Advanced Asymmetric Synthesis of (R)-5'-Methoxylaudanosine: A Scalable Route for High-Purity Muscle Relaxant Intermediates
The pharmaceutical industry's demand for high-purity neuromuscular blocking agents continues to drive innovation in intermediate synthesis, particularly for complex molecules like Mivacurium Chloride. Patent CN115073373A introduces a groundbreaking method for the asymmetric synthesis of (R)-5'-methoxylaudanosine, a pivotal chiral building block. This technology addresses long-standing challenges in the field by replacing hazardous reagents and low-yield resolution steps with a robust, chiral inducer-based strategy. By leveraging steric hindrance effects from specific amines such as R-(+)-alpha-methylbenzylamine, the process achieves exceptional stereocontrol without relying on prohibitively expensive noble metal catalysts. For R&D directors and procurement specialists, this represents a significant shift towards safer, more cost-effective manufacturing of critical pharmaceutical intermediates. The method not only enhances the overall mass yield to between 55% and 65% but also ensures the final product meets the rigorous purity standards required for injectable medications, thereby mitigating supply chain risks associated with quality failures.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of (R)-5'-methoxylaudanosine has been plagued by inefficiencies and safety hazards that complicate commercial scale-up. Traditional routes, as illustrated in the prior art, typically involve a five-step sequence starting from 3,4,5-trimethoxyphenylacetic acid. A major bottleneck in these legacy processes is the reliance on high-temperature reflux in xylene for dehydration steps, which consumes excessive energy and often results in poor intermediate quality requiring multiple recrystallizations. Furthermore, the introduction of methyl groups frequently necessitates the use of methyl iodide, a known genotoxic impurity that poses severe regulatory and safety challenges for industrial facilities. The cumulative effect of these harsh conditions is a low total yield, often hovering between 40% and 45%, with the final optical resolution step yielding merely 25% to 30% of the desired enantiomer. This inefficiency leads to substantial material waste, particularly the difficult recovery of the (S)-enantiomer from mother liquors, creating significant environmental disposal burdens and inflating the cost of goods sold.
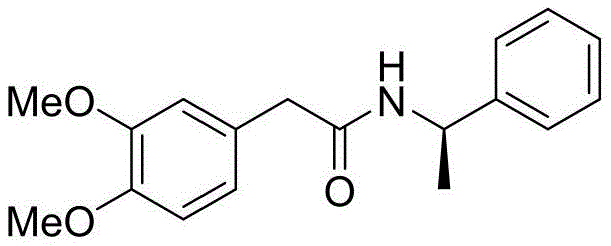
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent employs a sophisticated asymmetric synthesis strategy that fundamentally redesigns the molecular construction. Instead of relying on late-stage resolution of a racemate, this approach installs chirality early in the sequence using a chiral auxiliary. The process begins with the condensation of 3,4-dimethoxyphenylacetic acid with a chiral inducer, setting the stereochemical foundation for the entire molecule. This is followed by a streamlined sequence involving reduction, a second condensation, and a cyclization-reduction cascade that constructs the tetrahydroisoquinoline core with high diastereoselectivity. By avoiding the use of genotoxic alkylating agents and expensive homogeneous hydrogenation catalysts, the new route offers a cleaner, safer profile. The result is a dramatic improvement in process efficiency, with the total yield over six steps reaching 55% to 65%, effectively doubling the output compared to traditional resolution methods while simplifying the purification workflow.
Mechanistic Insights into Chiral Induction and Cyclization
The core innovation of this synthesis lies in the strategic use of steric hindrance to direct asymmetric induction. The process initiates with the formation of an amide bond between 3,4-dimethoxyphenylacetic acid and a chiral amine, such as R-(+)-alpha-methylbenzylamine. This chiral center acts as a temporary director, influencing the spatial arrangement of subsequent reactions. Following the reduction of this amide to the corresponding amine using a borane-tetrahydrofuran complex, the molecule undergoes a second acylation with 3,4,5-trimethoxyphenylacetic acid. The resulting diamide precursor is then subjected to Bischler-Napieralski type cyclization conditions using phosphorus oxychloride. This step is critical, as it forms the dihydroisoquinoline ring system. The subsequent reduction with sodium borohydride at low temperatures (-80°C to -76°C) is performed in batches to maximize stereoselectivity, ensuring that the newly formed chiral center aligns correctly with the existing chirality from the inducer. This precise control minimizes the formation of diastereomers, which is essential for achieving high optical purity in the final product.
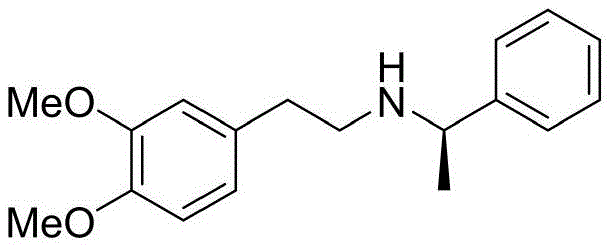
Following the construction of the chiral tetrahydroisoquinoline scaffold, the synthesis proceeds with the removal of the chiral auxiliary and functional group adjustments. The debenzylation step utilizes catalytic hydrogenation with palladium on carbon under a hydrogen atmosphere, a robust and scalable method that cleanly removes the benzyl protecting groups without affecting the sensitive methoxy substituents. The final N-methylation is achieved using formaldehyde and a phosphite reducing agent, avoiding the use of hazardous methyl iodide entirely. This sequence ensures that the final (R)-5'-methoxylaudanosine possesses an optical purity of 100% after a simple resolution and dissociation step, with chemical purity exceeding 99.95%. The control of impurities is paramount; the process is designed to keep all single impurities below 0.1%, a specification that is critical for intermediates destined for parenteral formulations where safety margins are extremely narrow.
How to Synthesize (R)-5'-Methoxylaudanosine Efficiently
The synthesis of this high-value intermediate requires precise control over reaction parameters to maintain stereochemical integrity. The process outlined in the patent provides a clear roadmap for transitioning from laboratory scale to commercial production. It emphasizes the importance of temperature control during the reduction steps and the selection of appropriate solvents to facilitate efficient workups. For detailed operational protocols, including specific molar ratios, reaction times, and purification techniques, please refer to the standardized guide below.
- Condense 3,4-dimethoxyphenylacetic acid with a chiral inducer like R-(+)-alpha-methylbenzylamine using EDCI to form the initial chiral amide intermediate.
- Reduce the amide to the corresponding amine using borane-THF complex, followed by a second condensation with 3,4,5-trimethoxyphenylacetic acid.
- Perform cyclization using phosphorus oxychloride, reduce with sodium borohydride, debenzyllate via hydrogenation, and finalize with N-methylation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers compelling economic and logistical benefits. The primary advantage lies in the substantial reduction of raw material costs and waste disposal expenses. By eliminating the need for expensive chiral catalysts like Ruthenium-BINAP complexes and hazardous reagents such as methyl iodide, the direct material cost of the synthesis is significantly lowered. Furthermore, the avoidance of genotoxic impurities simplifies the regulatory compliance burden, reducing the need for specialized containment equipment and extensive cleaning validation protocols. This translates directly into a lower cost of goods sold and a more competitive pricing structure for the final API. The improved yield profile also means that less starting material is required to produce the same amount of finished intermediate, enhancing overall resource efficiency.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts and toxic alkylating agents drastically reduces the cost of raw materials and waste treatment. The process utilizes common, inexpensive reagents like phosphorus oxychloride and sodium borohydride, which are readily available in the global chemical market. Additionally, the higher total yield means that the effective cost per kilogram of the active intermediate is significantly reduced compared to legacy methods. This efficiency allows manufacturers to offer more competitive pricing to downstream API producers while maintaining healthy margins.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals rather than specialized, single-source catalysts mitigates supply chain risks. Reagents such as R-(+)-alpha-methylbenzylamine and palladium on carbon are widely produced by multiple suppliers, ensuring continuity of supply even during market fluctuations. The robustness of the reaction conditions, which do not require extreme pressures or temperatures, further enhances equipment reliability and reduces downtime. This stability is crucial for maintaining consistent delivery schedules to pharmaceutical clients who operate on tight production timelines.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing unit operations such as filtration, extraction, and distillation that are standard in multipurpose chemical plants. The absence of genotoxic reagents simplifies environmental, health, and safety (EHS) management, reducing the regulatory hurdles associated with manufacturing. The higher atom economy and reduced solvent consumption contribute to a smaller environmental footprint, aligning with the sustainability goals of modern pharmaceutical companies. This makes the technology not only commercially viable but also environmentally responsible.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of (R)-5'-methoxylaudanosine. These answers are derived directly from the technical specifications and experimental data provided in the patent literature, ensuring accuracy and relevance for industry professionals.
Q: How does this new synthesis route improve upon traditional methods for (R)-5'-methoxylaudanosine?
A: The new route eliminates the use of genotoxic methyl iodide and toxic xylene reflux conditions found in prior art. It utilizes a chiral inducer strategy that achieves a total mass yield of 55-65%, significantly higher than the 25-30% yield of traditional resolution methods, while ensuring optical purity exceeds 99.95%.
Q: What are the critical quality control parameters for this intermediate?
A: Critical parameters include maintaining an ee value of greater than 90% prior to final resolution and achieving a final chemical purity of not less than 99.95%. The process ensures all single impurities remain below 0.1%, meeting the stringent requirements for injection-grade neuromuscular blocking agents.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process avoids expensive transition metal catalysts like Ruthenium-BINAP complexes and hazardous reagents. The use of standard reagents like phosphorus oxychloride and palladium on carbon, combined with mild reaction conditions, makes the route highly scalable and environmentally compliant for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-5'-Methoxylaudanosine Supplier
At NINGBO INNO PHARMCHEM, we understand the critical role that high-quality intermediates play in the successful development of neuromuscular blocking agents. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify identity and potency. Our facility is equipped to handle complex asymmetric syntheses safely and efficiently, adhering to the highest international standards for pharmaceutical manufacturing.
We invite you to collaborate with us to optimize your supply chain for Mivacurium Chloride intermediates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced synthesis capabilities can enhance the efficiency and profitability of your pharmaceutical projects.
