Revolutionizing Quinone Production via Ultrasonic-Assisted Indirect Electrosynthesis for Global Markets
Introduction to Advanced Quinone Synthesis Technologies
The global demand for high-purity quinone compounds, essential intermediates in the pharmaceutical and agrochemical sectors, has driven significant innovation in synthetic methodologies. Patent CN112626547B introduces a groundbreaking approach to the indirect electrosynthesis of quinone compounds, specifically targeting 1,4-naphthoquinone, 2-methyl-1,4-naphthoquinone (menadione), and 9,10-anthraquinone. This technology leverages a sophisticated cerium-mediated redox cycle within a novel mixed acid electrolyte system, enhanced by ultrasonic assistance to overcome traditional limitations in mass transfer and reaction kinetics. For R&D directors and procurement specialists seeking a reliable quinone supplier, this patent represents a pivotal shift towards greener, more efficient manufacturing processes that eliminate the reliance on toxic stoichiometric oxidants like chromic anhydride. The integration of ultrasound with electrochemical generation of the active oxidant ensures superior control over reaction parameters, resulting in exceptional product purity and yield profiles that meet stringent international quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of quinone compounds has been plagued by significant environmental and efficiency challenges. Traditional gas-phase catalytic oxidation methods, such as those employed for 1,4-naphthoquinone, often suffer from low conversion rates, typically hovering around 20 percent, which necessitates extensive recycling and purification steps. Alternatively, liquid-phase oxidation using chromic anhydride generates massive quantities of hazardous chromium-containing acidic wastewater, posing severe disposal costs and environmental compliance risks. Furthermore, conventional indirect electrosynthesis methods utilizing single acid media, such as pure sulfuric acid or pure methanesulfonic acid, face distinct drawbacks; sulfuric acid systems exhibit poor solubility for high concentrations of cerium ions leading to precipitation, while pure methanesulfonic acid systems incur high energy consumption due to solution viscosity and require expensive electrode materials to withstand corrosive conditions. These legacy processes often result in low space-time yields and inconsistent product quality due to the formation of stubborn by-products like binaphthyl.
The Novel Approach
The methodology disclosed in CN112626547B fundamentally reengineers the electrosynthesis landscape by introducing a synergistic mixed acid electrolyte composed of methanesulfonic acid and sulfuric acid. This specific formulation allows for the stable generation of high-concentration Ce4+ ions without the precipitation issues associated with pure sulfuric acid, while simultaneously reducing the viscosity and energy consumption typical of pure methanesulfonic acid systems. By coupling this optimized electrolyte with ultrasonic assistance during the chemical oxidation step, the process achieves rapid emulsification of the organic substrate and the aqueous oxidant phase. This enhancement drastically reduces reaction times and suppresses side reactions, enabling the continuous regeneration of the cerium mediator in a plate-and-frame electrolytic cell. The result is a robust, scalable process capable of delivering high-purity quinones with minimal waste generation, positioning it as a superior alternative for cost reduction in fine chemical manufacturing.
Mechanistic Insights into Cerium-Mediated Indirect Electrosynthesis
The core of this innovative process lies in the efficient cycling of cerium ions between the +3 and +4 oxidation states within a carefully balanced mixed acid environment. In the electrolytic cell, Ce3+ ions are anodically oxidized to Ce4+, which serves as a powerful, soluble chemical oxidant. The presence of sulfate ions from the sulfuric acid component facilitates the formation of cerium sulfate complexes, which, contrary to previous assumptions, stabilizes the solution against precipitation at higher concentrations while moderating the oxidizing power to prevent over-oxidation of the sensitive organic substrates. This mechanistic nuance is critical for maintaining high selectivity, as overly aggressive oxidants can degrade the aromatic rings or promote polymerization. The mixed acid system ensures that the Ce4+ species remains in solution long enough to effectively diffuse into the organic phase and react with the substrate, thereby maximizing the utilization of the electrical energy input.
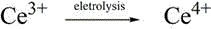
Following the electrochemical generation, the Ce4+-rich anolyte is introduced to the organic substrate under ultrasonic irradiation. The ultrasonic waves create cavitation bubbles that collapse violently, generating localized hot spots and intense micro-turbulence. This physical phenomenon dramatically increases the interfacial area between the immiscible aqueous electrolyte and the organic solvent containing the naphthalene or anthracene derivative. As the Ce4+ oxidizes the substrate to the corresponding quinone, it is reduced back to Ce3+, which is then returned to the electrolytic cell for regeneration. This closed-loop mechanism not only minimizes the consumption of the cerium mediator but also ensures that the reaction proceeds under mild thermal conditions, typically between 30°C and 60°C, preserving the structural integrity of the product and minimizing thermal degradation by-products.
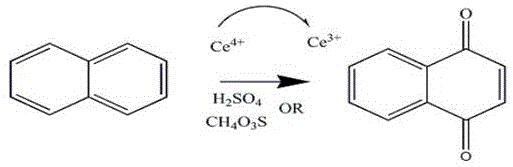
How to Synthesize 1,4-Naphthoquinone Efficiently
The synthesis of high-purity 1,4-naphthoquinone via this ultrasonic-assisted indirect electrosynthesis route involves a precise sequence of electrolyte preparation, electrochemical oxidation, and sonochemical reaction steps. The process begins with the formulation of the mixed acid electrolyte, where methanesulfonic acid and sulfuric acid are combined with cerium carbonate to achieve specific molar concentrations that optimize conductivity and solubility. Once the electrolyte is circulated through the plate-and-frame cell to generate the active Ce4+ species, it is immediately transferred to the synthesis reactor containing the naphthalene substrate dissolved in a non-polar solvent like n-heptane. The application of ultrasound at frequencies between 40Hz and 100Hz, combined with mechanical stirring, ensures homogeneous mixing and rapid reaction kinetics, allowing the transformation to proceed to completion within minutes rather than hours. Detailed standardized synthesis steps see the guide below.
- Prepare the electrolyte by mixing methanesulfonic acid (2 mol/L) and sulfuric acid (0.2-0.6 mol/L) with Ce3+ ions (0.8-1 mol/L) in deionized water.
- Circulate the electrolyte through a plate-and-frame electrolytic cell to oxidize Ce3+ to Ce4+ at the anode, maintaining a concentration below 0.8 mol/L.
- Mix the anolyte containing Ce4+ with the organic substrate (naphthalene, 2-methylnaphthalene, or anthracene) under ultrasonic assistance and mechanical stirring to obtain the quinone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ultrasonic-assisted electrosynthesis technology offers transformative benefits regarding cost structure and operational reliability. The elimination of stoichiometric heavy metal oxidants like chromium removes the substantial costs associated with hazardous waste treatment and regulatory compliance, directly impacting the bottom line through significant cost savings in waste management. Furthermore, the continuous nature of the plate-and-frame electrolytic system allows for uninterrupted production cycles, enhancing supply chain reliability by reducing the risk of batch-to-batch variability and equipment downtime common in batch-wise chemical oxidation processes. The ability to recycle both the aqueous electrolyte and the organic solvent phases contributes to a near-zero discharge manufacturing model, aligning with increasingly strict global environmental regulations and ensuring long-term operational sustainability without the threat of shutdowns due to environmental violations.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive and toxic stoichiometric oxidants, replacing them with electricity-driven regeneration of a catalytic amount of cerium mediator. This shift drastically reduces raw material costs and removes the financial burden of treating heavy metal wastewater, leading to substantial overall production cost optimization. Additionally, the mixed acid system lowers energy consumption compared to high-viscosity single acid systems, further enhancing the economic viability of large-scale operations.
- Enhanced Supply Chain Reliability: By utilizing a continuous flow electrolysis setup coupled with rapid sonochemical synthesis, the production lead time is significantly compressed compared to traditional batch methods. The robustness of the mixed acid electrolyte prevents clogging and precipitation issues that often halt production in conventional systems, ensuring a steady and predictable output of high-purity intermediates. This stability allows suppliers to maintain consistent inventory levels and meet tight delivery schedules for downstream pharmaceutical and agrochemical manufacturers.
- Scalability and Environmental Compliance: The modular design of the plate-and-frame electrolytic cells facilitates easy scale-up from pilot to commercial production volumes without the need for complex re-engineering of the reaction chemistry. The closed-loop recycling of solvents and acids minimizes the environmental footprint, making the facility compliant with rigorous international environmental standards. This green chemistry approach future-proofs the supply chain against tightening regulations on chemical emissions and waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ultrasonic-assisted electrosynthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear understanding of the operational parameters and benefits. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this method into existing production lines or for procurement teams assessing the quality and consistency of the supplied intermediates.
Q: How does the mixed acid system improve stability compared to single acid systems?
A: The mixed acid system comprising methanesulfonic acid and sulfuric acid prevents the precipitation of cerium sulfate that typically occurs in pure sulfuric acid at high concentrations, while maintaining lower viscosity and energy consumption compared to pure methanesulfonic acid systems.
Q: What role does ultrasonic assistance play in the synthesis of quinones?
A: Ultrasonic assistance significantly accelerates the emulsification of the organic and aqueous phases, enhancing the contact between the Ce4+ oxidant and the substrate, which reduces side reactions like binaphthyl formation and improves overall yield and selectivity.
Q: Is this electrosynthesis method suitable for large-scale industrial production?
A: Yes, the process utilizes a plate-and-frame electrolytic cell which allows for continuous circulation and regeneration of the electrolyte, facilitating zero liquid discharge and making it highly scalable for commercial manufacturing of quinone intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to meet the evolving demands of the global pharmaceutical and fine chemical industries. Our expertise extends beyond simple manufacturing; we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the ultrasonic-assisted electrosynthesis of quinones are translated into reliable, high-volume supply chains. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 1,4-naphthoquinone or menadione meets the exacting standards required for API synthesis. We are committed to delivering high-purity quinone intermediates that empower your R&D teams to develop next-generation therapeutics and agrochemicals with confidence.
We invite you to collaborate with us to leverage these cutting-edge manufacturing capabilities for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how our optimized processes can enhance your margin structures. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how NINGBO INNO PHARMCHEM can become your strategic partner in securing a sustainable and cost-effective supply of critical quinone intermediates.
