Advanced Manufacturing of Vecuronium Bromide via Optimized Epiandrosterone Route
Advanced Manufacturing of Vecuronium Bromide via Optimized Epiandrosterone Route
The pharmaceutical industry continuously seeks robust synthetic pathways for critical neuromuscular blocking agents, and Vecuronium Bromide stands out as a paramount example of a non-depolarizing muscle relaxant essential for modern anesthesia. Patent CN101684139A discloses a groundbreaking synthesis process that fundamentally shifts the production paradigm from expensive, complex precursors to a more accessible and economically viable starting material, epiandrosterone. This technical advancement addresses long-standing inefficiencies in the manufacturing of steroid-based muscle relaxants by streamlining the reaction sequence into eight distinct, high-yield steps. The structural integrity of the final product, characterized by its specific stereochemistry at the 2-beta and 16-beta positions, is meticulously preserved throughout this optimized route, ensuring therapeutic efficacy comparable to existing standards while offering substantial operational improvements.
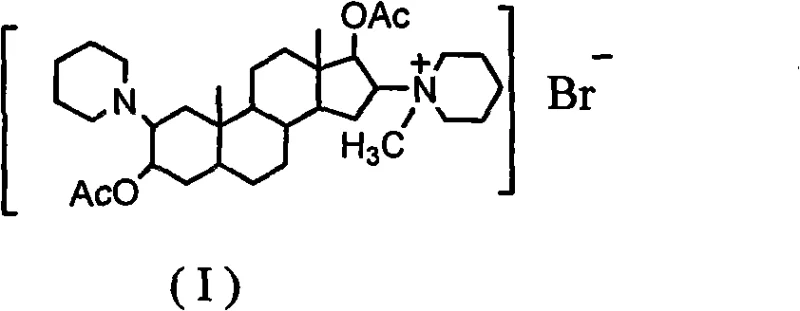
For procurement managers and supply chain directors, the implications of this patent are profound, as it promises a reliable vecuronium bromide supplier capable of delivering high-purity intermediates with reduced lead times. The transition away from scarce raw materials mitigates supply chain volatility, while the simplified purification protocols reduce the dependency on specialized chromatographic equipment. By adopting this methodology, manufacturers can achieve cost reduction in pharmaceutical intermediates manufacturing without compromising on the stringent purity specifications required for injectable anesthetics. The following analysis delves into the mechanistic nuances and commercial viability of this process, providing a comprehensive roadmap for stakeholders looking to optimize their production capabilities for this high-value API.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Vecuronium Bromide relied heavily on methodologies derived from Pancuronium Bromide production, which presented significant economic and operational bottlenecks for large-scale manufacturers. The traditional route typically utilized 5α-androstane-2-alkene-17-ketone as a starting material, a precursor that is not only prohibitively expensive but also difficult to source in bulk quantities consistent with commercial demand. Furthermore, the conventional synthetic pathway often necessitated the repeated use of column chromatography for purification at multiple stages, a technique that is notoriously labor-intensive, solvent-heavy, and challenging to scale beyond pilot plant operations. These factors combined to create a high-cost production environment with extended cycle times, making it difficult for suppliers to respond agilely to market fluctuations or sudden increases in surgical demand.
In addition to the economic drawbacks, the older methods frequently lacked detailed reporting on specific reaction conditions for certain transformation steps, leading to reproducibility issues and batch-to-batch variability in impurity profiles. The reliance on harsh reagents and the generation of complex waste streams further complicated the environmental compliance landscape, forcing manufacturers to invest heavily in waste treatment infrastructure. For a procurement manager evaluating potential partners, these inefficiencies translate into higher unit costs and increased risk of supply disruption. The inability to easily purify intermediates without chromatography meant that scaling up production often resulted in diminished yields and compromised quality, creating a significant barrier to entry for new competitors and limiting the overall capacity of the global supply chain for this critical anesthetic adjunct.
The Novel Approach
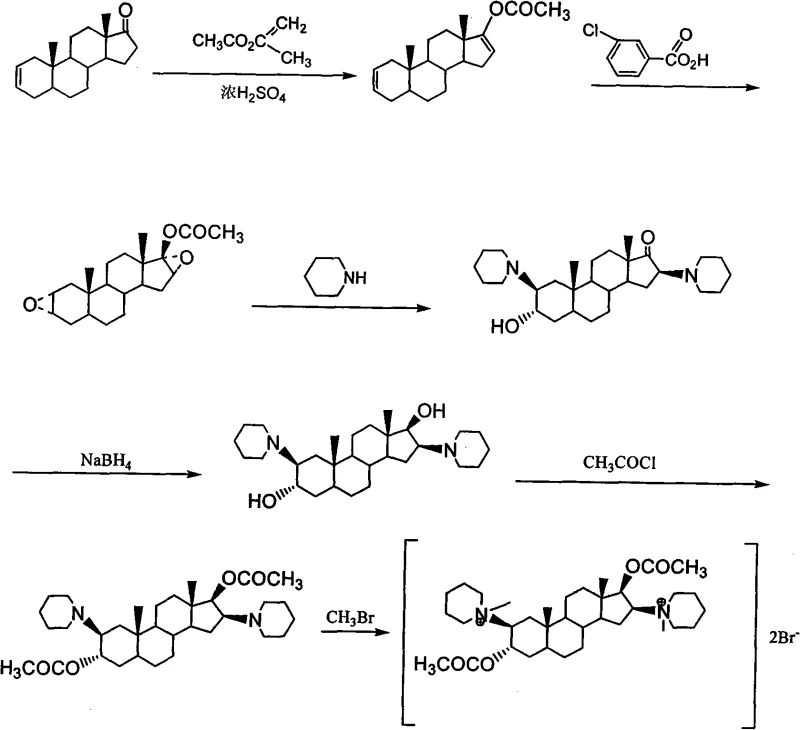
The innovative process detailed in the patent introduces a strategic pivot by utilizing epiandrosterone as the foundational building block, a steroid that is both commercially abundant and cost-effective compared to traditional precursors. This new eight-step synthesis begins with a straightforward esterification to form the sulfonyl ester, followed by a controlled elimination reaction to generate the key enone intermediate. By optimizing reaction conditions, such as using tosic acid as a catalyst during the enolization and esterification phase, the process achieves remarkable conversion rates, with yields reported up to 83.3% for critical intermediates. This approach effectively bypasses the need for the expensive 5α-androstane-2-alkene-17-ketone, thereby decoupling the production cost from the volatility of niche steroid markets and establishing a more stable economic baseline for manufacturing.
Moreover, the novel route incorporates a highly efficient epoxidation step using hydrogen peroxide and phthalic anhydride, which not only ensures high stereoselectivity but also generates water as the primary byproduct, significantly reducing the environmental footprint of the operation. The subsequent ring-opening and addition reactions with piperidine are conducted under conditions that favor the desired beta-configuration, minimizing the formation of diastereomeric impurities that would otherwise require complex separation techniques. Perhaps most importantly for industrial scalability, the final purification steps utilize recrystallization from solvents like acetone rather than column chromatography, drastically simplifying the downstream processing. This streamlined workflow enhances the commercial scale-up of complex pharmaceutical intermediates, allowing manufacturers to increase throughput while maintaining rigorous quality control standards essential for regulatory approval.
Mechanistic Insights into Steroid Functionalization and Quaternization
The core chemical innovation in this synthesis lies in the precise manipulation of the steroid backbone to install the requisite nitrogen functionalities and acetoxy groups with high regio- and stereoselectivity. The epoxidation of the diene intermediate (V) using hydrogen peroxide in the presence of phthalic anhydride generates a peroxyacid in situ, which selectively attacks the electron-rich double bonds to form the diepoxy compound (VI). This step is critical as it sets the stereochemistry for the subsequent nucleophilic attack by piperidine. The ring-opening reaction proceeds via an SN2-like mechanism where the piperidine nitrogen attacks the less hindered carbon of the epoxide ring, resulting in the trans-diaxial arrangement of the hydroxyl and amino groups. This mechanistic pathway ensures that the 2-beta and 16-beta configurations are established correctly, which is vital for the biological activity of the final neuromuscular blocker.
Following the installation of the piperidine rings, the reduction of the 17-ketone to the 17-beta-hydroxyl group using potassium borohydride is executed under mild conditions to prevent the degradation of the sensitive amine functionalities. The subsequent acetylation step protects these hydroxyl groups, stabilizing the molecule for the final quaternization reaction. The formation of the quaternary ammonium salt is achieved by reacting the tertiary amine intermediate with bromomethane, a reaction that must be carefully controlled to avoid over-alkylation or side reactions. The choice of solvent and temperature during this final step is paramount; the patent specifies the use of ether and acetone mixtures at low temperatures to precipitate the product efficiently. This meticulous control over reaction parameters minimizes the formation of organic impurities, ensuring that the final API meets the stringent purity specifications required for parenteral administration without the need for extensive chromatographic purification.
How to Synthesize Vecuronium Bromide Efficiently
The synthesis of Vecuronium Bromide via this optimized epiandrosterone route represents a significant leap forward in process chemistry, offering a clear path from raw materials to the final quaternary ammonium salt. The procedure is designed to be robust and scalable, leveraging common reagents and standard unit operations that are readily available in most fine chemical manufacturing facilities. By adhering to the specific reaction conditions outlined in the patent, such as the precise stoichiometry of tosic acid and the controlled addition of hydrogen peroxide, manufacturers can reproduce the high yields and purity levels demonstrated in the examples. The following guide summarizes the critical operational phases required to execute this synthesis successfully, serving as a foundational reference for process engineers and R&D teams aiming to implement this technology.
- Esterify epiandrosterone with Tosyl chloride to form epiandrosterone sulfonyl ester.
- Perform elimination and dehydration using 2,6-lutidine to generate 5Alpha-androst-2-alkene-17-ketone.
- Conduct enolization and esterification with isopropenyl acetate to form the diene intermediate.
- Execute epoxidation using hydrogen peroxide and phthalic anhydride to create the diepoxy compound.
- Open the epoxy rings via addition reaction with piperidine to introduce the nitrogen groups.
- Reduce the ketone group using potassium borohydride to form the diol intermediate.
- Acetylate the hydroxyl groups using acetic anhydride to protect the positions.
- Finalize the synthesis by reacting with bromomethane to form the quaternary ammonium salt.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders responsible for sourcing and logistics, the adoption of this synthesis process offers tangible benefits that extend far beyond simple chemical efficiency. The primary advantage lies in the drastic reduction of raw material costs, driven by the substitution of expensive, specialized steroids with widely available epiandrosterone. This shift not only lowers the direct cost of goods sold but also insulates the supply chain from the price volatility often associated with niche pharmaceutical intermediates. Furthermore, the elimination of column chromatography in the later stages of synthesis translates to significant savings in solvent consumption and labor hours, directly impacting the operating expenditure of the manufacturing facility. These efficiencies allow suppliers to offer more competitive pricing structures while maintaining healthy margins, a crucial factor in the highly competitive generic pharmaceutical market.
- Cost Reduction in Manufacturing: The utilization of epiandrosterone as a starting material fundamentally alters the cost structure of Vecuronium Bromide production by removing the dependency on high-cost precursors. Additionally, the high yields achieved in key steps, such as the acetylation and reduction phases, minimize material loss and maximize the output per batch. The avoidance of chromatographic purification further reduces the consumption of expensive silica gel and organic solvents, leading to substantial cost savings in waste disposal and raw material procurement. These cumulative effects result in a leaner, more cost-effective manufacturing process that enhances the overall profitability of the supply chain.
- Enhanced Supply Chain Reliability: By relying on commoditized starting materials like epiandrosterone and common reagents such as piperidine and acetic anhydride, manufacturers can secure a more stable and resilient supply base. This reduces the risk of production stoppages due to the unavailability of specialized chemicals, ensuring consistent delivery schedules for downstream pharmaceutical clients. The simplified process flow also shortens the overall production cycle time, enabling faster response to market demand surges and improving the agility of the supply network. This reliability is paramount for hospital formularies and anesthesia providers who depend on uninterrupted access to critical muscle relaxants.
- Scalability and Environmental Compliance: The process is inherently designed for scale, utilizing reaction conditions that do not require extreme temperatures or pressures, thus reducing energy consumption and equipment stress. The use of hydrogen peroxide as an oxidant generates water as a benign byproduct, significantly lowering the environmental burden and simplifying wastewater treatment protocols. This alignment with green chemistry principles not only reduces regulatory compliance costs but also enhances the corporate sustainability profile of the manufacturer. The ability to scale this process from pilot batches to multi-ton annual production without significant re-engineering makes it an ideal candidate for long-term commercial deployment.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical feasibility and commercial implications of this synthesis method. These insights are derived directly from the patent data and are intended to clarify the operational advantages for potential partners and investors. Understanding these details is crucial for making informed decisions about technology transfer and supply agreements.
Q: Why is epiandrosterone preferred over traditional precursors for Vecuronium Bromide synthesis?
A: Epiandrosterone is significantly cheaper and more readily available than the traditional 5α-androstane-2-alkene-17-ketone precursor used in pancuronium bromide synthesis. This switch drastically reduces raw material costs and simplifies the supply chain logistics for large-scale manufacturing.
Q: How does this process improve environmental compliance compared to older methods?
A: The novel process utilizes hydrogen peroxide and phthalic anhydride for the epoxidation step, where the primary oxidation byproduct is water. This eliminates the generation of heavy metal waste or toxic organic byproducts often associated with traditional oxidants, facilitating easier waste treatment and regulatory compliance.
Q: What are the key operational advantages regarding purification in this synthesis?
A: Unlike previous methods that required repeated column chromatography for purification, this optimized route allows for recrystallization using solvents like acetone in the final steps. This simplifies the post-reaction treatment, reduces solvent consumption, and makes the process much more suitable for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vecuronium Bromide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and compliant manufacturing processes for high-value pharmaceutical intermediates like Vecuronium Bromide. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of the global healthcare market. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of intermediate or API we produce meets the highest international standards. Our capability to implement the optimized epiandrosterone route positions us as a strategic partner capable of delivering cost-effective solutions without compromising on quality or safety.
We invite procurement leaders and R&D directors to engage with our technical procurement team to discuss how this advanced synthesis technology can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic potential of this route for your portfolio. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project needs, ensuring a seamless integration of high-quality Vecuronium Bromide into your manufacturing pipeline.
