Revolutionizing TPO Production: A Deep Dive into One-Pot Synthesis Technology for Commercial Scale-Up
The global demand for high-efficiency photoinitiators in UV-curing applications has necessitated a rigorous re-evaluation of synthetic methodologies, particularly for critical compounds like 2,4,6-trimethylbenzoyl-diphenylphosphine oxide (TPO). Patent CN107304220B introduces a transformative one-pot synthesis strategy that fundamentally alters the production landscape for this essential optoelectronic material. Unlike conventional multi-step processes that suffer from cumulative yield losses and complex waste management, this novel approach utilizes a direct reaction between sodium, ethanol, diphenyl phosphine chloride, and 2,4,6-trimethyl benzoyl chloride. This technological leap not only streamlines the operational workflow but also addresses the pressing industry need for greener, more cost-effective manufacturing protocols. For R&D directors and procurement specialists alike, understanding the mechanistic underpinnings and commercial implications of this patent is crucial for maintaining competitiveness in the rapidly evolving specialty chemicals market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of TPO has relied heavily on a two-step pathway involving the initial preparation of diphenyl ethoxy phosphine followed by acylation. This traditional route, while chemically sound, is plagued by significant inefficiencies that impact both the bottom line and environmental footprint. A major bottleneck lies in the requirement for substantial quantities of acid-binding agents, typically triethylamine, to scavenge hydrochloric acid generated during the reaction. The subsequent formation of triethylamine hydrochloride salts creates a cumbersome waste stream that demands extensive separation and treatment procedures. Furthermore, the isolation of the intermediate diphenyl ethoxy phosphine often necessitates energy-intensive distillation steps, which not only consume valuable utility resources but also expose the thermally sensitive intermediates to potential degradation. These cumulative inefficiencies result in lower overall yields and higher production costs, making the conventional method increasingly unsustainable in a market driven by margin compression and strict environmental regulations.
The Novel Approach
In stark contrast, the methodology disclosed in the patent leverages a sophisticated one-pot design that bypasses the isolation of unstable intermediates entirely. By employing metallic sodium and ethanol directly in the reaction vessel, the process generates the necessary nucleophilic species in situ, effectively activating the diphenyl phosphine chloride for immediate attack on the acyl chloride. This telescoping of reactions eliminates the need for external amine bases and the associated salt waste, drastically simplifying the post-reaction workup. The operational simplicity is further enhanced by the use of robust, commodity solvents such as toluene or xylene, which facilitate easy phase separation and crystallization. This shift from a linear, multi-vessel process to a convergent, single-vessel operation represents a paradigm shift in process chemistry, offering a direct route to high-purity products with minimized unit operations and reduced capital expenditure requirements for manufacturing facilities.
Mechanistic Insights into Sodium-Mediated One-Pot Acylation
The core of this innovative synthesis lies in the precise orchestration of reagent addition and temperature control to manage the reactivity of the sodium-ethanol system. Initially, the reaction of metallic sodium with ethanol generates sodium ethoxide, a potent base and nucleophile, within the non-polar solvent matrix. This in situ generation is critical as it avoids the handling hazards associated with pre-formed alkoxides while ensuring a fresh, highly reactive species is available for the subsequent transformation. Upon the introduction of diphenyl phosphine chloride at controlled low temperatures ranging from -10 to 20°C, the system likely forms a reactive phosphide anion or a coordinated complex that is primed for acylation. The careful thermal management at this stage prevents exothermic runaways and suppresses side reactions such as homocoupling or premature hydrolysis, ensuring that the phosphorus center remains intact and reactive for the final coupling step.
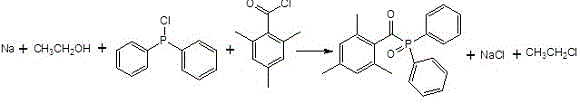
Following the activation of the phosphine component, the addition of 2,4,6-trimethylbenzoyl chloride at elevated temperatures between 70 and 90°C drives the acylation to completion. The steric bulk of the mesityl group on the acyl chloride typically hinders nucleophilic attack, but the high energy input combined with the activated phosphorus species overcomes this barrier efficiently. From an impurity control perspective, this mechanism is superior because it avoids the oxidative conditions often required in alternative pathways, thereby preventing the formation of phosphine oxide byproducts that are difficult to separate. The final workup involves a simple aqueous quench and pH adjustment using saturated sodium bicarbonate, which effectively removes inorganic salts like sodium chloride without requiring complex extraction protocols. This mechanistic elegance translates directly into the reported liquid spectrum content of approximately 99%, demonstrating the process's ability to deliver pharmaceutical-grade purity suitable for high-end electronic applications.
How to Synthesize 2,4,6-trimethylbenzoyl-diphenylphosphine oxide Efficiently
Implementing this synthesis route requires strict adherence to the sequential addition protocol and thermal profiles outlined in the patent data to ensure safety and reproducibility. The process begins with the suspension of sodium in a dry aromatic solvent, followed by the controlled dosing of ethanol to initiate the activation phase. Once the active species are generated, the phosphine chloride is introduced under cooling to manage the exotherm, followed by the heating phase for the acylation step. Detailed standardized operating procedures regarding stirring rates, addition times, and crystallization parameters are essential for transferring this laboratory success to pilot and commercial scales.
- Preparation of Reaction System: Add solvent (toluene/xylene) and metallic sodium to the reactor, then slowly introduce ethanol at 50-70°C to generate the active species in situ.
- Sequential Addition of Reagents: Carefully add diphenyl phosphine chloride at low temperatures (-10 to 20°C), followed by 2,4,6-trimethylbenzoyl chloride at elevated temperatures (70-90°C).
- Workup and Purification: Quench with water, separate layers, neutralize with sodium bicarbonate, and crystallize the organic phase to obtain >90% yield of pure TPO.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this one-pot technology offers tangible strategic advantages that extend beyond mere chemical yield. The elimination of triethylamine and the associated distillation steps results in a drastic reduction in raw material costs and utility consumption, directly improving the gross margin profile of the final product. By simplifying the manufacturing workflow, facilities can achieve higher throughput rates with existing infrastructure, effectively increasing capacity without the need for significant capital investment in new reactors or separation columns. This operational efficiency also mitigates supply chain risks associated with the sourcing of specialized reagents, as the process relies on widely available commodity chemicals like sodium metal and ethanol. Consequently, manufacturers can secure a more stable and resilient supply of high-purity TPO, ensuring continuity for downstream customers in the coatings and electronics sectors.
- Cost Reduction in Manufacturing: The economic benefits of this process are primarily derived from the removal of expensive acid scavengers and the energy savings associated with skipping distillation. By avoiding the generation of massive quantities of amine salt waste, companies also see a substantial decrease in waste disposal fees and environmental compliance costs. The high yield stability above 90% ensures that raw material utilization is maximized, reducing the effective cost per kilogram of the active ingredient. Furthermore, the simplified purification via crystallization lowers the demand for chromatographic media or complex solvent exchanges, contributing to an overall leaner cost structure.
- Enhanced Supply Chain Reliability: Relying on a robust one-pot synthesis reduces the number of critical control points in the manufacturing chain, thereby minimizing the risk of batch failures or delays. The use of common solvents like toluene and xylene ensures that supply disruptions for exotic reagents are unlikely to halt production. This reliability is crucial for maintaining just-in-time delivery schedules for major clients in the UV curing industry. Additionally, the shorter cycle time per batch allows for more flexible production planning, enabling suppliers to respond more agilely to fluctuations in market demand without building excessive inventory buffers.
- Scalability and Environmental Compliance: The inherent safety and simplicity of the reaction conditions make this process highly amenable to scale-up from pilot plants to multi-ton commercial reactors. The absence of hazardous oxidation steps and the reduction in volatile organic compound (VOC) emissions from distillation align perfectly with modern green chemistry initiatives. This environmental compatibility facilitates easier permitting and regulatory approval in jurisdictions with strict emission standards. As global regulations tighten around chemical manufacturing, adopting such a clean technology future-proofs the supply chain against potential regulatory shutdowns or fines, ensuring long-term operational viability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel TPO synthesis route. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this technology into their existing portfolios.
Q: How does the one-pot method improve yield compared to traditional TPO synthesis?
A: The one-pot method eliminates the intermediate isolation and distillation steps required in the traditional diphenyl ethoxy phosphine route. By avoiding the formation and subsequent removal of large quantities of triethylamine hydrochloride salt, the process minimizes product loss during workup, stabilizing yields above 90%.
Q: What are the primary cost drivers reduced by this new synthetic route?
A: Significant cost reductions are achieved by removing the need for expensive acid-binding agents like triethylamine and eliminating the energy-intensive distillation step. Additionally, the simplified waste stream reduces environmental compliance and disposal costs associated with amine salts.
Q: Is this process suitable for large-scale commercial manufacturing of photoinitiators?
A: Yes, the process uses common industrial solvents like toluene and xylene and operates at manageable temperature ranges. The high conversion rate and simplified purification via crystallization make it highly scalable for producing metric ton quantities of high-purity TPO.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,4,6-trimethylbenzoyl-diphenylphosphine oxide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to meet the rigorous demands of the global optoelectronic and polymer industries. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN107304220B are fully realized in practical manufacturing settings. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of TPO meets the highest performance standards required for UV curing applications. Our commitment to technical excellence allows us to navigate the complexities of process optimization, delivering consistent quality that supports our clients' product development goals.
We invite you to collaborate with us to leverage these cutting-edge synthesis methods for your specific application needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements and quality targets. We encourage potential partners to reach out for specific COA data and route feasibility assessments to understand how our optimized manufacturing capabilities can drive value for your organization. Let us be your strategic partner in securing a reliable, high-quality supply of next-generation photoinitiators.
