Advanced Catalytic Synthesis of Arformoterol Free Base for Commercial Scale-up
Introduction to Patent CN112279776B: A Paradigm Shift in Beta-Agonist Manufacturing
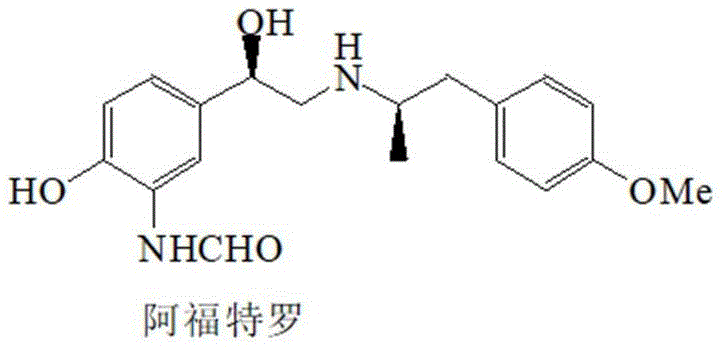
The pharmaceutical industry constantly seeks more efficient pathways for producing high-value active pharmaceutical ingredients (APIs) and their critical intermediates. Patent CN112279776B introduces a groundbreaking methodology for the synthesis of arformoterol free base, a potent long-acting beta-2 adrenergic receptor agonist used in treating severe chronic bronchial asthma. This technology addresses the longstanding inefficiencies of prior art by implementing a telescoped synthetic route that eliminates the need for isolating solid intermediates between reaction steps. By integrating chiral reduction, catalytic hydrogenation, and cyclization into a continuous flow of operations, the process achieves a total yield of approximately 50% with an enantiomeric excess (ee) exceeding 95%. For global procurement teams and R&D directors, this represents a significant opportunity for cost reduction in pharmaceutical intermediates manufacturing, as it removes the bottlenecks associated with traditional purification methods.
Furthermore, the strategic replacement of hazardous reagents with safer catalytic systems enhances the overall sustainability profile of the production line. The patent details a robust protocol that maintains mild reaction conditions, typically between 5°C and 40°C, which minimizes energy consumption and thermal degradation risks. This approach not only streamlines the supply chain for high-purity pharmaceutical intermediates but also aligns with modern green chemistry principles by reducing solvent waste. As we analyze the technical specifics, it becomes clear that this innovation offers a reliable arformoterol free base supplier pathway that is both economically and environmentally superior to legacy methods.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of arformoterol has been plagued by complex operational challenges that drive up costs and extend lead times. As illustrated in the prior art schematic, traditional routes often rely on Raney Nickel for reduction steps, a material known for its pyrophoric nature which introduces severe safety hazards during handling and storage. Moreover, conventional processes frequently necessitate the use of preparative column chromatography to separate optical isomers, a technique that is notoriously inefficient for large-scale commercial production due to high solvent consumption and low throughput. These factors combine to create a fragile supply chain where production batches are prone to delays and quality inconsistencies. The reliance on such archaic separation techniques also results in substantial material loss, driving the effective cost per kilogram to prohibitive levels for generic manufacturers.
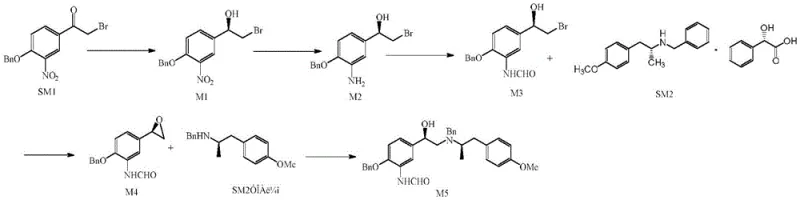
The Novel Approach
In stark contrast, the methodology disclosed in CN112279776B utilizes a sophisticated cascade of reactions that bypasses the need for intermediate isolation. The new route employs Platinum Dioxide (PtO2) and Palladium Carbon catalysts, which offer superior stability and activity compared to Raney Nickel, thereby mitigating safety risks while improving reaction kinetics. By designing the synthesis so that the reaction solution from one step flows directly into the next after simple filtration or concentration, the process eliminates the time-consuming drying and purification stages that typically dominate production schedules. This telescoping strategy not only accelerates the commercial scale-up of complex pharmaceutical intermediates but also ensures a more consistent impurity profile. The result is a streamlined workflow that significantly lowers the barrier to entry for high-volume manufacturing.
Mechanistic Insights into Chiral Reduction and Catalytic Hydrogenation
The cornerstone of this advanced synthesis lies in the precise stereochemical control exerted during the initial reduction phase. The process initiates with the carbonyl chiral reduction of 3'-nitro-4'-benzyloxy-2-bromoacetophenone (SM1) using borane dimethyl sulfide in the presence of a chiral modifier, specifically (1R,2S)-1-amino-2-indanone. This chiral auxiliary directs the hydride attack to favor the formation of the desired (R)-configured alcohol intermediate (M1) with high fidelity. The reaction is conducted at low temperatures, typically between 5°C and 10°C, to suppress non-selective background reduction and ensure that the enantiomeric excess remains above 95%. This level of stereocontrol is critical because the pharmacological activity of the (R,R)-formoterol isomer is reported to be 1000 times greater than its (S,S) counterpart, making purity a non-negotiable parameter for clinical efficacy.
Following the chiral induction, the nitro group is reduced to an amine via catalytic hydrogenation using Platinum Dioxide under a hydrogen pressure of 2.0 MPa. This step is performed in a mixed solvent system of tetrahydrofuran and toluene, which optimizes the solubility of the intermediate while maintaining catalyst suspension. The subsequent formylation and cyclization steps are equally critical, utilizing a mixture of formic acid and acetic anhydride to install the formamide group, followed by base-mediated ring closure with potassium carbonate. Each transformation is carefully tuned to proceed without precipitating the intermediate as a solid, allowing the reaction mixture to remain homogeneous or easily filterable. This continuity prevents the exposure of sensitive functional groups to harsh isolation conditions, thereby preserving the integrity of the chiral centers throughout the synthesis.
How to Synthesize Arformoterol Free Base Efficiently
The implementation of this synthesis route requires strict adherence to the defined solvent ratios and temperature profiles to maximize yield and purity. The process begins with the dissolution of the starting ketone in anhydrous tetrahydrofuran, followed by the controlled addition of the borane complex under nitrogen protection. Subsequent steps involve sequential additions of hydrogenation catalysts, formylating agents, and cyclization bases without breaking the vacuum or exposing the mixture to atmospheric moisture. Detailed standardized operating procedures regarding specific molar equivalents, stirring rates, and quenching protocols are essential for reproducibility. For a comprehensive breakdown of the exact experimental parameters and stoichiometry required for each stage, please refer to the technical guide below.
- Perform chiral reduction of 3'-nitro-4'-benzyloxy-2-bromoacetophenone (SM1) using borane dimethyl sulfide and a chiral amino-indanone catalyst in THF at 5-10°C.
- Conduct direct hydrogenation of the nitro group using Platinum Dioxide (PtO2) at 2.0 MPa pressure in a THF/Toluene mixture to generate the amine intermediate.
- Execute formylation using a formic acid/acetic anhydride mixture, followed by cyclization with potassium carbonate and condensation to form the precursor M5.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology translates into tangible operational improvements that go beyond simple yield metrics. The elimination of column chromatography and solid-state intermediate handling fundamentally alters the cost structure of production, removing some of the most expensive and time-intensive unit operations from the workflow. This simplification allows for a drastic reduction in solvent usage and waste generation, which directly correlates to lower disposal costs and a smaller environmental footprint. Furthermore, the use of stable catalysts like PtO2 and Pd-C reduces the risk of batch failures due to reagent instability, ensuring a more predictable delivery schedule for downstream API manufacturers.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the telescoping of reaction steps, which effectively merges multiple discrete processes into a single continuous operation. By avoiding the isolation of intermediates, the manufacturer saves significantly on filtration, drying, and packaging materials, while also reducing the labor hours required for each batch. The removal of column chromatography is particularly impactful, as this technique is often the most costly aspect of fine chemical purification due to the vast volumes of silica and solvents involved. Consequently, the overall cost of goods sold (COGS) is substantially lowered, providing a competitive pricing advantage in the global market.
- Enhanced Supply Chain Reliability: Safety is a critical component of supply chain continuity, and the replacement of pyrophoric Raney Nickel with stable platinum and palladium catalysts mitigates the risk of fire-related shutdowns. The milder reaction conditions, operating generally below 40°C for most steps, reduce the thermal load on reactor equipment and minimize the potential for runaway reactions. This stability ensures that production facilities can maintain consistent output without the frequent interruptions associated with hazardous material handling. Additionally, the high selectivity of the chiral reduction step reduces the burden on quality control labs, speeding up the release of finished goods for shipment.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing common industrial solvents like toluene, methanol, and THF that are readily available in bulk quantities. The reduction in total solvent volume per kilogram of product simplifies solvent recovery and recycling operations, aiding facilities in meeting stringent environmental regulations. The high atom economy of the catalytic steps means less chemical waste is generated per unit of product, aligning with the growing demand for sustainable manufacturing practices. This makes the technology an ideal candidate for expansion from pilot scale to multi-ton commercial production without requiring extensive re-engineering of the plant infrastructure.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis method is vital for stakeholders evaluating its potential for integration into existing supply chains. The following questions address common concerns regarding safety, purity, and scalability, drawing directly from the data provided in the patent documentation. These insights are intended to clarify how the new methodology overcomes the specific limitations of previous generations of synthetic routes. By addressing these points, we aim to provide a transparent view of the technology's capabilities and limitations.
Q: How does the new synthesis method improve safety compared to traditional Raney Nickel routes?
A: The patented method replaces pyrophoric Raney Nickel with stable Platinum Dioxide (PtO2) and Palladium Carbon catalysts under controlled hydrogen pressure, significantly reducing fire hazards and operational risks.
Q: What is the impact of telescoping reactions on the overall production cost?
A: By eliminating the isolation and purification of solid intermediates and avoiding expensive column chromatography for isomer separation, the process drastically reduces solvent consumption, labor time, and material loss.
Q: What optical purity (ee value) can be achieved with this chiral reduction strategy?
A: The use of (1R,2S)-1-amino-2-indanone as a chiral auxiliary in the borane reduction step ensures high stereocontrol, consistently achieving an ee value greater than 95%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Arformoterol Free Base Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the rigorous demands of the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the telescoped arformoterol synthesis can be seamlessly transferred from the laboratory to the manufacturing floor. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch meets the highest international standards. Our infrastructure is designed to handle complex chiral chemistries safely and efficiently, making us a strategic partner for companies seeking to optimize their API supply chains.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis that quantifies the potential efficiencies of switching to this new route. Please contact us to request specific COA data and route feasibility assessments tailored to your volume requirements. Together, we can drive down costs and enhance the reliability of your arformoterol supply chain.
