Advanced Manufacturing of Roflumilast Intermediates: A Technical Breakthrough for Global Supply Chains
Advanced Manufacturing of Roflumilast Intermediates: A Technical Breakthrough for Global Supply Chains
The pharmaceutical industry constantly seeks robust, scalable, and cost-effective pathways for producing critical active pharmaceutical ingredients (APIs) and their precursors. A pivotal development in this domain is detailed in Chinese Patent CN102775345A, which discloses a superior preparation method for Roflumilast and its key intermediates. Roflumilast, a selective phosphodiesterase 4 (PDE4) inhibitor approved for the treatment of chronic obstructive pulmonary disease (COPD), requires precise synthetic control to ensure high purity and safety. The traditional synthesis routes often suffer from harsh reaction conditions and challenging purification steps, which can bottleneck supply chains and inflate costs. This new technology addresses these pain points by introducing a modular approach that separates the amidation step from the etherification and difluoromethylation steps, allowing for better control over regioselectivity and impurity profiles.
For R&D directors and process chemists, the significance of this patent lies in its ability to bypass the formation of persistent byproducts that plague earlier methods. By utilizing protected resorcylic acid derivatives or direct amidation of dihydroxy benzoic acids followed by sequential functionalization, manufacturers can achieve higher overall yields. The method is versatile, supporting the production of the free base, N-oxides, and various acid addition salts, making it a comprehensive solution for diverse formulation needs. As a reliable pharmaceutical intermediates supplier, understanding these mechanistic nuances is crucial for delivering consistent quality to downstream drug manufacturers who demand rigorous adherence to specifications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods, such as those disclosed in International Patent Application WO 95/01338, typically rely on the activation of 3-(cyclopropylmethoxy)-4-(difluoromethoxy)benzoic acid into an acyl chloride using thionyl chloride, followed by coupling with 4-amino-3,5-dichloropyridine. This coupling step necessitates the use of strong bases like sodium hydride (NaH) in tetrahydrofuran (THF) to deprotonate the weakly nucleophilic aminopyridine. These conditions are inherently violent and pose significant safety risks on a large scale. More critically, the high basicity promotes side reactions, leading to the formation of 3-(cyclopropylmethoxy)-N-(3,5-dichloropyridine-4-yl)-4-hydroxybenzamide as a stubborn byproduct. This impurity is structurally similar to the target molecule, making it extremely difficult to remove even after repeated recrystallization, thereby depressing the total recovery rate and indirectly driving up production costs due to yield loss and extensive purification requirements.
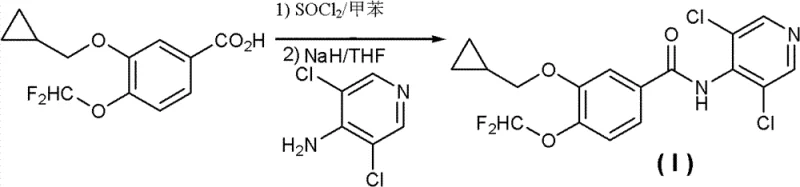
The Novel Approach
The innovative strategy presented in CN102775345A fundamentally reorders the synthetic sequence to mitigate these risks. Instead of forming the amide bond last under harsh conditions, the new method constructs the amide backbone early using 3,4-dihydroxybenzoic acid derivatives (Formula II) and 4-amino-3,5-dichloropyridine (Formula IX). This amidation can be performed on protected substrates (Formula VIII) or directly, followed by deprotection to yield the dihydroxy intermediate. Subsequently, the phenolic hydroxyl groups are selectively functionalized. One pathway involves alkylating the 3-position with a cyclopropylmethyl leaving group (Formula III) to form intermediate (IV), followed by difluoromethylation at the 4-position using reagents like difluorochloromethane. Alternatively, the difluoromethoxy group can be installed first to form intermediate (VI), followed by alkylation. This decoupling of steps allows the use of much milder bases, such as potassium carbonate or cesium carbonate, in solvents like acetone or toluene, drastically improving the safety profile and product purity.
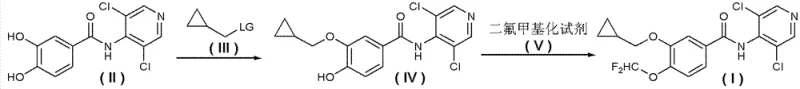
Mechanistic Insights into Sequential Functionalization Strategy
The core mechanistic advantage of this process lies in the enhanced nucleophilicity management of the phenolic hydroxyl groups on the benzamide scaffold. In the conventional route, the electron-withdrawing nature of the amide carbonyl and the pyridine ring can complicate late-stage substitutions. By establishing the amide bond first on a simpler dihydroxy scaffold, the electronic environment is more predictable. The alkylation step, converting the phenol to a cyclopropylmethyl ether, proceeds via a classic SN2 mechanism. The patent specifies the use of phase-transfer catalysts like tetrabutylammonium bromide (TBAB) to facilitate this reaction in biphasic systems (e.g., toluene/water or acetone/solid base). This ensures efficient mass transfer and complete conversion without requiring cryogenic temperatures or pyrophoric reagents. The subsequent difluoromethylation introduces the critical -OCF2H moiety, a bioisostere often used to enhance metabolic stability and lipophilicity in drug molecules.
Impurity control is intrinsically built into this mechanism. By avoiding the highly basic NaH conditions during the final coupling, the hydrolysis of the amide bond or the cleavage of the newly formed ether bonds is minimized. Furthermore, the use of protecting groups (PG) such as acetyl, benzoyl, or silyl groups in the alternative Route 4 (Reaction Formula 4) provides an additional layer of control. For instance, protecting the 3,4-dihydroxybenzoic acid as a diacetate prevents unwanted side reactions during the initial amidation. The deprotection step is then carried out under mild acidic or basic hydrolysis conditions before the final functionalization. This modular "protect-react-deprotect-functionalize" logic ensures that the final API intermediate meets stringent purity specifications required by regulatory bodies, reducing the burden on quality control laboratories.
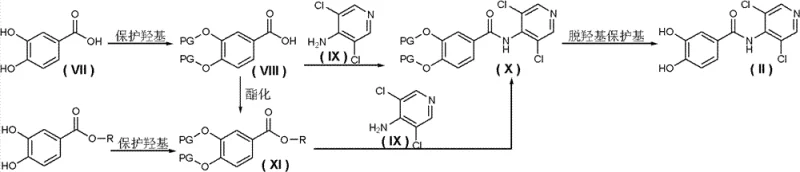
How to Synthesize 3-(cyclopropylmethoxy)-N-(3,5-dichloropyridine-4-yl)-4-(difluoromethoxy)benzamide Efficiently
The synthesis of this critical respiratory drug intermediate requires precise control over reaction parameters to maximize yield and minimize waste. The patented process outlines several viable pathways, with the most industrially relevant involving the reaction of a dihydroxy benzamide precursor with specific alkylating agents and difluoromethylating reagents. Operators must carefully select the base and solvent system; for example, using potassium carbonate in acetone at temperatures between 40°C and 80°C provides an optimal balance between reaction rate and selectivity. The detailed standardized synthesis steps, including specific molar ratios, workup procedures, and purification techniques, are outlined below to guide process engineers in implementing this technology.
- Prepare the key dihydroxy benzamide intermediate (Formula II) by reacting protected 3,4-dihydroxybenzoic acid derivatives with 4-amino-3,5-dichloropyridine, followed by deprotection.
- Perform selective alkylation of the phenolic hydroxyl group using cyclopropylmethyl halides in the presence of mild bases like potassium carbonate in acetone or toluene.
- Introduce the difluoromethoxy group using difluoromethylating reagents (e.g., difluorochloromethane) under phase-transfer catalysis conditions to yield the final high-purity intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route translates into tangible operational efficiencies and risk mitigation. Traditional methods relying on acyl chlorides and sodium hydride introduce significant logistical hazards, requiring specialized handling equipment and strict safety protocols that can delay production schedules. By shifting to a route that utilizes stable carboxylic acid derivatives and mild inorganic bases, the manufacturing process becomes inherently safer and more manageable. This reduction in hazard potential lowers insurance costs and simplifies facility requirements, allowing for more flexible production planning. Furthermore, the improved impurity profile means fewer batches are rejected due to out-of-specification results, ensuring a more consistent flow of materials to the final drug product assembly lines.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents like sodium hydride, coupled with the avoidance of complex purification steps to remove hydroxybenzamide byproducts, leads to substantial cost savings. Higher overall yields mean that less raw material is required to produce the same amount of finished intermediate, directly improving the cost of goods sold (COGS). Additionally, the use of common industrial solvents such as acetone, toluene, and ethyl acetate reduces solvent procurement costs and simplifies recycling processes, contributing to a leaner manufacturing budget without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials like 3,4-dihydroxybenzoic acid and 4-amino-3,5-dichloropyridine ensures a stable supply base. Unlike specialized acyl chlorides which may have limited suppliers and short shelf-lives, the precursors used in this new method are commodity chemicals with robust global supply chains. This diversification of raw material sources reduces the risk of supply disruptions caused by vendor-specific issues. Moreover, the milder reaction conditions reduce equipment wear and tear, minimizing unplanned maintenance downtime and ensuring that delivery commitments to pharmaceutical partners are met consistently.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to multi-ton commercial production often exposes hidden inefficiencies, but this route is designed with scalability in mind. The exothermic risks associated with NaH additions are removed, making the scale-up trajectory smoother and safer. From an environmental perspective, the reduction in byproduct formation decreases the load on wastewater treatment facilities. The ability to use phase-transfer catalysis also allows for reactions in greener solvent systems or reduced solvent volumes, aligning with modern green chemistry principles and helping manufacturers meet increasingly stringent environmental regulations regarding waste discharge and solvent emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Roflumilast intermediates using this patented methodology. These insights are derived directly from the experimental data and claims within CN102775345A, providing clarity on how this technology resolves historical manufacturing bottlenecks. Understanding these details helps stakeholders make informed decisions about sourcing and process validation.
Q: How does the new synthesis route improve impurity profiles compared to conventional methods?
A: Conventional methods using acyl chlorides and sodium hydride often generate difficult-to-remove hydroxybenzamide byproducts. The new route utilizes milder bases and sequential functionalization, significantly reducing side reactions and simplifying purification.
Q: What are the scalability advantages of this patented process?
A: The process avoids hazardous reagents like sodium hydride in the final coupling steps and uses common industrial solvents such as acetone and toluene. This enhances operational safety and allows for easier scale-up from kilogram to multi-ton production.
Q: Can this method be adapted for N-oxide or salt forms of Roflumilast?
A: Yes, the patent explicitly covers the preparation of the base compound (Formula I) as well as its N-oxide derivatives and pharmaceutically acceptable acid addition salts, providing flexibility for different drug formulation requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Roflumilast Intermediate Supplier
The technological advancements detailed in this report underscore the complexity and precision required to manufacture high-quality respiratory disease treatments. At NINGBO INNO PHARMCHEM, we possess the technical expertise to translate these patented laboratory methods into robust, commercial-scale operations. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch against the highest industry standards, guaranteeing that our intermediates seamlessly integrate into your downstream API synthesis.
We invite you to collaborate with us to optimize your supply chain for Roflumilast and related pharmaceutical projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us today to request specific COA data, route feasibility assessments, and competitive quotations. Let us be your partner in delivering life-saving medications to patients worldwide through superior chemical manufacturing excellence.
