Advanced Synthesis of 16α-Hydroxyprednisolone for Commercial Budesonide Production
The pharmaceutical industry continuously seeks robust synthetic routes for high-value corticosteroids, particularly for respiratory therapies where purity is paramount. Patent CN115819487A introduces a groundbreaking preparation method for a key budesonide intermediate, specifically 16α-hydroxyprednisolone (Compound V). This innovation addresses long-standing challenges in steroid synthesis by utilizing tetraene acetate (Compound I) as the starting material instead of the traditional prednisolone acetate. The disclosed process streamlines production into four critical chemical transformations: oxidation, bromohydrin formation, debromination, and a highly controlled hydrolysis. By integrating a specific antioxidant strategy during the final hydrolysis step, this method effectively suppresses the formation of oxidative and rearrangement impurities that have historically plagued the quality of budesonide precursors. For global procurement teams and R&D directors, this patent represents a significant leap forward in cost reduction in pharmaceutical intermediates manufacturing, offering a pathway to >99% purity intermediates essential for high-quality asthma medications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of budesonide has relied heavily on prednisolone acetate as the primary feedstock. This conventional route is fraught with inefficiencies, typically requiring a lengthy sequence of esterification, reduction, elimination, oxidation, condensation, and hydrolysis reactions. A major bottleneck in these traditional processes is the necessity to protect the 11-position hydroxyl group to prevent unwanted side reactions during subsequent dehydration steps. This protection-deprotection strategy not only extends the reaction timeline but also introduces additional reagents and purification stages, drastically increasing the cost of goods sold (COGS). Furthermore, the dehydration reactions involved often suffer from poor regioselectivity and generate substantial amounts of byproducts, leading to lower overall yields and complicating the downstream purification of the final active pharmaceutical ingredient (API). These structural inefficiencies make the conventional supply chain vulnerable to fluctuations in raw material costs and extended lead times.
The Novel Approach
In stark contrast, the methodology outlined in CN115819487A leverages tetraene acetate (3TR) as a superior starting point, fundamentally altering the synthetic landscape. This novel approach bypasses the cumbersome protection steps entirely, utilizing a direct four-step sequence to construct the necessary stereochemistry and functional groups. The process begins with a selective oxidation to install the 11-oxygen functionality, followed by a precise bromohydrin addition and subsequent debromination to establish the 16α-hydroxyl configuration. The culmination of this route is a specialized hydrolysis step that cleaves the 21-acetate group while preserving the integrity of the sensitive steroid nucleus. As illustrated in the comprehensive reaction scheme below, this streamlined pathway minimizes unit operations and maximizes atom economy.
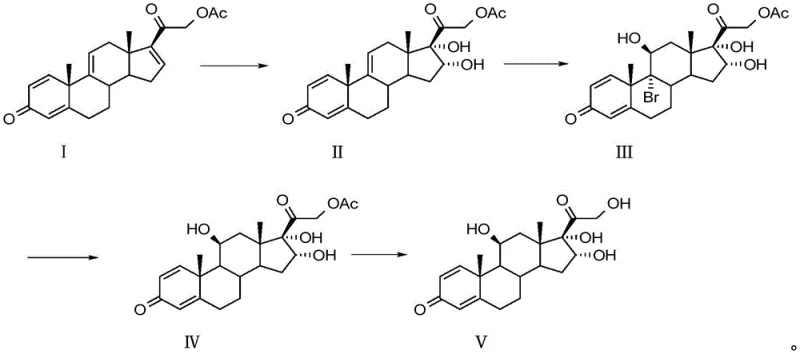
By eliminating the dehydration and protection hurdles inherent to the prednisolone-based route, this new method offers a reliable pharmaceutical intermediates supplier a distinct competitive advantage in terms of throughput and operational simplicity. The reduction in step count directly correlates to reduced solvent consumption, lower waste generation, and a more robust process capable of consistent commercial scale-up.
Mechanistic Insights into Antioxidant-Stabilized Hydrolysis
The crown jewel of this patented technology lies in the mechanistic understanding and control of the final hydrolysis step (Step S4). The inventors identified that the conversion of Compound IV to Compound V under alkaline conditions is highly susceptible to oxidative degradation. In the absence of specific controls, atmospheric oxygen can attack the reactive side chain, leading to the formation of 17-ketone and 21-aldehyde impurities through a complex rearrangement mechanism. These impurities are structurally similar to the target molecule, making them exceptionally difficult to remove via standard crystallization or chromatography, thereby compromising the purity profile required for regulatory approval.
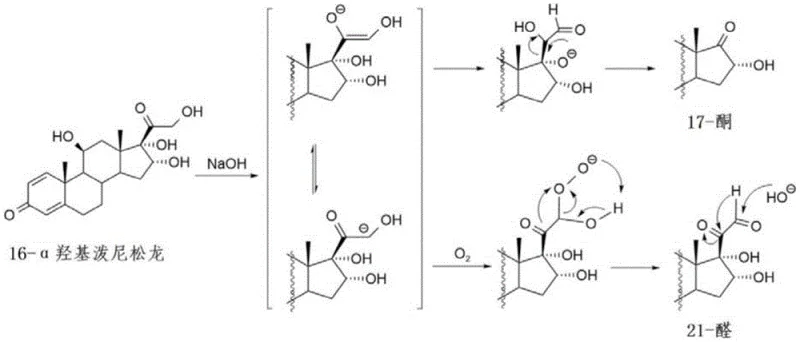
To counteract this, the process incorporates tert-butyl hydroquinone (TBHQ) as a critical process additive. Unlike common antioxidants such as BHA or BHT, which may carry carcinogenic risks or fail to suppress specific steroid rearrangements, TBHQ demonstrates superior efficacy in this specific solvent system. The mechanism involves the scavenging of free radicals generated during the base-catalyzed hydrolysis, effectively interrupting the oxidation cascade before rearrangement can occur. Crucially, the patent specifies a narrow mass ratio window for TBHQ (0.002:1 to 0.006:1 relative to Compound IV). Deviating from this range results in either insufficient impurity suppression or the introduction of a pink discoloration in the final product, which fails pharmacopoeia appearance tests. This precise control ensures the production of high-purity pharmaceutical intermediates with a single impurity content maintained below 0.50%, setting a new benchmark for quality in corticosteroid synthesis.
How to Synthesize 16α-Hydroxyprednisolone Efficiently
The execution of this synthesis requires strict adherence to temperature controls and reagent stoichiometry to ensure safety and reproducibility. The oxidation step utilizes potassium permanganate at sub-zero temperatures (-10°C to 0°C) to prevent over-oxidation of the diene system. Subsequent bromination and debromination steps employ mild conditions to preserve the delicate 1,4-diene-3-one structure of the A-ring. The final hydrolysis is conducted in a mixed solvent system of dichloromethane and methanol with sodium hydroxide, strictly maintaining the temperature between -5°C and 10°C. Detailed standardized operating procedures for each reaction stage, including workup and purification protocols, are essential for transferring this laboratory success to pilot and commercial plants.
- Oxidation of Tetraene Acetate (Compound I) using potassium permanganate in organic solvent at low temperature to form Compound II.
- Bromohydrin reaction of Compound II with N-bromosuccinimide or dibromohydantoin to generate Compound III.
- Debromination of Compound III using zinc powder and thioglycolic acid to yield Compound IV.
- Hydrolysis of Compound IV with alkali in the presence of tert-butyl hydroquinone (TBHQ) to obtain high-purity Compound V.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology translates into tangible strategic benefits beyond mere chemical elegance. The shift to a shorter, four-step synthesis from readily available tetraene acetate significantly de-risks the supply chain by reducing dependency on complex, multi-step precursor availability. The elimination of protection groups and dehydration steps removes entire categories of expensive reagents and hazardous waste streams, driving down the variable cost of production. Furthermore, the high purity of the intermediate (>99%) reduces the burden on downstream processing facilities, allowing for faster batch release times and higher throughput of the final budesonide API.
- Cost Reduction in Manufacturing: The streamlined synthetic route eliminates the need for costly protecting group chemistry and reduces the total number of isolation steps. By avoiding the use of expensive reagents required for traditional dehydration and protection strategies, the overall material cost is substantially lowered. Additionally, the high yield and purity minimize the loss of valuable steroid intermediates during purification, leading to significant savings in raw material consumption and waste disposal costs associated with failed batches or extensive recrystallization.
- Enhanced Supply Chain Reliability: Utilizing tetraene acetate as a starting material leverages a more stable and abundant supply base compared to modified prednisolone derivatives. The simplified process flow reduces the potential for bottlenecks at intermediate stages, ensuring a more consistent flow of materials to the final formulation units. This robustness allows manufacturers to maintain tighter delivery schedules and respond more agilely to market demand fluctuations for respiratory medications without the risk of prolonged campaign delays caused by complex synthetic failures.
- Scalability and Environmental Compliance: The reaction conditions described, such as low-temperature oxidations and ambient pressure hydrolysis, are inherently safer and easier to scale from kilogram to multi-ton quantities. The reduction in solvent usage and the avoidance of heavy metal catalysts or toxic protecting agents align with modern green chemistry principles. This facilitates easier regulatory compliance regarding environmental discharge and worker safety, making the process more sustainable and attractive for long-term commercial investment in GMP-compliant facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. Understanding these nuances is critical for R&D teams evaluating technology transfer and procurement officers assessing supplier capabilities. The answers are derived directly from the experimental data and mechanistic insights provided in the patent documentation.
Q: Why is TBHQ added during the hydrolysis step?
A: TBHQ acts as a critical antioxidant that prevents air oxidation of the steroid side chain during alkaline hydrolysis. Without it, oxidative impurities form and rearrange into difficult-to-remove byproducts, lowering purity and causing discoloration.
Q: What is the advantage of using Tetraene Acetate over Prednisolone Acetate?
A: Using Tetraene Acetate (Compound I) eliminates the need for complex protection and dehydration steps required when starting from Prednisolone Acetate. This shortens the synthetic route, reduces side reactions, and significantly improves overall yield.
Q: What purity levels can be achieved with this method?
A: The patented method consistently produces 16α-hydroxyprednisolone (Compound V) with a purity greater than 99%, with maximum single impurity content controlled below 0.50%, meeting stringent pharmacopoeia standards for downstream budesonide synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 16α-Hydroxyprednisolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the production of life-saving respiratory drugs. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate details of the CN115819487A process are executed with precision. We operate stringent purity specifications and maintain rigorous QC labs equipped to detect trace impurities at the ppm level, guaranteeing that every batch of 16α-hydroxyprednisolone meets the exacting standards required for budesonide synthesis. Our commitment to quality assurance ensures that our clients receive intermediates that facilitate smooth downstream processing and final API registration.
We invite global partners to collaborate with us to leverage this advanced technology for their supply chains. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our implementation of this patent can enhance your operational efficiency and product quality.
