Scalable Production of High-Purity Prothioconazole via Optimized Oxidation Catalysis
Scalable Production of High-Purity Prothioconazole via Optimized Oxidation Catalysis
The global demand for high-efficacy fungicides continues to drive innovation in agrochemical intermediate manufacturing, particularly for broad-spectrum agents like Prothioconazole. Patent CN111662240B introduces a groundbreaking preparation method that addresses the longstanding challenges of purity and yield in Prothioconazole synthesis. This technology leverages a sophisticated oxidative cyclization strategy, utilizing a tailored solvent system and mild reaction conditions to achieve product purity exceeding 99 percent and yields surpassing 93 percent. For R&D directors and procurement specialists, this represents a significant leap forward in process reliability, offering a robust pathway to produce stable, high-quality active ingredients suitable for demanding agricultural applications.
The core innovation lies in the precise control of the oxidation environment. Unlike conventional methods that struggle with byproduct formation and thermal instability, this approach employs a mixture of water and specific organic solvents—such as ethers, ketones, or esters—to modulate reaction selectivity. By maintaining temperatures between 0°C and 60°C, the process effectively mitigates the decomposition risks associated with acidic or high-heat environments. This technical advancement not only ensures the structural integrity of the final molecule but also streamlines the downstream purification process, making it an ideal candidate for large-scale industrial adoption by a reliable agrochemical intermediate supplier.
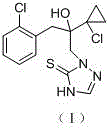
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Prothioconazole has been plagued by inefficiencies that compromise both economic viability and product quality. Prior art methods, such as those described in CN106986838A and CN109232452A, often rely on harsh oxidants like peroxybenzoic acid or complex solvent systems involving lower alcohols and dichloromethane. These traditional routes frequently necessitate the use of sodium sulfite to quench excess oxidants and chelating agents to remove residual metal ions, resulting in substantial salt-containing wastewater that poses significant environmental disposal challenges. Furthermore, the yields in these legacy processes typically hover around 85 to 90 percent, with purity levels struggling to exceed 98 percent, leading to stability issues in the final fungicide formulation.
Another critical drawback of conventional synthesis is the thermal sensitivity of the intermediate and final product. Many existing protocols require elevated temperatures or acidic workups that trigger decomposition, generating tarry byproducts that are difficult to separate. This not only reduces the overall mass balance but also complicates the crystallization step, often yielding products with undesirable coloration (e.g., yellowing due to iron entrainment) that fail to meet stringent international quality standards. For supply chain managers, these inefficiencies translate into higher production costs, longer lead times, and inconsistent batch-to-batch quality, undermining the reliability of the supply chain for high-purity agrochemical intermediates.
The Novel Approach
The methodology disclosed in CN111662240B fundamentally reengineers the oxidation step to overcome these historical bottlenecks. By selecting a biphasic solvent system comprising water and organic solvents like methyl tert-butyl ether or methyl isobutyl ketone, the invention creates an environment that maximizes the conversion of the triazolidine precursor while minimizing side reactions. The use of ferric chloride, either alone or in combination with air/oxygen, serves as a highly effective yet manageable oxidant. Crucially, the post-reaction workup is drastically simplified; the oxidant and inorganic byproducts can be removed simply by washing with water, eliminating the need for expensive chelating agents and complex extraction sequences.
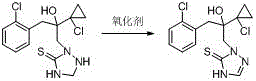
This novel approach also introduces a highly efficient recrystallization protocol using aromatic hydrocarbons such as xylene or toluene. This step is pivotal in elevating the purity from the crude range to over 99 percent, ensuring the removal of trace impurities that could affect the color and stability of the final API. The result is a white, crystalline product with superior physicochemical properties compared to commercially available alternatives. For manufacturers seeking cost reduction in fungicide manufacturing, this process offers a direct route to higher value output with significantly reduced waste generation and operational complexity.
Mechanistic Insights into FeCl3-Catalyzed Oxidative Cyclization
The chemical transformation at the heart of this patent involves the oxidative dehydrogenation of the 1,2,4-triazolidine-5-thione ring to form the aromatic 1,2,4-triazole-3-thione system found in Prothioconazole. Mechanistically, ferric chloride acts as a single-electron oxidant, facilitating the removal of hydrogen atoms from the saturated nitrogen-containing ring. The presence of water in the solvent mixture plays a dual role: it helps dissolve the inorganic ferric species, ensuring homogeneous contact with the substrate at the interface, and it suppresses the formation of acidic byproducts that could catalyze the degradation of the sensitive triazole ring. This delicate balance prevents the ring-opening reactions that typically lead to tar formation in anhydrous or strongly acidic conditions.
Furthermore, the choice of organic co-solvent is critical for controlling the reaction kinetics and thermodynamics. Solvents like ethers and ketones possess moderate polarity that enhances the solubility of the organic precursor while maintaining immiscibility with the aqueous phase containing the spent oxidant. This phase separation capability is key to the purification mechanism; once the oxidation is complete, the product remains in the organic layer while iron salts partition into the aqueous layer. This intrinsic separation efficiency eliminates the need for additional filtration or adsorption steps to remove metal contaminants, thereby preserving the yield and ensuring the high purity required for pharmaceutical and agrochemical grade materials.
Impurity control is achieved through the suppression of over-oxidation and thermal degradation. By strictly maintaining the reaction temperature between 5°C and 30°C during the oxidation phase, the process avoids the activation energy thresholds required for unwanted side reactions. The subsequent recrystallization step acts as a final polishing mechanism, leveraging the differential solubility of Prothioconazole and its potential isomers or degradation products in hot versus cold aromatic solvents. This multi-stage purification strategy ensures that the final impurity profile is minimal, meeting the rigorous specifications demanded by global regulatory bodies for crop protection agents.
How to Synthesize Prothioconazole Efficiently
The synthesis of high-purity Prothioconazole via this patented route involves a sequence of carefully controlled unit operations designed to maximize yield and minimize environmental impact. The process begins with the preparation of the reaction mixture, where the triazolidine precursor is suspended in a pre-mixed solvent system of water and an organic ether or ketone. Ferric chloride is then introduced as the oxidant, and the mixture is stirred under controlled temperature conditions to effect the cyclization. Following the reaction, the phases are allowed to separate, and the organic layer is washed to remove inorganic residues before solvent removal. The detailed standardized synthesis steps, including specific molar ratios, stirring rates, and crystallization parameters, are outlined in the guide below.
- React 2-(1-chloro-cyclopropane-1-yl)-1-(2-chlorophenyl)-2-hydroxy-3-(1,2,4-triazolidine-5-sulfur-1-yl) propane with an oxidant (ferric chloride/air) in a mixed solvent of water and organic ether/ketone at 0-60°C.
- Upon completion, separate layers, wash the organic phase with water to remove inorganic salts, and remove solvent under reduced pressure to isolate the crude product.
- Recrystallize the crude prothioconazole using an aromatic hydrocarbon solvent (e.g., xylene or toluene) to achieve purity levels exceeding 99%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis technology offers transformative benefits in terms of cost structure and operational reliability. The elimination of complex post-treatment reagents, such as chelating agents and reducing agents like sodium sulfite, directly reduces the bill of materials and simplifies inventory management. Moreover, the ability to recover and reuse solvents like methyl tert-butyl ether and xylene through standard distillation processes significantly lowers the variable costs associated with raw material consumption. This streamlined workflow translates into substantial cost savings in agrochemical intermediate manufacturing without compromising on the quality of the final output.
- Cost Reduction in Manufacturing: The process design inherently lowers production costs by removing the need for expensive purification additives and reducing the number of unit operations. Traditional methods often require multiple extraction and washing steps to remove metal impurities, which increases labor, energy, and solvent usage. In contrast, this method achieves high purity through a simple water wash and recrystallization, drastically cutting down on processing time and utility consumption. The high yield (>93 percent) further amplifies these savings by maximizing the output from each batch of starting material, ensuring better asset utilization and return on investment for production facilities.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply chain continuity by reducing the risk of batch failures due to impurity buildup or thermal runaway. The mild reaction conditions (0-60°C) allow for safer operation in standard glass-lined or stainless steel reactors, reducing maintenance downtime and safety incidents. Additionally, the use of commercially available and stable reagents like ferric chloride and common organic solvents ensures that raw material sourcing is not subject to the volatility associated with specialized or hazardous oxidants. This stability allows suppliers to offer more consistent lead times and reliable delivery schedules for high-purity agrochemical intermediates.
- Scalability and Environmental Compliance: From an environmental perspective, this process aligns with modern green chemistry principles by minimizing waste generation. The absence of heavy salt loads in the wastewater simplifies effluent treatment, reducing the burden on environmental compliance teams and lowering disposal fees. The scalability of the process is proven by its tolerance to variations in solvent ratios and its ability to be performed in batch or semi-continuous modes. This flexibility enables manufacturers to easily scale up from pilot plant quantities to multi-ton commercial production, supporting the growing global demand for sustainable and high-performance crop protection solutions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Prothioconazole using this advanced oxidation method. These insights are derived directly from the patent data and are intended to clarify the operational benefits and quality assurances associated with this technology. Understanding these details is crucial for partners looking to integrate this intermediate into their broader supply chains or formulation portfolios.
Q: Why is the purity of Prothioconazole critical for its stability?
A: Prothioconazole is chemically unstable in acidic and high-temperature environments. Lower purity grades degrade faster under these conditions, leading to tar formation and loss of efficacy. Achieving >99% purity ensures long-term storage stability and consistent fungicidal performance.
Q: How does this new oxidation method improve upon traditional synthesis routes?
A: Traditional methods often require complex post-treatment steps like chelating agents to remove metal impurities or multiple extractions. This patented process utilizes a specific water-organic solvent system that allows for simple water washing to remove oxidants, significantly reducing wastewater treatment complexity and operational costs.
Q: What are the advantages of the solvent system used in this process?
A: The use of ketone, ester, or ether solvents mixed with water enhances reaction selectivity and raw material conversion compared to traditional alcohol or aromatic solvents. This specific polarity balance minimizes side reactions and facilitates easier product isolation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Prothioconazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high-purity standards defined in patent CN111662240B are maintained at every scale. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced process control systems capable of meeting stringent purity specifications, guaranteeing that every batch of Prothioconazole delivers the stability and efficacy your formulations require.
We invite you to collaborate with us to leverage this cutting-edge synthesis technology for your agrochemical portfolio. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this optimized route can enhance your margin structure. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and samples that validate our commitment to quality and innovation in the agrochemical sector.
