Revolutionizing Sartan Intermediate Production via Solar-Initiated Bromination Technology
Revolutionizing Sartan Intermediate Production via Solar-Initiated Bromination Technology
The pharmaceutical industry is constantly seeking more sustainable and efficient pathways for the synthesis of critical active pharmaceutical ingredient (API) intermediates, particularly those used in the vast class of antihypertensive drugs known as Sartans. Patent CN101648839A introduces a groundbreaking green synthesis method for producing bromomethylbiphenyl compounds, which serve as the foundational building blocks for these life-saving medications. This technology represents a significant paradigm shift from traditional thermal initiation methods to a photochemical approach driven by natural sunlight. By leveraging renewable solar energy, this process not only aligns with modern environmental sustainability goals but also addresses critical pain points regarding safety, cost, and product purity that have long plagued the manufacturing of biphenyl derivatives. The core innovation lies in the ability to achieve high conversion rates and exceptional selectivity without the need for hazardous chemical initiators, thereby streamlining the supply chain for reliable pharmaceutical intermediate suppliers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
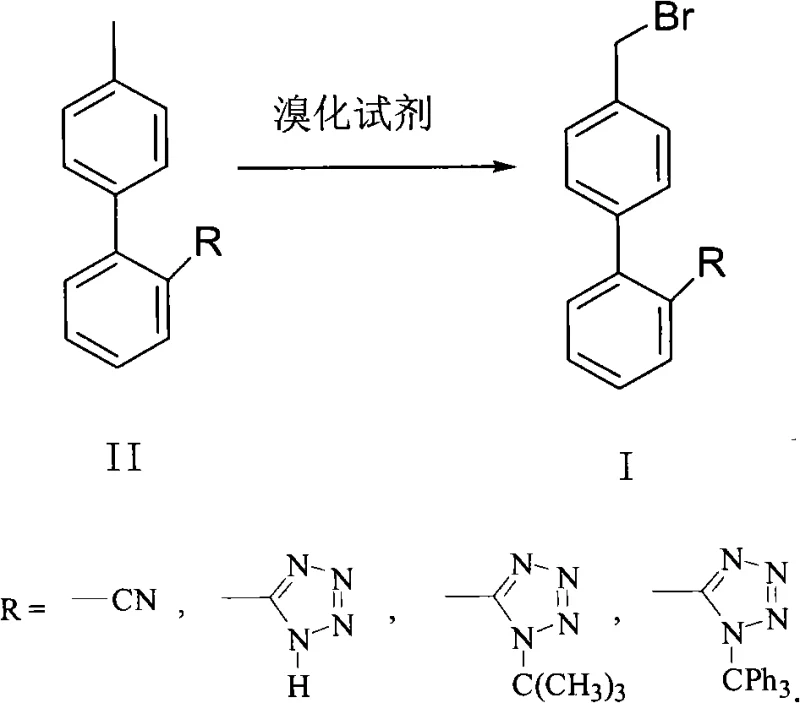
Historically, the synthesis of 4'-bromomethyl-2-substituted biphenyl compounds, essential for drugs like Losartan and Valsartan, has relied heavily on thermal radical bromination. Traditional protocols typically employ elemental bromine or N-bromosuccinimide (NBS) in conjunction with thermal initiators such as AIBN (azobisisobutyronitrile), ABVN, or BPO (benzoyl peroxide). These conventional methods suffer from inherent inefficiencies and safety hazards; they strictly require elevated reaction temperatures, often exceeding 40°C, to effectively decompose the initiators and generate the necessary radicals. If the temperature is not maintained sufficiently high, the initiating effect is poor, leading to incomplete reactions and low yields. Furthermore, the use of excessive amounts of these chemical initiators introduces potential safety hazards due to their explosive nature and creates significant cost pressure. From a quality perspective, these thermal processes often struggle with selectivity, typically achieving conversion rates of only 80-84% and selectivity of 85-90%, necessitating complex downstream purification to remove toxic initiator residues and byproducts.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes natural sunlight as the sole initiation source, fundamentally altering the energy profile of the reaction. This photo-initiation method operates effectively at much milder temperatures, preferably between 0°C and 20°C, eliminating the need for energy-intensive heating systems. By replacing hazardous thermal initiators with renewable solar energy, the process drastically reduces the risk of thermal runaway and explosion, enhancing overall plant safety. The use of dibromohydantoin as a preferred brominating agent in this photochemical system further enhances the reaction profile, allowing for conversion rates reaching 87-92% and selectivity up to 92-95%. This method not only simplifies the operational procedure by removing the handling of sensitive initiators but also significantly improves the atom economy. The result is a cleaner reaction profile that facilitates easier purification, yielding high-purity products suitable for direct use in subsequent coupling reactions for API synthesis, thus positioning this technology as a superior choice for cost reduction in API manufacturing.
Mechanistic Insights into Photo-Initiated Radical Bromination
The mechanistic foundation of this green synthesis relies on the homolytic cleavage of the brominating agent induced by photon absorption rather than thermal decomposition. When the reaction mixture containing the 4'-methyl-2-substituted biphenyl substrate and dibromohydantoin is exposed to sunlight, the energy from the photons promotes the brominating agent to an excited state, facilitating the generation of bromine radicals. These highly reactive radicals selectively abstract a hydrogen atom from the benzylic methyl group of the biphenyl substrate, forming a stable benzylic radical intermediate. This intermediate subsequently reacts with another molecule of the brominating agent to yield the desired 4'-bromomethyl product while regenerating a bromine radical to propagate the chain reaction. The absence of thermal initiators like AIBN prevents the formation of cyano-isopropyl radicals or other initiator-derived fragments that could otherwise participate in side reactions or remain as difficult-to-remove impurities. This clean radical generation mechanism is crucial for maintaining the integrity of sensitive functional groups, such as the tetrazole ring found in many Sartan precursors, which might degrade under harsher thermal conditions.
Controlling the selectivity between monobromination (Formula I) and dibromination (Formula III) is a critical aspect of this mechanism, governed largely by the stoichiometry of the brominating agent and the reaction temperature. The patent highlights that maintaining the reaction temperature within the 0-20°C range is vital for suppressing the formation of the dibrominated byproduct. At higher temperatures or with excessive brominating agent, the reactivity of the newly formed bromomethyl group increases, leading to further substitution. However, under the optimized photochemical conditions with controlled light intensity and temperature, the kinetic preference for the initial hydrogen abstraction at the methyl group is maximized. Additionally, the use of solvents like dichloromethane provides an ideal medium that solubilizes both the organic substrate and the brominating agent while remaining inert to the radical species. The ability to fine-tune the reaction time (30 minutes to 6 hours) based on light intensity allows operators to quench the reaction precisely at the point of maximum monobromide yield, ensuring that the impurity profile remains well within the stringent specifications required for high-purity pharmaceutical intermediates.
How to Synthesize 4'-Bromomethyl-2-substituted Biphenyl Efficiently
The synthesis protocol outlined in the patent offers a straightforward yet highly effective pathway for producing these critical intermediates, emphasizing operational simplicity and reproducibility. The process begins by dissolving the starting material, such as 4'-methyl-2-cyanobiphenyl (OTBN), in a halogenated hydrocarbon solvent, with dichloromethane being the preferred choice due to its excellent solvation properties and ease of removal. A stoichiometric amount of dibromohydantoin is then added to the solution, and the mixture is cooled to a low temperature range of 0-20°C using a standard low-temperature bath. Unlike traditional methods that require heating mantles or oil baths, this setup relies on ambient or artificial light sources, though natural sunlight is highlighted for its cost-effectiveness and sustainability. The reaction progress is monitored via HPLC, and upon completion, the workup involves simple aqueous washes to remove succinimide byproducts and unreacted reagents. For a comprehensive understanding of the specific operational parameters and safety precautions, the detailed standardized synthesis steps are provided in the guide below.
- Dissolve the 4'-methyl-2-substituted biphenyl compound in an organic solvent such as dichloromethane.
- Add a brominating agent, preferably dibromohydantoin, to the solution while maintaining the temperature between 0°C and 20°C.
- Expose the reaction mixture to natural sunlight irradiation for 2 to 4 hours to initiate the bromination without chemical initiators.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this sunlight-driven synthesis method presents a compelling value proposition centered around cost efficiency, safety, and supply continuity. The elimination of expensive and hazardous chemical initiators like AIBN and BPO directly translates to reduced raw material costs and lowers the regulatory burden associated with storing and handling explosive substances. Furthermore, the simplified purification process, resulting from higher selectivity and fewer byproducts, reduces the consumption of solvents and adsorbents during the workup phase, contributing to substantial cost savings in waste management and material usage. The reliance on natural sunlight as an energy source not only reduces utility costs associated with heating but also aligns with corporate sustainability mandates, potentially qualifying the manufacturing site for green chemistry incentives. This robust and scalable process ensures a steady supply of high-quality intermediates, mitigating the risk of production delays caused by initiator shortages or thermal regulation failures.
- Cost Reduction in Manufacturing: The removal of thermal initiators eliminates a significant line item in the bill of materials, as reagents like AIBN are not only costly but also require specialized storage and handling protocols that add to overhead expenses. By utilizing dibromohydantoin and sunlight, the process reduces the complexity of the reaction setup, removing the need for precise thermal control systems required to decompose initiators safely. The high selectivity of the reaction minimizes the loss of valuable starting materials to over-brominated byproducts, thereby improving the overall mass balance and yield efficiency. Additionally, the simplified downstream processing reduces the labor and time required for purification, leading to a leaner manufacturing operation with lower operational expenditures.
- Enhanced Supply Chain Reliability: The raw materials required for this process, including the biphenyl precursors and dibromohydantoin, are commercially available in bulk quantities from multiple global suppliers, ensuring a resilient supply chain that is not dependent on single-source specialty chemicals. The mild reaction conditions reduce the wear and tear on reactor equipment and minimize the risk of unplanned shutdowns due to thermal excursions or initiator-related safety incidents. This reliability is crucial for maintaining consistent delivery schedules to API manufacturers, who depend on a uninterrupted flow of intermediates to meet their own production targets. The scalability of the photochemical process allows for flexible production volumes, enabling suppliers to respond quickly to fluctuations in market demand without significant capital investment in new infrastructure.
- Scalability and Environmental Compliance: The green nature of this synthesis method significantly reduces the generation of hazardous waste, particularly the nitrogen-containing residues associated with azo initiators, simplifying compliance with increasingly stringent environmental regulations. The use of dichloromethane, while requiring proper containment, is a well-understood solvent in the industry with established recovery and recycling protocols, further minimizing the environmental footprint. The process is inherently safer, reducing the potential for catastrophic accidents related to thermal runaway, which is a major concern for insurance and liability purposes in large-scale chemical manufacturing. This combination of safety, environmental friendliness, and operational simplicity makes the technology highly attractive for commercial scale-up of complex pharmaceutical intermediates, ensuring long-term viability in a regulated market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photochemical bromination technology, derived directly from the patent specifications and experimental data. Understanding these details is essential for technical teams evaluating the feasibility of integrating this green method into existing production lines. The answers provided reflect the specific advantages observed in the patent examples, highlighting the practical benefits of switching from thermal to solar initiation.
Q: What are the advantages of using sunlight over thermal initiators like AIBN?
A: Using sunlight eliminates the need for hazardous chemical initiators like AIBN or BPO, which require high temperatures (>40°C) and pose safety risks. The photochemical method operates at mild temperatures (0-20°C), significantly improving process safety and reducing toxic residue in the final product.
Q: How does this method control selectivity against dibromination?
A: By carefully controlling the reaction temperature (0-20°C) and utilizing specific brominating agents like dibromohydantoin under light irradiation, the process achieves high selectivity (92-95%) for the monobrominated product, minimizing the formation of unwanted dibrominated byproducts.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method is designed for scalability. It uses cheap, readily available raw materials and solvents, avoids expensive catalysts, and simplifies purification steps due to high product purity, making it highly suitable for small-batch to industrial-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bromomethylbiphenyl Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the green synthesis route described in patent CN101648839A for the production of high-value Sartan intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped to handle photochemical reactions safely and effectively, adhering to stringent purity specifications and rigorous QC labs to guarantee that every batch meets the exacting standards required by global pharmaceutical regulators. We are committed to delivering not just a product, but a reliable supply solution that enhances your manufacturing efficiency.
We invite you to collaborate with us to optimize your supply chain for bromomethylbiphenyl compounds and other critical pharmaceutical building blocks. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and competitiveness in your API production. Let us help you engineer a more sustainable and cost-effective future for your pharmaceutical projects.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
