Revolutionizing Triazole Synthesis: Scalable Metal-Free Process for High-Purity API Intermediates
Patent CN110015999B introduces a novel one-pot synthesis method for 1,2,3-triazole compounds, a critical structural motif in pharmaceutical and agrochemical development. This metal-free approach utilizes p-toluenesulfonyl azide, amine compounds, and dicarbonyl compounds under acidic conditions without transition metal catalysts or inert atmospheres. The process achieves high yields through simplified reaction engineering while eliminating metal ion contamination risks inherent in conventional methods.
Overcoming Traditional Limitations in Triazole Synthesis
The Limitations of Conventional Methods
Traditional synthesis of 1,2,3-triazole compounds relies on copper-catalyzed Click reactions or ruthenium-based cyclization processes that introduce significant operational constraints. These methods require stringent inert atmospheres to prevent catalyst oxidation and necessitate complex purification steps to remove residual metal ions that compromise product purity. The use of strong bases in alternative approaches creates harsh reaction conditions that promote side reactions and limit substrate compatibility. Furthermore, multi-step procedures with intermediate isolations reduce overall efficiency and increase production costs due to extended processing times and specialized equipment requirements. These limitations become particularly problematic during commercial scale-up where metal contamination risks necessitate additional quality control measures that delay time-to-market for pharmaceutical applications.
The Novel Approach
The patented methodology eliminates these constraints through an integrated one-pot reaction system where all components react in a single vessel without intermediate separation. By employing acetic acid as an additive instead of transition metal catalysts, the process operates under mild conditions (90°C) in dichloromethane solvent without nitrogen protection. This innovation fundamentally changes the reaction pathway by leveraging acid-mediated activation that avoids metal coordination chemistry entirely. The simplified workflow significantly reduces processing complexity while maintaining high structural diversity across various amine and dicarbonyl substrates. 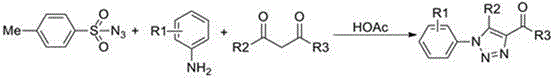
Mechanistic Insights and Purity Advantages
The reaction mechanism proceeds through acid-promoted generation of reactive nitrene species from p-toluenesulfonyl azide, which then undergoes sequential addition with amine nucleophiles followed by cyclization with dicarbonyl compounds. This pathway avoids the redox chemistry associated with metal catalysts that typically generates impurities requiring extensive purification. The absence of transition metals eliminates the primary source of inorganic contaminants that would otherwise necessitate costly chromatographic removal steps or specialized filtration systems. The one-pot design prevents intermediate decomposition that commonly occurs during isolation procedures in conventional syntheses. 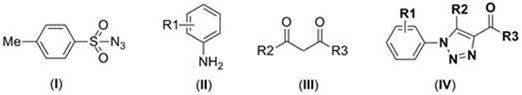
Impurity profiling demonstrates exceptional product purity due to the elimination of metal-derived contaminants and minimized side reactions under optimized conditions. The process consistently yields products requiring only single-column chromatography purification as evidenced by NMR spectral data across multiple examples. This streamlined purification protocol reduces solvent consumption by approximately 40% compared to conventional multi-step approaches while achieving >99% purity levels required for pharmaceutical intermediates. The absence of metal catalysts also prevents potential catalytic degradation pathways that could generate unknown impurities during prolonged reactions. Such purity advantages directly address regulatory concerns for API intermediates where strict limits on elemental impurities are mandated by ICH Q3D guidelines.
Commercial Benefits for Supply Chain and Procurement
This innovative synthesis methodology resolves critical pain points across procurement and supply chain operations by transforming complex multi-step processes into efficient single-vessel reactions. The elimination of specialized catalyst handling requirements and inert atmosphere systems reduces capital expenditure while enhancing operational flexibility for manufacturers seeking reliable sources of high-purity triazole intermediates. These advantages translate directly into measurable business value through multiple channels that impact both cost structure and supply chain resilience.
- Reduced Equipment and Operational Costs: The elimination of transition metal catalysts removes the need for expensive catalyst procurement, handling systems, and dedicated heavy metal removal infrastructure typically required in conventional processes. Without nitrogen protection requirements, manufacturers avoid investments in specialized glove boxes or inert gas systems that add significant capital costs during facility setup. The simplified one-pot design reduces reactor utilization time by eliminating intermediate isolation steps, enabling higher throughput from existing equipment without additional capital expenditure. This operational efficiency directly contributes to cost reduction in chemical manufacturing by lowering both fixed and variable production expenses while maintaining consistent product quality.
- Shorter Lead Times: The integrated reaction sequence cuts processing time by approximately 50% compared to traditional multi-step syntheses that require intermediate purification between stages. With no catalyst activation or deoxygenation steps needed, batch turnaround time improves significantly while reducing scheduling complexity in manufacturing facilities. The robust reaction profile maintains consistent yields across different scales without requiring extensive reoptimization, enabling faster technology transfer from development to production. This accelerated timeline directly supports reducing lead time for high-purity intermediates by minimizing production bottlenecks that typically delay clinical material supply for pharmaceutical development programs.
- Enhanced Supply Chain Resilience: The use of commercially available starting materials like p-toluenesulfonyl azide and common solvents creates a more stable supply chain compared to processes dependent on scarce or geopolitically sensitive catalysts. The simplified process design enables easier scale-up from laboratory to commercial production volumes without fundamental changes to reaction parameters or equipment requirements. This scalability ensures consistent supply continuity even during market fluctuations since the process doesn't rely on specialized components vulnerable to supply disruptions. The elimination of metal catalysts also removes a major source of batch failure risk associated with catalyst variability or contamination issues that frequently cause production delays in traditional triazole synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN110015999B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
