Advanced Synthesis Of 3,4-Disubstituted Benzaldehydes: Scaling Waste Valorization For Global Supply Chains
Introduction To Patent CN102503788B: Transforming Byproducts Into Premium Intermediates
The global demand for high-purity aromatic aldehydes, such as Veratraldehyde and Vanillin, continues to surge across the pharmaceutical and flavor industries, driving the need for more sustainable and cost-effective manufacturing routes. Patent CN102503788B introduces a groundbreaking methodology that leverages 4-(2-methylallyl)-1,2-benzenediol, a historically underutilized byproduct from furanol synthesis, as a versatile starting material. This innovation represents a paradigm shift in fine chemical manufacturing, moving away from traditional petrochemical-derived precursors towards waste valorization strategies that align with modern green chemistry principles. By establishing a robust chemical pathway to convert this specific phenolic intermediate into a spectrum of 3,4-disubstituted benzaldehydes, the technology offers a compelling solution for reducing environmental footprints while securing a stable supply of critical organic building blocks. The strategic importance of this patent lies not only in its chemical elegance but also in its potential to decouple production costs from volatile commodity markets, providing a distinct competitive advantage for manufacturers capable of implementing this divergent synthesis platform.

The Limitations Of Conventional Methods vs. The Novel Approach
The Limitations Of Conventional Methods
Traditional industrial routes for producing key aldehydes like Vanillin and Veratraldehyde have long been plagued by significant environmental and economic inefficiencies that burden modern supply chains. The prevalent nitrosation method for Vanillin, for instance, generates substantial quantities of toxic nitroso-waste and requires complex purification steps to remove heavy metal contaminants, leading to escalating disposal costs and regulatory compliance risks. Similarly, the synthesis of Veratraldehyde often relies on the Vilsmeier-Haack reaction using catechol derivatives, which necessitates the handling of hazardous phosphoryl chloride and produces corrosive acidic waste streams that demand neutralization before discharge. Furthermore, these conventional processes typically lack modularity; a production line dedicated to Vanillin cannot easily be switched to produce Piperonal or Ethyl Vanillin without extensive retooling, creating rigidity in the face of fluctuating market demands. The reliance on distinct, linear pathways for each product also means that raw material procurement must be managed separately for each SKU, increasing logistical complexity and exposing the supply chain to multiple points of failure and price volatility.
The Novel Approach
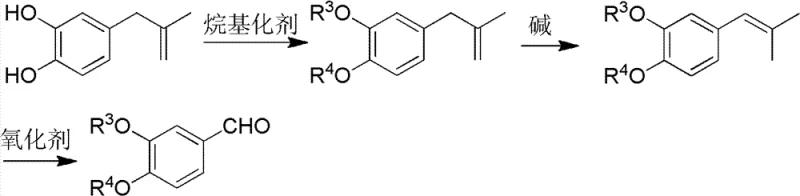
In stark contrast, the novel approach detailed in the patent capitalizes on the structural versatility of 4-(2-methylallyl)-1,2-benzenediol to create a unified, modular production platform capable of generating multiple high-value outputs from a single feedstock source. This methodology fundamentally alters the economic equation by utilizing a byproduct that is otherwise discarded or sold at negligible value, effectively negating the raw material cost component that dominates traditional pricing models. The process employs a logical sequence of alkylation, thermal isomerization, and oxidative cleavage, allowing manufacturers to tune the final product profile simply by selecting different alkylating agents such as dimethyl sulfate for Veratraldehyde or dichloromethane for Piperonal. This flexibility enables a single facility to pivot production between different aldehydes based on real-time market signals, optimizing asset utilization and inventory management. Moreover, the adoption of ozonolysis as a primary oxidation tool offers a cleaner alternative to stoichiometric metal oxidants, significantly reducing the load of inorganic salts in wastewater and simplifying the downstream purification process to achieve pharmaceutical-grade purity specifications with fewer unit operations.
Mechanistic Insights Into The Divergent Aldehyde Synthesis Platform
The core chemical ingenuity of this process rests on a precise three-step sequence that transforms the allyl side chain into an aldehyde functionality while simultaneously establishing the requisite substitution pattern on the aromatic ring. The initial alkylation step serves a dual purpose: it protects the phenolic hydroxyl groups from unwanted side reactions during subsequent thermal treatment and defines the identity of the final substituent groups (methoxy, ethoxy, or methylenedioxy). Following protection, the critical isomerization step utilizes a base-catalyzed Claisen rearrangement mechanism, where the allyl group migrates from the para-position to the ortho-position relative to the oxygen atom. This [1,3]-sigmatropic shift is thermodynamically driven and is typically conducted in high-boiling solvents like n-butanol with potassium hydroxide to ensure complete conversion to the propenyl isomer, which is the essential precursor for oxidative cleavage. The final transformation involves the scission of the carbon-carbon double bond in the propenyl side chain, a reaction that can be achieved through various oxidative protocols including ozonolysis followed by reductive workup with zinc and acetic acid, or via chemical oxidants like potassium permanganate stabilized against over-oxidation.
Controlling impurity profiles in this synthesis is paramount, particularly given the sensitivity of the aldehyde moiety to further oxidation into carboxylic acids, which would degrade product quality and yield. The patent explicitly addresses this challenge by incorporating specific stabilizers such as p-aminobenzenesulfonic acid when using strong oxidants like potassium dichromate or potassium permanganate. These additives function as sacrificial reagents or radical scavengers that moderate the oxidation potential of the reaction medium, ensuring that the cleavage stops selectively at the aldehyde stage rather than proceeding to the corresponding benzoic acid derivatives. Additionally, the use of ozonolysis provides an inherently cleaner mechanistic pathway where the formation of the ozonide intermediate allows for a controlled reductive decomposition, minimizing the formation of polymeric byproducts or ring-opened degradation species. This rigorous control over the oxidation step ensures that the resulting crude product possesses a high assay value, reducing the burden on final crystallization or distillation steps and enabling the production of high-purity pharmaceutical intermediates that meet stringent international pharmacopoeia standards without extensive reprocessing.
How To Synthesize 3,4-Disubstituted Benzaldehydes Efficiently
Implementing this synthesis route requires careful attention to reaction conditions, particularly during the isomerization and oxidation phases where temperature and reagent stoichiometry dictate success. The process begins with the alkylation of the dihydroxy starting material, followed by the base-mediated rearrangement in n-butanol at elevated temperatures to secure the ortho-propenyl configuration. Once the isomerized intermediate is isolated, the final oxidative cleavage can be tailored based on available infrastructure, choosing between catalytic air oxidation for cost sensitivity or ozonolysis for maximum purity and environmental compliance. The detailed standardized operating procedures, including specific molar ratios, temperature ramps, and workup protocols necessary to replicate the high yields reported in the patent examples, are critical for successful technology transfer and commercial adoption.
- Perform alkylation on 4-(2-methylallyl)-1,2-benzenediol using agents like dimethyl sulfate or dichloromethane to protect hydroxyl groups.
- Execute thermal isomerization using potassium hydroxide in n-butanol to rearrange the allyl group to the ortho-position.
- Conduct oxidative cleavage of the isomerized alkene using ozone or permanganate to yield the final 3,4-disubstituted benzaldehyde.
Commercial Advantages For Procurement And Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this technology offers profound strategic benefits that extend far beyond simple unit cost savings, fundamentally reshaping the risk profile of sourcing critical aromatic aldehydes. By anchoring production to a byproduct stream from the furanol industry, manufacturers can insulate their operations from the price volatility associated with virgin petrochemical feedstocks like guaiacol or catechol, which are subject to fluctuating oil prices and supply disruptions. This waste-to-value model creates a circular economy dynamic where the cost of goods sold is structurally lower, allowing for more aggressive pricing strategies or improved margin retention in a competitive marketplace. Furthermore, the divergent nature of the synthesis platform means that a single production asset can service multiple market segments, from food flavors to active pharmaceutical ingredients, enhancing capital efficiency and reducing the need for dedicated, single-product facilities that sit idle during market downturns. This operational agility translates directly into enhanced supply chain reliability, as producers can rapidly reallocate capacity to meet urgent customer demands for specific aldehydes without the long lead times associated with building new production lines.
- Cost Reduction In Manufacturing: The elimination of expensive virgin raw materials in favor of valorized byproducts drastically reduces the direct material cost component, which is typically the largest expense in fine chemical manufacturing. Additionally, the ability to use ozonolysis eliminates the need for stoichiometric amounts of heavy metal oxidants and the subsequent costly waste treatment required to remove chromium or manganese residues from the effluent. This reduction in auxiliary chemical consumption and waste disposal fees contributes to a significantly leaner cost structure, enabling substantial cost savings that can be passed down to customers or reinvested into process optimization. The streamlined purification process, resulting from higher selectivity and fewer side reactions, also lowers energy consumption and solvent usage, further compounding the economic advantages of this novel route over legacy technologies.
- Enhanced Supply Chain Reliability: Sourcing raw materials from an established byproduct stream ensures a consistent and predictable supply of the key starting material, 4-(2-methylallyl)-1,2-benzenediol, which is generated continuously during furanol production. This inherent link to another large-scale industrial process mitigates the risk of raw material shortages that often plague specialty chemical supply chains dependent on niche suppliers. Moreover, the modular design of the synthesis allows for geographic diversification of production; since the chemistry relies on common unit operations and widely available reagents, it can be replicated in multiple regions to create a resilient, multi-site supply network. This redundancy is crucial for multinational corporations seeking to reduce lead time for high-purity intermediates and ensure business continuity in the face of regional logistical disruptions or geopolitical instability.
- Scalability And Environmental Compliance: The unit operations described in the patent, such as liquid-liquid extraction, distillation, and standard oxidation reactors, are well-understood and easily scalable from pilot plant to multi-ton commercial production without encountering significant engineering bottlenecks. The process aligns perfectly with increasingly stringent global environmental regulations by minimizing the generation of hazardous waste and avoiding the use of persistent organic pollutants or toxic heavy metals in the final product stream. This environmental compatibility simplifies the permitting process for new facilities and reduces the long-term liability associated with environmental remediation, making it a future-proof investment for sustainable chemical manufacturing. The ability to produce pharmaceutical-grade intermediates with a cleaner environmental profile also enhances the brand value of downstream customers who are under pressure to demonstrate sustainability in their own supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this synthesis technology, providing clarity for stakeholders evaluating its potential impact on their operations. These insights are derived directly from the experimental data and claims within the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these nuances is essential for assessing the feasibility of integrating this route into existing manufacturing portfolios and for leveraging its unique advantages in negotiations with suppliers.
Q: What is the primary raw material advantage of this synthesis method?
A: The process utilizes 4-(2-methylallyl)-1,2-benzenediol, which is a byproduct generated during the industrial synthesis of furanol. This transforms a low-value waste stream into high-value fragrance and pharmaceutical intermediates, significantly reducing raw material costs compared to starting from catechol or guaiacol.
Q: How does the patent address the issue of over-oxidation during the final step?
A: When using strong oxidants like potassium dichromate or potassium permanganate, the method specifies the addition of p-aminobenzenesulfonic acid or p-aminobenzaldehyde. These additives act as stabilizers to prevent the deep oxidation of the sensitive aldehyde group into carboxylic acids, thereby improving yield and purity.
Q: Can this platform technology produce multiple different end products?
A: Yes, the technology is highly divergent. By varying the alkylation agent (e.g., methyl vs. ethyl vs. methylene bridging), the same core intermediate can be converted into Veratraldehyde, Ethyl Vanillin, Piperonal (Heliotropin), or 3,4-Dihydroxybenzaldehyde, offering immense supply chain flexibility.
Partnering With NINGBO INNO PHARMCHEM: Your Reliable Veratraldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthesis methods described in CN102503788B and possess the technical expertise to bring this innovative chemistry to commercial reality. As a leading CDMO partner, we have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory concept to industrial volume is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific unit operations required for this process, including safe ozonolysis capabilities and advanced distillation columns for high-purity separation, guaranteeing stringent purity specifications that meet the rigorous demands of the global pharmaceutical and flavor industries. Our rigorous QC labs employ advanced analytical techniques to monitor every stage of the synthesis, ensuring that impurity profiles are tightly controlled and that every batch delivers consistent quality.
We invite you to collaborate with us to unlock the value of this waste-to-value technology for your specific application needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this novel route for your specific volume requirements. We encourage you to contact us to request specific COA data from our pilot runs and to discuss route feasibility assessments tailored to your supply chain goals. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable flavor & fragrance intermediate supplier committed to driving innovation, sustainability, and cost efficiency in the fine chemical sector.
