Advanced Manufacturing of 5-Substituted Thiophenyl-Benzimidazole Intermediates for Veterinary Applications
Advanced Manufacturing of 5-Substituted Thiophenyl-Benzimidazole Intermediates for Veterinary Applications
The global demand for high-efficacy veterinary anthelmintics continues to drive innovation in the synthesis of benzimidazole derivatives, particularly those serving as precursors to drugs like fenbendazole. Patent CN102304090A introduces a robust and economically viable methodology for preparing 5-substituted thiophenyl-benzimidazol-2-N-methoxycarbonyl compounds, addressing critical bottlenecks in traditional manufacturing. This technical disclosure outlines a streamlined four-step sequence that replaces hazardous reagents with safer alternatives while maintaining high purity standards essential for regulatory compliance. By leveraging a novel combination of nucleophilic substitution and chemoselective reduction, the process achieves significant improvements in overall yield and environmental profile. For R&D directors and procurement specialists, this route represents a strategic opportunity to optimize the supply chain for broad-spectrum antiparasitic agents. The structural versatility of the method allows for various substituents on the phenyl ring, enhancing its utility across a range of related pharmaceutical targets.
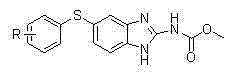
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzimidazole carbamates has relied on methodologies that pose significant safety and economic challenges for large-scale operations. One prevalent prior art method utilizes sodium hydride (NaH) as a base for the initial coupling reaction, which introduces severe safety hazards due to the pyrophoric nature of the reagent and the requirement for strictly anhydrous conditions. Furthermore, alternative routes often depend on expensive and unstable starting materials, such as N-(methoxycarbonyl) amino azomethine acid esters or S-methyl-isourea, which drive up the cost of goods sold and complicate inventory management. These traditional pathways also frequently employ catalytic hydrogenation for nitro reduction, necessitating high-pressure reactors and introducing the risk of residual heavy metal contamination that requires costly purification steps. The cumulative effect of these factors is a manufacturing process that is difficult to scale safely and economically, limiting the ability of suppliers to meet fluctuating market demands efficiently.
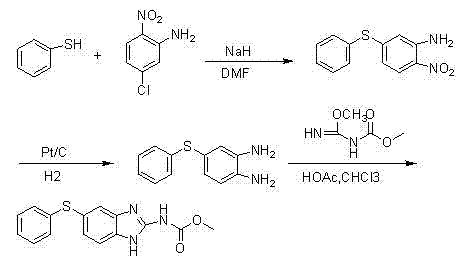
The Novel Approach
The methodology disclosed in the patent fundamentally reengineers the synthetic pathway to prioritize safety, cost-efficiency, and operational simplicity without compromising yield. Instead of hazardous sodium hydride, the new process employs anhydrous potassium carbonate in DMF for the nucleophilic substitution, creating a much safer reaction environment that is easier to control on a multi-ton scale. A key innovation lies in the reduction step, where a sodium borohydride and ammonium chloride system is used to selectively reduce the nitro group to an amine, avoiding the need for expensive noble metal catalysts. This approach not only lowers raw material costs but also simplifies the downstream processing by eliminating the need for complex catalyst removal protocols. The final cyclization utilizes an in-situ generated cyanamide derivative, ensuring high reactivity and minimizing the formation of side products. This holistic redesign results in a shorter synthesis route with higher overall recovery rates, making it an ideal candidate for modern green chemistry initiatives in the fine chemical sector.
Mechanistic Insights into Chemoselective Reduction and Cyclization
The core chemical innovation of this process centers on the chemoselective reduction of the nitro-thioether intermediate using the NaBH4-NH4Cl system in methanol. Mechanistically, the ammonium chloride serves to modulate the reducing power of sodium borohydride, generating reactive species that effectively target the nitro group while leaving the sensitive thioether linkage intact. This selectivity is crucial because harsher reducing conditions could potentially cleave the carbon-sulfur bond or reduce other functional groups, leading to complex impurity profiles that are difficult to separate. The reaction proceeds through the formation of hydroxylamine intermediates which are rapidly reduced to the corresponding aniline, ensuring a clean conversion to the diamine precursor required for cyclization. By avoiding transition metal catalysts, the process inherently reduces the risk of metal leaching, a critical quality attribute for veterinary active pharmaceutical ingredients (APIs) intended for food-producing animals. This mechanistic precision ensures that the resulting diamine is of sufficient purity to drive the subsequent cyclization reaction to completion with minimal byproduct formation.
Following the reduction, the cyclization mechanism involves the condensation of the diamine with a cyanamide derivative generated from thiourea and methyl chloroformate. The cyanamide acts as a potent electrophile, attacking the nucleophilic amine groups on the benzene ring to close the imidazole ring system. The use of methyl chloroformate activates the thiourea, creating a highly reactive intermediate that facilitates rapid ring closure under reflux conditions in ethanol. This step is thermodynamically driven and benefits from the precipitation of the final product upon cooling, which simplifies isolation and enhances purity. The reaction conditions are optimized to prevent the hydrolysis of the methoxycarbonyl group, preserving the integrity of the final carbamate functionality which is essential for the biological activity of the end drug. Understanding these mechanistic nuances allows process chemists to fine-tune reaction parameters such as temperature and stoichiometry to maximize yield and minimize waste generation.
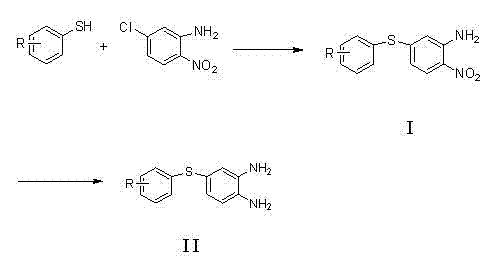
How to Synthesize 5-Substituted Thiophenyl-Benzimidazole Efficiently
The synthesis protocol described in the patent provides a clear, step-by-step framework for producing high-purity benzimidazole intermediates suitable for veterinary applications. The process begins with the coupling of substituted thiophenols with chloronitroanilines, followed by a mild reduction and a final condensation step. Each stage is designed to be operationally simple, utilizing standard laboratory and plant equipment such as reflux condensers and filtration units. The detailed standardized synthesis steps below outline the specific molar ratios and conditions required to replicate the high yields reported in the patent examples. Adhering to these parameters ensures consistent quality and reproducibility, which are paramount for GMP-compliant manufacturing environments.
- Perform nucleophilic substitution between substituted thiophenol and 2-nitro-5-chloroaniline using potassium carbonate in DMF to form the nitro-thioether intermediate.
- Execute chemoselective reduction of the nitro group using sodium borohydride and ammonium chloride in methanol to generate the diamine precursor.
- React the diamine with an in-situ generated cyanamide derivative (from thiourea and methyl chloroformate) in ethanol under reflux to cyclize into the final benzimidazole product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthesis route offers substantial advantages that directly impact the bottom line and supply chain resilience for manufacturers of veterinary intermediates. The elimination of hazardous reagents like sodium hydride reduces the need for specialized safety infrastructure and insurance costs, while the use of commodity chemicals like potassium carbonate and sodium borohydride stabilizes raw material pricing against market volatility. The simplified workup procedures, which rely on basic extraction and crystallization rather than complex chromatography or distillation, significantly reduce processing time and solvent consumption. These efficiencies translate into a more agile manufacturing process capable of responding quickly to market surges in demand for anthelmintic drugs. Furthermore, the high purity of the crude product minimizes the need for extensive recrystallization, further driving down production costs and energy usage.
- Cost Reduction in Manufacturing: The replacement of expensive and unstable reagents with cheap, commercially available alternatives creates a direct reduction in the bill of materials. By avoiding the use of noble metal catalysts and specialized esters, the process removes significant cost drivers associated with both procurement and waste disposal. The high yield reported in the patent examples indicates efficient atom economy, meaning less raw material is wasted per unit of product produced. Additionally, the milder reaction conditions reduce energy consumption for heating and cooling, contributing to lower utility costs over the lifecycle of the production campaign. These cumulative savings allow for a more competitive pricing strategy in the global market for veterinary drug intermediates.
- Enhanced Supply Chain Reliability: The reliance on bulk commodity chemicals ensures that the supply chain is not vulnerable to the shortages often associated with specialized fine chemical reagents. Thiophenols and chloronitroanilines are produced at scale by multiple suppliers globally, providing redundancy and security of supply. The robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality, reducing the risk of batch failures. This stability is critical for maintaining continuous production schedules and meeting strict delivery deadlines for downstream API manufacturers. Consequently, partners adopting this technology can offer greater reliability to their customers, strengthening long-term contractual relationships.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from kilogram-scale development to multi-ton commercial production without requiring fundamental changes to the chemistry. The avoidance of heavy metals and hazardous bases simplifies wastewater treatment and waste disposal, aligning with increasingly stringent environmental regulations. The use of common solvents like DMF, methanol, and ethanol facilitates solvent recovery and recycling programs, further reducing the environmental footprint. This alignment with green chemistry principles not only mitigates regulatory risk but also enhances the corporate sustainability profile of the manufacturer. Such environmental stewardship is becoming a key differentiator in supplier selection processes for major pharmaceutical and agrochemical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation. They serve to clarify the operational benefits and chemical rationale behind the new methodology for stakeholders evaluating its adoption.
Q: Why is the NaBH4/NH4Cl reduction method preferred over catalytic hydrogenation for this intermediate?
A: The sodium borohydride/ammonium chloride system offers superior chemoselectivity for reducing the nitro group without requiring high-pressure equipment or expensive noble metal catalysts like Pd/C, thereby eliminating risks of heavy metal contamination in the final veterinary active ingredient.
Q: What are the safety advantages of this synthesis route compared to traditional methods?
A: This route eliminates the use of sodium hydride (NaH), a pyrophoric and hazardous reagent used in conventional methods, significantly improving operational safety and reducing the need for specialized inert atmosphere handling during the initial substitution step.
Q: Can this process be scaled for industrial production of fenbendazole precursors?
A: Yes, the process utilizes cheap, readily available raw materials like thiophenol and chloronitroaniline, and involves standard unit operations such as reflux, filtration, and extraction, making it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Substituted Thiophenyl-Benzimidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and safe synthesis routes in the production of high-value veterinary intermediates. Our team of expert process chemists has extensively analyzed the technology disclosed in CN102304090A and is fully prepared to implement this advanced methodology for our global clients. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab to plant is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications to guarantee that every batch meets the highest international standards. By leveraging this optimized route, we can deliver superior quality intermediates with improved lead times and cost structures.
We invite procurement leaders and R&D directors to collaborate with us to evaluate the feasibility of this route for your specific supply chain needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your product portfolio. Let us be your partner in driving innovation and efficiency in the veterinary pharmaceutical sector.
