Advanced Lewis Acid Catalysis for High-Purity Carboxylic Acid Derivatives Manufacturing
Advanced Lewis Acid Catalysis for High-Purity Carboxylic Acid Derivatives Manufacturing
The landscape of fine chemical synthesis is undergoing a significant transformation driven by the urgent need for greener, more efficient manufacturing processes. A pivotal development in this arena is detailed in patent CN115819215A, which discloses a novel method for synthesizing carboxylic acid derivatives using Lewis acid catalysis. This technology represents a substantial departure from conventional hydrolysis methods that rely on harsh inorganic acids or bases. By utilizing organic monobasic acids as additives and Lewis acids as catalysts, this process achieves high conversion rates under remarkably mild thermal conditions. For R&D directors and procurement managers in the pharmaceutical and agrochemical sectors, this innovation offers a compelling pathway to reduce operational costs while enhancing product quality. The ability to produce critical intermediates like benzoic acid and phenylacetic acid derivatives with yields reaching up to 96% and purity exceeding 98% underscores the commercial viability of this approach.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the industrial production of carboxylic acids from nitrile compounds has been dominated by hydrolysis reactions conducted under extreme conditions. These legacy processes typically require the use of concentrated inorganic strong acids, such as sulfuric acid or hydrochloric acid, or strong alkalis like sodium hydroxide and potassium hydroxide. Such aggressive reagents necessitate high-temperature environments to drive the reaction to completion, often leading to severe equipment corrosion and significant safety hazards for plant personnel. Furthermore, the neutralization of these strong acids or bases post-reaction generates vast quantities of saline wastewater, imposing a heavy burden on environmental compliance and waste treatment infrastructure. The energy intensity required to maintain high temperatures over extended periods also contributes to inflated operational expenditures, making these conventional routes increasingly unsustainable in a modern regulatory environment focused on green chemistry principles.
The Novel Approach
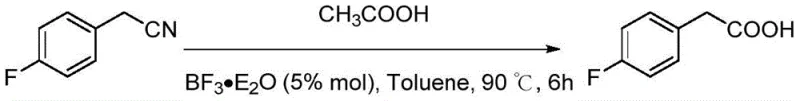
In stark contrast, the methodology outlined in patent CN115819215A introduces a sophisticated catalytic system that circumvents the drawbacks of traditional hydrolysis. By employing Lewis acids such as Ferric Chloride (FeCl3) or Boron Trifluoride Etherate (BF3·Et2O) in conjunction with organic monobasic acids like acetic acid, the reaction proceeds efficiently at temperatures between 80°C and 90°C. This moderate thermal profile not only enhances energy efficiency but also drastically reduces the risk of thermal runaway and equipment degradation. The substitution of inorganic strong acids with organic additives eliminates the generation of corrosive inorganic wastewater at the source, aligning perfectly with stringent environmental standards. Moreover, the use of high-boiling solvents like toluene facilitates easy recovery and recycling, further optimizing the material balance of the process.
Mechanistic Insights into FeCl3-Catalyzed Nitrile Hydrolysis
The efficacy of this synthetic route lies in the unique activation mechanism provided by the Lewis acid catalyst. In the presence of a Lewis acid like FeCl3, the electron-deficient metal center coordinates with the nitrogen atom of the nitrile group, thereby increasing the electrophilicity of the cyano carbon. This activation lowers the energy barrier for nucleophilic attack by water molecules or the organic acid additive, facilitating the formation of the intermediate amide and subsequent hydrolysis to the carboxylic acid. The organic monobasic acid serves a dual role: it acts as a proton source to assist in the hydrolysis steps and helps maintain a mildly acidic environment that prevents side reactions often seen under strongly alkaline conditions. This precise control over the reaction microenvironment is crucial for minimizing the formation of by-products, ensuring that the final product profile remains clean and易于 purification.
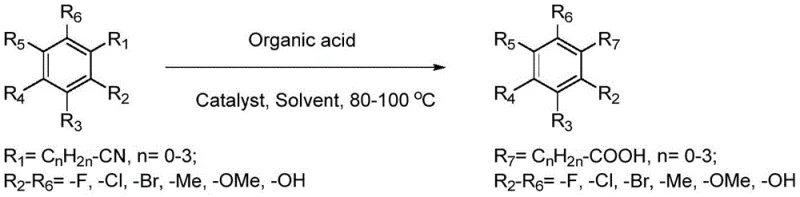
From an impurity control perspective, the mild reaction conditions play a pivotal role in preserving the integrity of sensitive functional groups on the aromatic ring. Traditional strong acid hydrolysis can often lead to undesired halogenation, sulfonation, or decomposition of labile substituents, particularly in complex pharmaceutical intermediates. The Lewis acid-catalyzed method operates under conditions that are gentle enough to tolerate a wide range of substituents, including halogens like fluorine, chlorine, and bromine, as well as alkoxy and hydroxyl groups. This chemoselectivity is evidenced by the successful synthesis of diverse derivatives such as 4-fluorobenzoic acid and 2,4-dichlorophenylacetic acid with high fidelity. The result is a product stream that requires less aggressive purification steps, directly translating to higher overall recovery rates and reduced solvent consumption during the work-up phase.
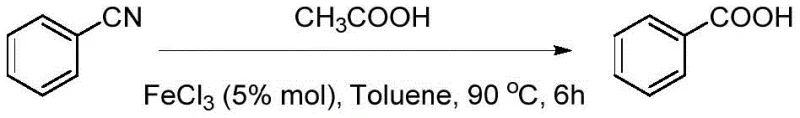
How to Synthesize Carboxylic Acid Derivatives Efficiently
Implementing this Lewis acid-catalyzed protocol requires careful attention to stoichiometry and reaction parameters to maximize yield and purity. The process begins with the precise weighing of the nitrile substrate and the organic acid additive, typically in a molar ratio ranging from 1:1 to 1:5, with 1:3 being optimal for many substrates. The choice of solvent is critical; high-boiling organic solvents such as toluene are preferred to ensure the reaction mixture remains homogeneous at the operating temperature of 90°C. The catalyst loading is kept low, generally around 5 mol% (1:0.05 molar ratio relative to the substrate), which is sufficient to drive the reaction to completion within 4 to 6 hours. Following the reaction, the work-up involves vacuum distillation to recover the solvent and unreacted acid, followed by a decolorization step using activated carbon and final recrystallization from aqueous ethanol to achieve pharmaceutical-grade purity.
- Combine the nitrile compound, organic monobasic acid (e.g., acetic acid), Lewis acid catalyst (e.g., FeCl3), and high-boiling solvent (e.g., toluene) in a reaction vessel.
- Heat the reaction mixture to 80-90°C and maintain for 4-6 hours to ensure complete conversion of the nitrile group.
- Purify the crude product through vacuum distillation to recover solvents, followed by decolorization with activated carbon and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this Lewis acid-catalyzed synthesis offers tangible strategic benefits that extend beyond mere technical performance. The elimination of corrosive inorganic acids and bases fundamentally alters the cost structure of manufacturing carboxylic acid derivatives. By removing the need for expensive corrosion-resistant reactors and extensive wastewater neutralization facilities, capital expenditure (CAPEX) and operational expenditure (OPEX) are significantly reduced. The milder reaction conditions also imply lower energy consumption for heating and cooling cycles, contributing to a smaller carbon footprint and reduced utility costs. Furthermore, the high selectivity of the process minimizes the formation of difficult-to-remove impurities, which streamlines the purification workflow and reduces the volume of solvents required for recrystallization, leading to substantial cost savings in raw material procurement.
- Cost Reduction in Manufacturing: The replacement of strong mineral acids with inexpensive Lewis acids like Ferric Chloride and organic acids like acetic acid results in a drastic reduction in raw material costs. Unlike traditional methods that generate large volumes of saline waste requiring costly disposal, this process produces minimal hazardous waste, thereby lowering environmental compliance fees. The ability to recover and recycle the toluene solvent further enhances the economic efficiency of the process, ensuring that the cost per kilogram of the final active pharmaceutical ingredient (API) intermediate is optimized for maximum margin.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures consistent batch-to-batch quality, which is critical for maintaining uninterrupted supply chains in the pharmaceutical industry. The tolerance of the method towards various functional groups means that a single platform technology can be applied to synthesize a wide array of intermediates, reducing the need for multiple specialized production lines. This flexibility allows manufacturers to respond rapidly to market demands for different carboxylic acid derivatives without significant retooling or process re-validation, thereby shortening lead times for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the absence of exothermic hazards associated with strong acid dilution. The mild operating temperature of 90°C is easily manageable in standard glass-lined or stainless steel reactors, facilitating seamless scale-up from 100 kgs to 100 MT annual capacity. Additionally, the process aligns with green chemistry principles by avoiding the generation of corrosive inorganic wastewater, simplifying the permitting process for new facilities and ensuring long-term operational sustainability in regions with strict environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Lewis acid-catalyzed synthesis method. These insights are derived directly from the experimental data and technical specifications provided in patent CN115819215A, offering a clear understanding of the process capabilities and limitations. Understanding these details is essential for R&D teams evaluating the feasibility of adopting this technology for their specific product portfolios and for procurement professionals assessing the reliability of suppliers utilizing this method.
Q: What are the primary advantages of this Lewis acid method over traditional hydrolysis?
A: This method replaces corrosive strong acids or bases with organic monobasic acids and Lewis acid catalysts, significantly reducing wastewater treatment costs and equipment corrosion while operating at lower temperatures (80-90°C).
Q: Which catalysts are preferred for this synthesis according to the patent?
A: The patent specifically highlights Ferric Chloride (FeCl3) and Boron Trifluoride Etherate (BF3·Et2O) as highly effective and cost-efficient catalysts for this transformation.
Q: What purity levels can be achieved with this process?
A: Experimental data in the patent demonstrates that products obtained via this route consistently achieve purity levels exceeding 98%, with reaction yields ranging from 85% to 96%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carboxylic Acid Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in CN115819215A for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of critical materials. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of carboxylic acid derivatives meets the exacting standards required for drug substance manufacturing. We are committed to leveraging such innovative synthetic routes to deliver superior quality products while maintaining competitive pricing structures for our global partners.
We invite you to collaborate with us to explore how this efficient Lewis acid-catalyzed method can be integrated into your supply chain to drive down costs and improve sustainability. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs, demonstrating the economic benefits of switching to this greener synthesis platform. Please contact our technical procurement team today to request specific COA data for our available carboxylic acid derivatives and to discuss route feasibility assessments for your upcoming projects. Let us be your trusted partner in navigating the complexities of modern fine chemical manufacturing.
