity of the crude product reduces the load on wastewater treatment systems and minimizes the volume of solvent waste generated during purification. This alignment with green chemistry principles not only facilitates smoother regulatory approvals but also enhances the corporate social responsibility profile of the supply chain partners involved.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a transparent view of the process capabilities. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for commercial production.
Q: How does this synthesis method address genotoxic impurity concerns?
A: Unlike prior art methods utilizing p-toluenesulfonyl-containing catalysts which risk genotoxic residue, this process employs Ru(CH3COO)2((R)-BINAP), an industrially mature catalyst free from genotoxic structural alerts, ensuring safer API intermediates.
Q: What optical purity levels can be achieved with this route?
A: The direct synthesis yields product with over 99.79% ee. Following a standard refinement step using organic acids like D-quinic acid, the optical purity exceeds 99.98% with chemical purity greater than 99.93%.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process operates under mild conditions (23-35°C, low hydrogen pressure) using commercially available solvents and catalysts, facilitating easy scale-up from kilogram to multi-ton production without harsh operational requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-5'-Methoxylaudanosine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the synthesis of complex chiral intermediates like (R)-5'-methoxylaudanosine requires more than just a patent; it demands deep process engineering expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced chiral HPLC and NMR capabilities to guarantee that every batch meets the >99.98% ee and >99.93% chemical purity standards required for Mivacurium Chloride synthesis. Our infrastructure is designed to handle sensitive catalytic hydrogenations safely and consistently, providing a secure foundation for your long-term supply needs.
We invite you to collaborate with us to leverage this advanced technology for your pipeline. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data from our pilot batches and comprehensive route feasibility assessments to demonstrate how this non-genotoxic, high-yield process can optimize your supply chain and reduce your overall cost of goods. Let us be your partner in delivering high-quality pharmaceutical intermediates with speed, precision, and reliability.
