Scalable Manufacturing of High-Purity Budesonide via Optimized Prednisolone Esterification and Oxidation
The pharmaceutical industry continuously seeks robust synthetic routes for high-value corticosteroids, and the preparation of Budesonide stands as a prime example of process optimization driving commercial viability. Patent CN101863952B discloses a novel preparation method that fundamentally shifts the starting material paradigm from prednisone acetate to prednisolone, offering a streamlined five-step sequence that significantly enhances efficiency. This technical breakthrough addresses long-standing challenges in steroid synthesis, specifically focusing on yield improvement, impurity control, and the precise management of stereochemistry required for clinical efficacy. By leveraging specific catalytic systems and tightly controlled reaction parameters, this methodology achieves a total molar yield of approximately 45%, a substantial leap compared to traditional routes that often struggle to exceed 12%. For R&D directors and procurement strategists, understanding the nuances of this pathway is essential for securing a reliable supply chain of high-purity pharmaceutical intermediates that meet stringent European Pharmacopoeia standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Budesonide has relied heavily on prednisone acetate as the primary starting material, a route fraught with kinetic and thermodynamic inefficiencies. Existing literature and prior art, such as the methods referenced in the background of the patent, often suffer from slow reaction rates in key intermediate stages, particularly during the introduction of the 16-alpha hydroxyl group. These conventional pathways frequently require harsh conditions that lead to extensive by-product formation, necessitating complex purification protocols that erode overall profitability. Furthermore, the inability to effectively control the ratio of R and S isomers in the final acetalization step has been a persistent bottleneck, often resulting in products that fail to meet the strict 41-50% S-isomer requirement of the European Pharmacopoeia without costly recrystallization cycles. The cumulative effect of these inefficiencies is a low overall molar yield, typically hovering around 12%, which drastically inflates the cost of goods sold and creates supply chain vulnerabilities due to material loss at every unit operation.
The Novel Approach
In stark contrast, the methodology outlined in CN101863952B introduces a sophisticated protection-deprotection strategy starting directly from prednisolone, which is readily available as a bulk API原料. The process initiates with a highly efficient esterification using acetic anhydride and 4-methylaminopyridine, followed by a selective elimination reaction to form the critical 16-ene intermediate. This route is distinguished by its use of mild yet effective catalytic conditions, such as the use of alkali metal salts for deacetylation and potassium permanganate for oxidation under strictly controlled low-temperature environments. By systematically protecting the 11 and 21 hydroxyl groups as esters before the oxidation step, the process prevents unwanted side reactions, thereby preserving the integrity of the steroid nucleus. This strategic sequencing not only accelerates reaction kinetics but also ensures that the final acetalization with n-butyraldehyde proceeds with high stereoselectivity, delivering a product profile that consistently aligns with global regulatory expectations.
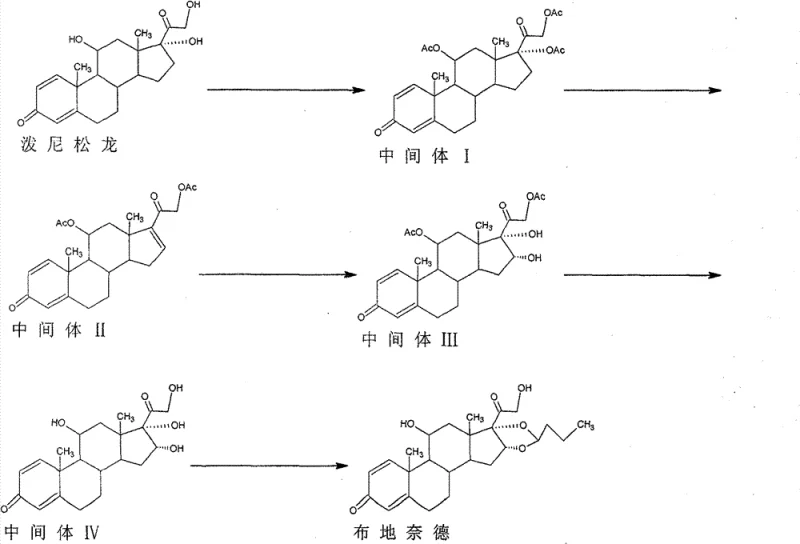
Mechanistic Insights into KMnO4-Catalyzed Oxidation and Acetalization
The core chemical innovation of this process lies in the precise execution of the oxidation and acetalization steps, which dictate both the yield and the pharmacological quality of the final API. The oxidation of the 16-ene intermediate (Intermediate II) using potassium permanganate is performed in an acidic environment at temperatures between -10°C and 0°C. This low-temperature regime is mechanistically crucial; it suppresses the oxidative cleavage of the double bond and prevents the over-oxidation of sensitive functional groups elsewhere on the molecule. The addition of sodium sulfite post-reaction serves to quench excess oxidant, ensuring a clean workup that minimizes heavy metal contamination risks. Following this, the transesterification step utilizes alcohols like methanol or ethanol under alkaline catalysis to selectively remove the acetyl protecting groups, revealing the 16α-hydroxyprednisolone structure without compromising the newly formed diol system.
Furthermore, the final condensation reaction with n-butyraldehyde is engineered to maximize the formation of the therapeutically active isomers. By employing p-toluenesulfonic acid as a catalyst in solvents such as dichloromethane, acetonitrile, or isooctane, the reaction environment is tuned to favor the formation of the 16α,17α-acetal linkage. The choice of solvent plays a pivotal role in the thermodynamic equilibrium of the R and S epimers at the 16-position. The patent data indicates that this specific catalytic system allows manufacturers to reliably achieve an S-isomer content of 41-50%, which is critical because the R-configuration possesses significantly higher anti-inflammatory activity. This level of control eliminates the need for expensive chiral separations downstream, representing a major advantage for commercial scale-up of complex pharmaceutical intermediates.
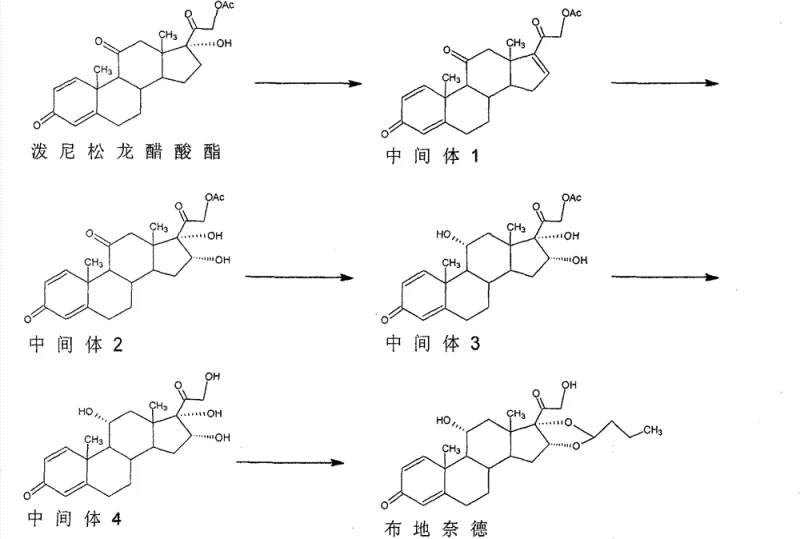
How to Synthesize Budesonide Efficiently
The synthesis of Budesonide via this optimized route requires strict adherence to the sequential addition of reagents and temperature controls defined in the patent examples. The process begins with the dissolution of prednisolone in dimethylformamide, followed by the addition of acetic anhydride and the amine catalyst to drive the initial esterification to completion within 2 to 3 hours. Subsequent steps involve careful monitoring via thin-layer chromatography (TLC) to ensure full conversion before quenching into ice water, a technique that precipitates the intermediates in high purity. The detailed standardized synthesis steps, including specific molar ratios, solvent volumes, and agitation speeds for each of the five stages, are provided in the structured guide below to assist process engineers in replicating these results.
- Esterify Prednisolone with acetic anhydride using 4-methylaminopyridine catalyst to form the triacetoxy intermediate.
- Perform catalytic deacetylation and elimination using alkali metal salts to generate the 16-ene diacetoxy intermediate.
- Oxidize the 16-ene intermediate with potassium permanganate under acidic conditions at low temperature (-10 to 0°C) to introduce 16α,17α-dihydroxy groups.
- Conduct transesterification with alcohol under alkaline conditions to remove protecting groups and obtain 16α-hydroxyprednisolone.
- Condense 16α-hydroxyprednisolone with n-butyraldehyde using p-toluenesulfonic acid to finalize the Budesonide structure with controlled isomer ratio.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers tangible benefits that extend beyond mere technical elegance, directly impacting the bottom line through operational efficiency and risk mitigation. The shift to prednisolone as a starting material leverages a commodity chemical that is widely produced and cost-effective, reducing dependency on specialized precursors that may face supply bottlenecks. Moreover, the significant improvement in overall yield translates directly into reduced raw material consumption per kilogram of finished product, lowering the variable costs associated with manufacturing. The mild reaction conditions also imply lower energy consumption for heating and cooling, contributing to a more sustainable and cost-efficient production footprint that aligns with modern green chemistry initiatives.
- Cost Reduction in Manufacturing: The elimination of harsh reaction conditions and the reduction of purification steps lead to substantial cost savings in utility and waste treatment. By avoiding the use of expensive transition metal catalysts that require rigorous removal processes, the manufacturing overhead is significantly decreased. The high yield of intermediates, particularly the oxidation step which reaches over 94% molar yield in optimized examples, ensures that valuable starting materials are not lost to side reactions, maximizing the return on investment for every batch produced.
- Enhanced Supply Chain Reliability: Utilizing prednisolone, a stable and commercially abundant API intermediate, ensures a consistent supply of raw materials不受 market fluctuations affecting niche steroids. The robustness of the reaction conditions, which tolerate slight variations without catastrophic failure, enhances the reliability of production schedules. This stability allows for better inventory planning and reduces the risk of stockouts, ensuring that downstream formulation teams receive their required volumes of high-purity Budesonide on time.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing common organic solvents and reagents that are easily sourced in bulk quantities for multi-ton production. The waste stream is simplified due to the high selectivity of the reactions, reducing the burden on effluent treatment plants and facilitating compliance with increasingly strict environmental regulations. The ability to scale from laboratory grams to commercial tons without re-optimizing the core chemistry provides a clear path for rapid capacity expansion to meet market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Budesonide synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a factual basis for decision-making. Understanding these specifics helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What is the overall molar yield of this Budesonide synthesis method?
A: According to patent CN101863952B, the total molar yield of the product can reach approximately 45%, which is a significant improvement over conventional methods that often yield around 12%.
Q: How does this process control the R/S isomer ratio of Budesonide?
A: The process utilizes p-toluenesulfonic acid as a catalyst and specific solvents like dichloromethane or acetonitrile during the final condensation step. This allows precise control over the S-type isomer proportion, ensuring it remains between 41% and 50%, compliant with European Pharmacopoeia standards.
Q: Why is Potassium Permanganate used at low temperatures in this synthesis?
A: Potassium permanganate is used for the oxidation step to introduce hydroxyl groups at the 16 and 17 positions. Conducting this reaction at low temperatures (-10 to 0°C) is critical to reduce side reactions, prevent over-oxidation, and ensure the safety and selectivity of the process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Budesonide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of process robustness in the production of high-value corticosteroids like Budesonide. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a GMP-compliant environment. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced HPLC and NMR capabilities to verify that every batch meets the 41-50% S-isomer ratio and 99% purity standards required by global pharmacopoeias. Our commitment to quality assurance ensures that our clients receive intermediates that are ready for immediate formulation without additional purification burdens.
We invite pharmaceutical partners to engage with our technical procurement team to discuss how this optimized synthesis route can enhance your supply chain resilience. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this high-yield methodology. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume requirements, ensuring a seamless transition to a more efficient and reliable source of Budesonide.
