Advanced One-Pot Synthesis of Triazolinethione Compounds for Commercial Fungicide Production
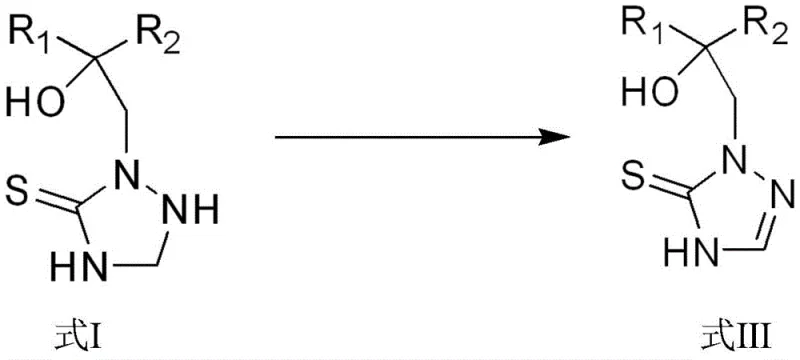
The pharmaceutical and agrochemical industries are constantly seeking more efficient pathways to synthesize critical active ingredients, particularly broad-spectrum fungicides like prothioconazole. Patent CN111269190A introduces a groundbreaking one-pot method for synthesizing triazoline thioketone compounds, addressing long-standing inefficiencies in traditional manufacturing. This technology transforms the production landscape by utilizing a sequential oxidation-reduction strategy that converts triazoline thioketone precursors (Formula I) directly into high-value intermediates (Formula III) within a single reactor vessel. By eliminating the need for isolation of unstable intermediates and avoiding harsh reaction conditions, this innovation offers a robust solution for reliable agrochemical intermediate supplier networks aiming to enhance sustainability and throughput.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of triazolinethione compounds has been plagued by significant operational hurdles that impede cost reduction in fungicide manufacturing. Prior art methods, such as those disclosed in WO9616048a1, often rely on reacting triazole compounds with elemental sulfur under high temperatures in polar solvents, resulting in prolonged reaction times and difficult removal of residual sulfur. Other approaches, like the use of n-butyllithium described in US5789430, necessitate ultra-low temperature conditions and strictly anhydrous environments, posing severe safety risks and requiring expensive specialized equipment. Furthermore, oxidative methods utilizing ferric trichloride, as seen in CN1411450A, generate substantial amounts of heavy metal waste, creating immense pressure on wastewater treatment facilities and complicating the purification process due to the solubility of iron salts in alcohol solvents.
The Novel Approach
The methodology outlined in CN111269190A represents a paradigm shift by employing molecular oxygen as a green oxidant followed by a mild reduction step, effectively bypassing the drawbacks of previous techniques. This novel approach allows for the direct conversion of raw materials into the target triazolinethione structure without the need for intermediate isolation, thereby streamlining the workflow and minimizing material loss. The process operates under mild temperatures ranging from 0 to 50 degrees Celsius, significantly lowering energy consumption compared to high-temperature sulfurization methods. By integrating oxidation and reduction into a single pot, the technique not only simplifies the post-treatment process but also ensures high raw material conversion rates and superior product purity, making it an ideal candidate for commercial scale-up of complex agrochemical intermediates.
Mechanistic Insights into Oxygen-Mediated Oxidation and Reduction
The core of this synthetic breakthrough lies in the controlled oxidation of the triazolidine thione compound (Formula I) to form a disulfide dimer intermediate (Formula II), followed immediately by reduction back to the monomeric triazolinethione (Formula III). In the first stage, oxygen acts as the oxidant in the presence of a diluent such as methanol, ethanol, or acetonitrile, facilitating the formation of the S-S bond without generating toxic byproducts. This oxidative step is carefully monitored to ensure complete conversion before the introduction of the reducing agent. The subsequent reduction phase utilizes agents like sodium thiosulfate or sodium sulfite to cleave the disulfide bond, regenerating the thione functionality with high fidelity. This two-step sequence within one vessel prevents the accumulation of impurities that typically arise from multi-step isolations.
Impurity control is inherently built into this mechanism through the choice of reagents and the one-pot nature of the reaction. Unlike iron-catalyzed oxidations where metal residues persist, the use of oxygen and soluble sulfur-based reducing agents ensures that inorganic byproducts are easily removed during the aqueous workup. The reaction stoichiometry is optimized with a molar ratio of raw material to oxygen and reductant typically between 1:0.1-2:0.1-2, ensuring that excess reagents do not lead to side reactions. The result is a product with purity levels exceeding 95 percent, achieved through a mechanism that prioritizes atom economy and minimizes the generation of hazardous waste streams, aligning perfectly with modern green chemistry principles.
How to Synthesize Prothioconazole Efficiently
For manufacturers targeting the production of prothioconazole, a leading triazole fungicide, this patent provides a clear roadmap starting from the specific precursor shown in Formula IV. The process involves dissolving the triazolidine thione raw material in a suitable solvent like methanol or acetonitrile and introducing oxygen at ambient temperatures to drive the oxidation. Once the intermediate is formed, a reducing agent is added to complete the transformation into prothioconazole (Formula V). The detailed standardized synthesis steps below outline the precise conditions, stoichiometry, and workup procedures required to achieve the reported high yields and purity specifications.
- Oxidize the triazolidine thione compound (Formula I) with oxygen in a diluent solvent to form the disulfide intermediate (Formula II).
- Add a reducing agent such as sodium thiosulfate or sodium sulfite directly to the reaction mixture to reduce the intermediate to the final product (Formula III).
- Remove the solvent under reduced pressure and perform crystallization using water and toluene to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this one-pot synthesis method offers transformative benefits that directly impact the bottom line and operational resilience. By shifting away from hazardous reagents like n-butyllithium and heavy metal oxidants, companies can significantly reduce the costs associated with safety compliance, specialized storage, and waste disposal. The ability to use common, recyclable solvents such as methanol and acetonitrile further enhances cost efficiency, as these materials are readily available in the global market and do not suffer from the supply volatility seen with exotic reagents. Additionally, the simplified process flow reduces the number of unit operations, leading to shorter production cycles and faster time-to-market for critical crop protection products.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the reduction in solvent usage per batch contribute to a substantial decrease in overall production costs. Without the need for complex purification steps to remove heavy metals or residual sulfur, the downstream processing becomes far more economical, allowing for better margin management in competitive agrochemical markets.
- Enhanced Supply Chain Reliability: Utilizing oxygen as the primary oxidant removes dependency on specialized chemical oxidants that may face supply constraints. The mild reaction conditions allow for production in standard stainless steel reactors without the need for glass-lined or Hastelloy equipment, broadening the base of potential contract manufacturing organizations and ensuring consistent supply continuity.
- Scalability and Environmental Compliance: The drastic reduction in three wastes (wastewater, waste gas, and solid waste) simplifies environmental permitting and lowers the burden on effluent treatment plants. This eco-friendly profile facilitates easier scale-up from pilot to commercial production, as regulatory hurdles related to hazardous waste generation are minimized, supporting sustainable growth strategies.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this one-pot synthesis technology. These answers are derived directly from the experimental data and embodiments provided in the patent documentation, ensuring accuracy for technical decision-makers evaluating this route for adoption.
Q: How does this one-pot method compare to traditional ferric chloride oxidation?
A: Unlike the ferric chloride method which generates significant heavy metal waste and requires difficult separation, this oxygen-based one-pot method drastically reduces three wastes and simplifies purification, making it more environmentally compliant and cost-effective.
Q: What reducing agents are compatible with this synthesis route?
A: The process is versatile and supports various reducing agents including sodium thiosulfate, sodium sulfite, zinc powder, and sodium borohydride, allowing flexibility based on availability and cost considerations.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the reaction operates under mild conditions (0-50°C) using common solvents like methanol or acetonitrile, eliminating the need for specialized ultra-low temperature or anhydrous equipment required by older methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Prothioconazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of adopting advanced synthetic routes like the one described in CN111269190A to maintain a competitive edge in the agrochemical sector. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities.
We invite you to collaborate with us to leverage this cutting-edge technology for your supply chain needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to help you optimize your manufacturing strategy and secure a reliable source of high-quality triazolinethione compounds.
