Optimizing Roflumilast Production: A Technical Breakthrough for Commercial Scale-Up
The pharmaceutical landscape for Chronic Obstructive Pulmonary Disease (COPD) treatments continues to evolve, with Roflumilast standing out as a critical therapeutic agent. Recent advancements in synthetic methodology, specifically detailed in patent CN102336704A, have introduced a transformative approach to manufacturing this complex molecule. This technical insight report analyzes the novel synthetic pathway that bypasses traditional bottlenecks, offering a robust solution for reliable roflumilast intermediate supplier networks globally. The core innovation lies in the strategic utilization of Isovanillin as a foundational building block, replacing scarce and expensive precursors mandated by earlier intellectual property such as US Pat 5712298. By re-engineering the synthesis of the key intermediate 3-cyclopropylmethoxy-4-hydroxybenzaldehyde, this method addresses fundamental challenges in availability and cost. 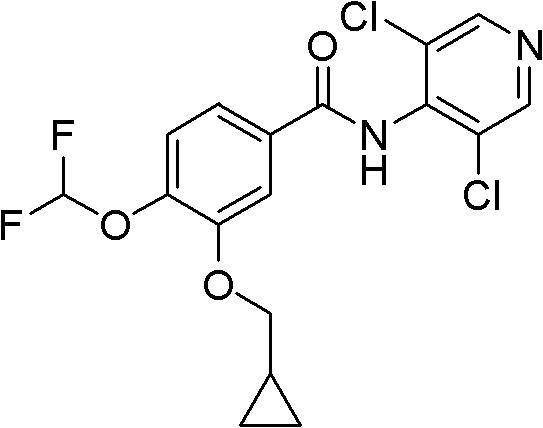 . The structural integrity of the final API is maintained while drastically optimizing the upstream process efficiency. For R&D Directors and Supply Chain Heads, understanding this shift is paramount, as it represents a move towards more sustainable and scalable high-purity roflumilast production capabilities.
. The structural integrity of the final API is maintained while drastically optimizing the upstream process efficiency. For R&D Directors and Supply Chain Heads, understanding this shift is paramount, as it represents a move towards more sustainable and scalable high-purity roflumilast production capabilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical synthesis routes for Roflumilast have been plagued by significant logistical and economic hurdles that hinder efficient cost reduction in API manufacturing. The method described in US Pat 5712298, while chemically valid, relies on a starting material, formula (3), which is notoriously difficult to source commercially, creating a fragile supply chain dependency. Furthermore, alternative pathways reported in World Patent WO2004033430 introduce severe safety and environmental liabilities by utilizing toxic carbon monoxide gas and expensive palladium catalysts for carbonylation steps. These conventional methods often suffer from poor selectivity during the initial alkylation phases, leading to the formation of difficult-to-remove by-products such as di-cyclopropylated species. The necessity for harsh reaction conditions, including cryogenic temperatures around -55°C for bromination steps in older literature, further exacerbates energy consumption and operational complexity. Consequently, these factors combine to create a high barrier to entry for commercial scale-up of complex pharmaceutical intermediates, resulting in inflated production costs and extended lead times that are untenable in a competitive generic market.
The Novel Approach
The methodology outlined in CN102336704A presents a paradigm shift by establishing a concise, high-yielding route starting from the abundant and inexpensive Isovanillin. This novel approach eliminates the dependency on non-commercial starting materials and avoids the use of hazardous reagents like carbon monoxide, thereby enhancing overall process safety. The synthesis is streamlined into a logical sequence where Isovanillin undergoes cyclopropyl methylation followed by a highly selective demethylation to yield the critical intermediate (3). 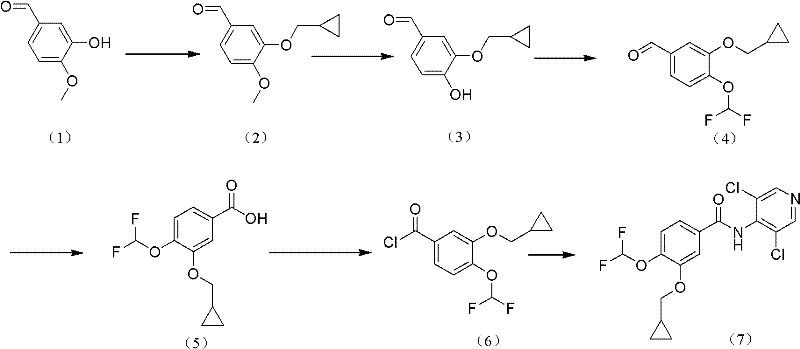 . This pathway demonstrates superior reaction selectivity, minimizing the generation of regio-isomers that typically complicate purification. The post-treatment procedures are notably simplified, avoiding complex chromatographic separations in favor of efficient crystallization techniques. For procurement teams, this translates to a more resilient supply chain with reducing lead time for high-purity pharmaceutical intermediates, as the raw material base is stable and widely available. The elimination of transition metal catalysts also removes the need for costly and time-consuming heavy metal scavenging steps, directly contributing to substantial operational expenditure savings.
. This pathway demonstrates superior reaction selectivity, minimizing the generation of regio-isomers that typically complicate purification. The post-treatment procedures are notably simplified, avoiding complex chromatographic separations in favor of efficient crystallization techniques. For procurement teams, this translates to a more resilient supply chain with reducing lead time for high-purity pharmaceutical intermediates, as the raw material base is stable and widely available. The elimination of transition metal catalysts also removes the need for costly and time-consuming heavy metal scavenging steps, directly contributing to substantial operational expenditure savings.
Mechanistic Insights into Isovanillin-Based Cyclization and Demethylation
The chemical elegance of this process is rooted in the precise control of reaction parameters during the cyclopropyl methylation and subsequent demethylation stages. In the first step, Isovanillin reacts with chloromethyl cyclopropane or bromomethyl cyclopropane in the presence of a base such as anhydrous potassium carbonate and a phase-transfer catalyst like benzyltriethylammonium chloride. The reaction is conducted in polar aprotic solvents such as DMF or NMP at moderate temperatures ranging from 20°C to 90°C. This specific thermal window is crucial; it provides sufficient energy to drive the nucleophilic substitution without promoting excessive side reactions or degradation of the sensitive aldehyde moiety. The phase-transfer catalyst plays a pivotal role in facilitating the interaction between the organic halide and the phenolic salt in the organic phase, ensuring high conversion rates and minimizing the formation of O-alkylated by-products. The molar ratios are tightly controlled, typically maintaining a slight excess of the alkylating agent (1:1.5 to 1:1.6) to ensure complete consumption of the valuable Isovanillin starting material.
Following the methylation, the selective demethylation of the 4-methoxy group is achieved using dodecyl mercaptan or similar thiol reagents in the presence of strong bases like sodium methylate or sodium hydroxide. This step is performed at elevated temperatures, preferably around 100°C, in solvents such as NMP or DMSO. The mechanism involves the nucleophilic attack of the thiolate anion on the methyl group of the methoxy functionality, cleaving the ether bond to reveal the phenolic hydroxyl group while leaving the cyclopropylmethoxy group intact due to its steric and electronic stability. This selectivity is the cornerstone of the process, as it prevents the formation of the fully demethylated catechol derivative, which would be a difficult impurity to separate. The subsequent workup involves careful pH adjustment to approximately 4.0 using hydrochloric acid, which precipitates the product or facilitates its extraction into organic solvents like ethyl acetate. This meticulous control over acidity ensures that the aldehyde functionality remains stable and that the product is isolated with minimal contamination from inorganic salts or residual thiols.
How to Synthesize Roflumilast Efficiently
Implementing this synthesis route requires strict adherence to the optimized reaction conditions and purification protocols defined in the patent to ensure consistent quality and yield. The process begins with the preparation of 3-cyclopropylmethoxy-4-methoxybenzaldehyde, followed by the critical demethylation to generate intermediate (3), which is then converted to the difluoromethoxy derivative and coupled with the dichloropyridine amine. Each step demands precise monitoring of temperature, stoichiometry, and reaction time to maintain the integrity of the intermediates. The final purification stage is particularly vital, utilizing a dual-solvent recrystallization system to achieve pharmaceutical-grade purity.
- Perform cyclopropyl methylation on Isovanillin using chloromethyl cyclopropane or bromomethyl cyclopropane with a phase-transfer catalyst in DMF or NMP at 20-90°C.
- Execute selective demethylation of the resulting 3-cyclopropylmethoxy-4-methoxybenzaldehyde using dodecyl mercaptan and sodium methylate at 100°C.
- Purify the final Roflumilast crude product via a dual-stage recrystallization process involving isopropanol/water followed by ethyl acetate/petroleum ether.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers profound advantages that extend beyond mere chemical efficiency, directly impacting the bottom line and supply chain resilience. The primary driver for cost optimization is the substitution of obscure, high-cost starting materials with Isovanillin, a commodity chemical with a stable global supply. This shift fundamentally alters the cost structure of the API, removing the premium associated with custom-synthesized precursors. Furthermore, the avoidance of noble metal catalysts such as palladium eliminates a significant variable cost component and the associated regulatory burden of residual metal testing. The simplified post-treatment workflow reduces solvent consumption and waste generation, aligning with modern green chemistry principles and reducing disposal costs. For supply chain managers, the robustness of this route means fewer points of failure; the reagents are standard industrial chemicals, mitigating the risk of shortages that often plague specialized reagent markets.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of readily available raw materials like Isovanillin drastically lower the direct material costs associated with production. By removing the need for complex heavy metal removal processes, manufacturers can also reduce the operational overhead related to specialized filtration media and analytical testing. The high selectivity of the reaction minimizes the loss of valuable intermediates to by-product formation, thereby improving the overall mass balance and yield efficiency. These factors collectively contribute to a leaner manufacturing model that supports aggressive pricing strategies without compromising margin integrity.
- Enhanced Supply Chain Reliability: Sourcing stability is significantly improved as the key starting materials are produced by multiple suppliers globally, reducing dependency on single-source vendors. The synthetic route avoids the use of highly regulated toxic gases like carbon monoxide, simplifying the safety compliance requirements and facility licensing needed for production. This ease of compliance accelerates the timeline for technology transfer and site validation, ensuring faster time-to-market for new generic filings. Additionally, the robust nature of the intermediates allows for more flexible inventory management, as they possess better stability profiles compared to those generated via harsher conventional methods.
- Scalability and Environmental Compliance: The process is inherently designed for scale, utilizing standard unit operations such as filtration, extraction, and crystallization that are easily replicated in multi-purpose reactors. The absence of cryogenic steps and toxic gases reduces the energy footprint and safety risks, making it suitable for production in a wider range of geographic locations. Waste streams are more manageable, consisting primarily of organic solvents and inorganic salts that can be treated using standard effluent processing protocols. This environmental compatibility ensures long-term sustainability and reduces the risk of regulatory shutdowns due to compliance violations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on feasibility and performance. Understanding these details is crucial for stakeholders evaluating the potential for technology adoption or partnership.
Q: Why is the Isovanillin route superior to US Pat 5712298?
A: The conventional route relies on starting materials that are not commercially available, whereas the Isovanillin route utilizes readily accessible and cost-effective raw materials, significantly simplifying the supply chain.
Q: How does this method improve impurity profiles?
A: By employing selective demethylation and a specialized dual-solvent recrystallization technique, the process effectively removes by-products, achieving purity levels exceeding 99.5%.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method avoids toxic carbon monoxide and expensive palladium catalysts found in alternative routes, making it safer and more economically viable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Roflumilast Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of deploying advanced synthetic methodologies to meet the rigorous demands of the global pharmaceutical market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN102336704A are fully realized in practical manufacturing environments. We are committed to delivering stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of Roflumilast meets the highest international standards for safety and efficacy. Our infrastructure is designed to handle complex chemistries safely, providing a secure partner for your long-term supply needs.
We invite you to collaborate with us to optimize your supply chain and achieve significant competitive advantages. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities align with your project goals. Let us help you navigate the complexities of API sourcing with confidence and precision.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
