Advanced Synthetic Route for Telmisartan Intermediates Enhancing Commercial Scalability and Purity
Advanced Synthetic Route for Telmisartan Intermediates Enhancing Commercial Scalability and Purity
The pharmaceutical industry continuously seeks robust synthetic pathways that balance chemical efficiency with commercial viability, particularly for high-volume antihypertensive agents like Telmisartan. A pivotal advancement in this domain is documented in patent CN1182122C, which outlines a novel preparation method for Telmisartan, a potent non-peptide angiotensin II (ATII) receptor antagonist. This intellectual property addresses critical bottlenecks in traditional manufacturing by replacing unstable tert-butyl protecting groups with more resilient methyl and ethyl ester derivatives. The strategic shift not only stabilizes the key intermediate, 4'-bromomethylbiphenyl-2-carboxylic acid ester, but also streamlines the downstream hydrolysis process. For stakeholders evaluating the landscape of reliable pharmaceutical intermediates supplier options, understanding these mechanistic improvements is essential for securing long-term supply chain continuity.
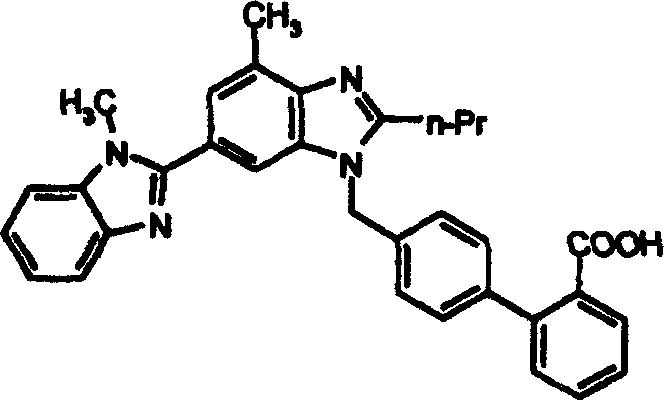
Telmisartan represents a cornerstone therapy in cardiovascular medicine, necessitating production methods that guarantee exceptional purity and consistency. The structural complexity of the molecule, featuring a biphenyl system linked to a benzimidazole core, demands precise control over functional group transformations. The innovation presented in the referenced patent focuses on the alkylation step where the benzimidazole nitrogen attacks the bromomethyl group of the biphenyl derivative. By optimizing the protecting group strategy on the carboxylic acid moiety, the process mitigates the risks associated with acid-sensitive intermediates. This technical refinement is crucial for any organization aiming for cost reduction in API manufacturing while maintaining rigorous quality standards required by global regulatory bodies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Telmisartan relied heavily on the use of 4'-bromomethylbiphenyl-2-carboxylic acid tert-butyl ester as a key coupling partner. While conceptually sound, this tert-butyl protected species exhibits significant instability, particularly under slightly acidic conditions often encountered during workup or storage. This inherent instability leads to the premature cleavage of the protecting group, generating free carboxylic acid byproducts that complicate the reaction matrix. Consequently, the nucleophilic substitution with the benzimidazole intermediate becomes difficult to control, resulting in a heterogeneous mixture of products and a proliferation of impurities. Furthermore, the removal of the tert-butyl group in the final deprotection step often requires harsh acidic conditions that can degrade the sensitive benzimidazole rings, thereby reducing the overall yield and compromising the quality of the final active pharmaceutical ingredient.
The Novel Approach
In stark contrast, the methodology described in patent CN1182122C utilizes methyl or ethyl esters as protecting groups for the biphenyl carboxylic acid component. These lower alkyl esters possess superior thermal and chemical stability compared to their tert-butyl counterparts, ensuring that the integrity of the molecule is maintained throughout the nucleophilic substitution phase. The reaction between the benzimidazole intermediate and the methyl or ethyl ester derivative proceeds with greater predictability and ease of control, significantly minimizing the formation of side products. Moreover, the hydrolysis of these methyl or ethyl esters to reveal the final carboxylic acid is straightforward and can be achieved under mild acidic or alkaline conditions. This operational simplicity translates directly into a more robust manufacturing process that is highly amenable to commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Nucleophilic Substitution and Ester Hydrolysis
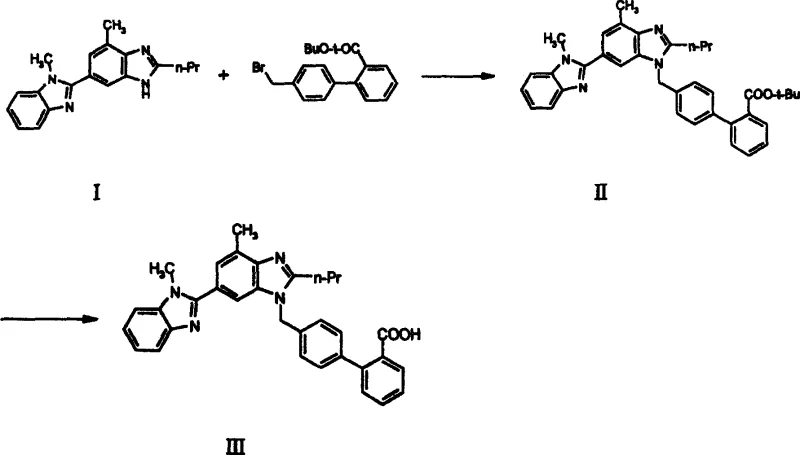
The core of this synthetic innovation lies in the optimization of the nucleophilic substitution reaction between 2-n-propyl-4-methyl-6-(1-methylbenzimidazol-2-yl)benzimidazole and the halogenated biphenyl derivative. In this transformation, the nitrogen atom of the benzimidazole ring acts as a nucleophile, displacing the bromide ion from the bromomethyl group. The choice of base is critical in this mechanism; the patent highlights the efficacy of both inorganic bases like potassium carbonate and organic bases such as sodium ethoxide or triethylamine. These bases serve to deprotonate the benzimidazole nitrogen, enhancing its nucleophilicity without inducing unwanted side reactions on the ester functionality. The stability of the methyl and ethyl esters allows for a wider range of reaction temperatures, typically between 20°C and 100°C, providing process engineers with flexibility to optimize reaction kinetics without fear of ester degradation.
Following the alkylation, the conversion of the intermediate ester to the final Telmisartan acid involves a hydrolysis mechanism that is markedly more efficient than traditional deprotection methods. Whether conducted under acidic conditions using hydrochloric or sulfuric acid, or under alkaline conditions using sodium hydroxide, the cleavage of the methyl or ethyl ester bond is rapid and quantitative. This step is crucial for impurity control, as the mild conditions prevent the decomposition of the biphenyl-benzimidazole scaffold. The ability to perform this hydrolysis in mixed solvent systems, such as alcohol-water combinations, further enhances the solubility of intermediates and facilitates the isolation of the final product. This refined mechanistic pathway ensures that the final high-purity Telmisartan meets stringent specifications with minimal downstream purification requirements.
How to Synthesize Telmisartan Efficiently
The implementation of this synthetic route involves a streamlined sequence designed to maximize throughput and minimize waste generation. The process begins with the careful selection of reagents, specifically opting for the methyl or ethyl ester variants of the bromomethyl biphenyl precursor. The reaction environment must be controlled to ensure complete conversion while avoiding excessive thermal stress. Following the coupling reaction, standard workup procedures involving extraction and washing are employed to isolate the crude ester intermediate. This intermediate is sufficiently stable to be carried forward without extensive purification, although recrystallization can be performed if higher purity is desired prior to hydrolysis. The final hydrolysis step is the gateway to the active drug substance, requiring precise pH control to ensure complete deprotection and precipitation of the product.
- Perform nucleophilic substitution between 2-n-propyl-4-methyl-6-(1-methylbenzimidazol-2-yl)benzimidazole and methyl or ethyl 4'-bromomethylbiphenyl-2-carboxylate using a base catalyst.
- Isolate the intermediate ester (IVa or IVb) through extraction and crystallization, ensuring high purity before the next step.
- Hydrolyze the ester intermediate under acidic or alkaline conditions to remove the protecting group and obtain the final Telmisartan acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this methyl/ethyl ester-based synthesis offers profound logistical and economic benefits. The primary advantage stems from the enhanced stability of the starting materials and intermediates. Unlike the tert-butyl protected analogs which require careful handling and specific storage conditions to prevent degradation, the methyl and ethyl esters are robust commodities that can be sourced reliably and stored for extended periods without quality loss. This stability drastically simplifies inventory management and reduces the risk of raw material spoilage, contributing to substantial cost savings in the overall procurement strategy. Furthermore, the simplified reaction profile reduces the need for complex monitoring and intervention during production, lowering operational overheads.
- Cost Reduction in Manufacturing: The elimination of unstable protecting groups removes the necessity for specialized handling protocols and reduces the incidence of batch failures due to reagent degradation. By utilizing common methyl and ethyl esters, the process leverages widely available and cost-effective raw materials. The improved yield and reduced impurity profile mean that less material is wasted during purification, directly lowering the cost of goods sold. Additionally, the milder hydrolysis conditions reduce energy consumption and extend the lifespan of reactor equipment by minimizing exposure to corrosive environments.
- Enhanced Supply Chain Reliability: The robustness of the methyl and ethyl ester intermediates ensures a consistent supply of high-quality precursors, mitigating the risk of production delays caused by raw material variability. Since these esters are easier to synthesize and stabilize, suppliers can maintain higher safety stocks and respond more agilely to fluctuations in demand. This reliability is critical for maintaining uninterrupted production schedules for life-saving antihypertensive medications. The simplified process also reduces dependency on niche reagents, diversifying the supplier base and strengthening the overall resilience of the supply network.
- Scalability and Environmental Compliance: The operational simplicity of this route makes it ideally suited for large-scale industrial production. The ability to run reactions at moderate temperatures and use standard solvents facilitates easy technology transfer from pilot plants to commercial manufacturing facilities. Moreover, the reduction in impurities and the use of less hazardous deprotection conditions contribute to a greener manufacturing footprint. Waste streams are easier to treat, and the overall atom economy of the process is improved, aligning with modern environmental regulations and sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this optimized synthetic pathway. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature. Understanding these nuances is vital for technical teams evaluating the feasibility of adopting this route for commercial production. The answers reflect the consensus on best practices for maximizing yield and purity while minimizing operational risks.
Q: Why is the methyl/ethyl ester protection superior to the tert-butyl group in Telmisartan synthesis?
A: The tert-butyl group is acid-labile and unstable during operation, leading to premature deprotection and impurity formation. Methyl and ethyl esters offer superior stability, allowing for better reaction control and easier purification.
Q: What are the optimal conditions for the nucleophilic substitution step?
A: The reaction proceeds efficiently at temperatures between 20°C and 100°C using bases such as potassium carbonate or sodium ethoxide in solvents like DMF or DMSO, typically completing within 8 to 10 hours.
Q: How does this new route impact the final product quality?
A: By minimizing side reactions associated with unstable protecting groups, this route significantly reduces impurity levels, resulting in higher yields and improved overall product quality suitable for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Telmisartan Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of employing advanced synthetic strategies to deliver high-value pharmaceutical intermediates. Our technical team has extensively analyzed routes such as the one described in CN1182122C to ensure our manufacturing processes are aligned with the latest industry standards for efficiency and quality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that whether you require pilot quantities or full-scale commercial supply, our capabilities match your needs. Our commitment to stringent purity specifications is backed by state-of-the-art rigorous QC labs that monitor every batch for impurities and identity, guaranteeing that our Telmisartan intermediates meet the highest global pharmacopoeial standards.
We invite potential partners to engage with our technical procurement team to discuss how our optimized manufacturing capabilities can support your specific project requirements. By leveraging our expertise in process optimization, we can provide a Customized Cost-Saving Analysis tailored to your volume needs and quality targets. We encourage you to contact us to request specific COA data and comprehensive route feasibility assessments that demonstrate our commitment to being a dependable partner in your supply chain. Let us collaborate to bring efficient, high-quality antihypertensive solutions to the market faster and more economically.
