Advanced Veratraldehyde Manufacturing: High-Purity Synthesis from Catechol for Global Pharma Supply Chains
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes that balance cost-efficiency with high purity standards, a challenge effectively addressed by the methodology disclosed in patent CN102070421A. This intellectual property outlines a sophisticated two-step synthesis of veratraldehyde, a critical intermediate for numerous active pharmaceutical ingredients (APIs) including antihypertensives and anti-allergics, starting from the readily available raw material catechol. Unlike traditional methods that rely on the methylation of expensive vanillin, this novel approach utilizes a sequential methylation and formylation strategy that fundamentally alters the economic landscape of production. By shifting the feedstock to catechol, manufacturers can bypass the high costs associated with vanillin while simultaneously achieving superior yield metrics and process stability. The patent details a comprehensive utilization of methylating and formylating reagents, ensuring that the operational path is not only shortened but also optimized for industrial throughput. This technological breakthrough represents a significant pivot in how high-value aromatic aldehydes are manufactured, offering a reliable veratraldehyde supplier pathway that is both economically and technically superior to legacy processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of veratraldehyde has been heavily reliant on the O-methylation of vanillin, a process that, while chemically straightforward, suffers from severe economic and supply chain constraints. The primary bottleneck lies in the raw material itself; food-grade vanillin is a high-value commodity with a price point that significantly inflates the final cost of the synthetic intermediate, making it difficult to apply in cost-sensitive market sectors. Furthermore, conventional methylation protocols often struggle to consistently achieve yields beyond the 50% to 80% range, leading to substantial material loss and increased waste generation. The reliance on alkaline conditions for etherification can also introduce variability in product quality, necessitating rigorous downstream purification steps that further erode profit margins. These factors combined create a fragile supply chain where fluctuations in vanillin pricing directly impact the viability of veratraldehyde production, limiting the ability of manufacturers to offer competitive pricing for cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
In stark contrast, the innovative method described in the patent leverages catechol as the foundational building block, initiating a transformative two-stage reaction sequence that decouples production costs from the volatile vanillin market. The first stage involves a highly controlled methylation reaction where catechol is converted into veratrole (1,2-dimethoxybenzene) through a dual-temperature protocol that maximizes conversion efficiency. This is followed by a precise formylation step where veratrole reacts with specific formylating agents to yield the target aldehyde with exceptional purity. This route effectively shortens the operational complexity while enhancing the overall yield, as the intermediate veratrole can be produced with yields exceeding 90% and purity approaching 99.5%. By establishing a synthesis path that is independent of vanillin, this approach not only reduces the bill of materials cost but also stabilizes the entire process flow, ensuring smooth and consistent output that is essential for meeting the stringent demands of global pharmaceutical supply chains.
Mechanistic Insights into Composite Catalyst Methylation and Formylation
The core of this synthetic breakthrough lies in the intricate manipulation of phase transfer catalysis during the methylation stage, where a composite catalyst system plays a pivotal role in driving the reaction kinetics. The process employs a sophisticated blend of quaternary ammonium salts, such as benzyltriethylammonium chloride (TEBAC) or tetrabutylammonium hydrogen sulfate (TBAHS), often combined with organic bases like tetramethylammonium hydroxide (TMAH) or pyridine. These catalysts function by facilitating the transport of hydroxide ions into the organic phase, where they deprotonate the catechol to form a reactive phenoxide species capable of attacking the dimethyl sulfate. This mechanism effectively suppresses the hydrolysis rate of the methylating agent, a common side reaction that plagues traditional methods, thereby ensuring that the reaction proceeds steadily even at moderate temperatures ranging from 20°C to 105°C. The result is a highly efficient conversion where the formation of veratrole is maximized, and the presence of residual raw materials or by-products is minimized to negligible levels.
Following the successful isolation of veratrole, the process transitions to the formylation stage, which operates through an electrophilic aromatic substitution mechanism akin to the Vilsmeier-Haack reaction. In this step, a formylation reagent such as N-methylformanilide or dimethylformamide (DMF) reacts with phosphorus oxychloride to generate a highly reactive chloroiminium ion complex. This weak electrophilic reagent then attacks the electron-rich aromatic ring of the veratrole, preferentially targeting the position para to the methoxy groups due to electronic activation. The reaction is conducted at a controlled temperature of 50°C to 60°C to manage the exothermic nature of the complex formation and subsequent rearrangement. This precise thermal control, combined with the specific choice of reagents, ensures that the formyl group is introduced with high regioselectivity, ultimately yielding veratraldehyde with a content greater than 99.5% after simple rectification. 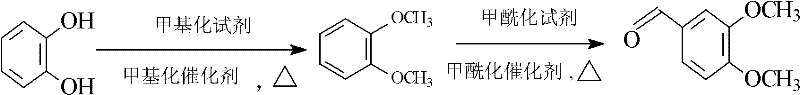
How to Synthesize Veratraldehyde Efficiently
Implementing this synthesis route requires careful attention to the sequential addition of reagents and strict temperature profiling to ensure the safety and efficacy of the transformation. The process begins with the dissolution of catechol in a suitable solvent like toluene, followed by the gradual introduction of dimethyl sulfate and the composite catalyst system under agitation. Operators must maintain the initial reaction temperature between 20°C and 40°C to control the exotherm during the formation of the methyl catechol intermediate before ramping up to 70°C and finally 105°C to drive the completion of veratrole synthesis. Once the veratrole is isolated and purified, it is subjected to the formylation conditions using phosphorus oxychloride and the chosen formyl donor, with the reaction mixture subsequently quenched in a toluene-water system for phase separation. The detailed standardized synthesis steps, including specific molar ratios, stirring speeds, and distillation parameters required for GMP-compliant manufacturing, are outlined in the guide below.
- Perform a two-stage methylation reaction on catechol using dimethyl sulfate and a composite phase-transfer catalyst system (e.g., TEBAC/TMAH) at controlled temperatures between 20°C and 105°C to generate veratrole.
- Conduct a formylation reaction on the isolated veratrole using a formylation reagent like N-methylformanilide or DMF in the presence of phosphorus oxychloride at 50-60°C.
- Quench the reaction mixture in a toluene-water system, separate the organic phase, and purify the crude product via rectification to obtain veratraldehyde with purity exceeding 99.5%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catechol-based synthesis route offers profound strategic benefits that extend far beyond simple chemical conversion metrics. By eliminating the dependency on vanillin, a specialty chemical with fluctuating market dynamics, manufacturers can secure a more stable and predictable cost structure for their veratraldehyde supply. The ability to source catechol, a bulk commodity chemical, significantly insulates the production budget from the volatility associated with flavor and fragrance grade raw materials. Furthermore, the high yield and purity achieved in the intermediate veratrole stage mean that less material is wasted, and downstream purification costs are drastically reduced, contributing to substantial cost savings in the overall manufacturing budget. This process optimization translates directly into a more competitive pricing model for the final API intermediate, allowing partners to maintain healthy margins even in price-sensitive markets.
- Cost Reduction in Manufacturing: The fundamental shift from vanillin to catechol as the starting material inherently lowers the raw material expenditure, as catechol is produced on a much larger industrial scale with lower unit costs. Additionally, the high efficiency of the composite catalyst system minimizes the consumption of methylating agents and reduces the generation of waste streams that require expensive treatment. The elimination of transition metal catalysts in favor of organic phase transfer catalysts also removes the need for costly heavy metal scavenging steps, further streamlining the production expense profile. These cumulative efficiencies ensure that the cost of goods sold (COGS) is significantly optimized without compromising on the quality or specification of the final product.
- Enhanced Supply Chain Reliability: Sourcing catechol is generally more reliable than sourcing high-purity vanillin, as it is a staple in the polymer and antioxidant industries with multiple global suppliers. This diversification of the supply base reduces the risk of single-source bottlenecks and ensures continuity of supply even during market disruptions. The robustness of the reaction conditions, which do not require high-pressure vessels or cryogenic cooling, also means that production can be scaled across a wider range of manufacturing facilities, increasing the overall resilience of the supply network. Consequently, lead times for high-purity pharmaceutical intermediates can be stabilized, providing downstream drug manufacturers with the certainty they need for their own production planning.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, operating under atmospheric pressure and moderate temperatures that are easily managed in standard glass-lined or stainless steel reactors. The absence of extreme conditions reduces energy consumption and lowers the safety risks associated with high-pressure operations, facilitating easier regulatory approval for commercial scale-up of complex pharmaceutical intermediates. Moreover, the ability to recover and recycle excess formylating reagents and solvents aligns with modern green chemistry principles, minimizing the environmental footprint and simplifying waste disposal compliance. This combination of operational safety and environmental stewardship makes the technology highly attractive for long-term sustainable manufacturing partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catechol-to-veratraldehyde synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear understanding of the method's capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers below reflect the specific advantages in yield, purity, and operational stability that distinguish this method from conventional vanillin-based approaches.
Q: What are the primary advantages of using catechol over vanillin for veratraldehyde production?
A: The primary advantage is significant cost reduction, as catechol is a widely available bulk commodity chemical compared to the more expensive food-grade vanillin traditionally used. Additionally, this route allows for the independent production and marketing of the intermediate veratrole, adding economic flexibility.
Q: How does the composite catalyst system improve the methylation yield?
A: The use of composite catalysts, such as mixtures of quaternary ammonium salts (TEBAC, TBAHS) and organic bases (TMAH, pyridine), effectively suppresses the hydrolysis of the methylating agent. This ensures a stable reaction environment, pushing the yield of veratrole above 90% while maintaining purity levels around 99.5%.
Q: Is this synthesis method suitable for large-scale industrial manufacturing?
A: Yes, the process is designed for industrial scalability as it operates under atmospheric pressure without requiring extreme vacuum or high-pressure conditions. The mild temperature ranges (20-105°C) and the ability to recover and recycle excess reagents make it highly suitable for continuous commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Veratraldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering high-purity veratraldehyde that meets stringent purity specifications, utilizing rigorous QC labs to verify every batch against the highest international standards. Our capability to implement the catechol-based synthesis route allows us to offer a product that is not only chemically superior but also economically advantageous for our partners.
We invite you to collaborate with us to explore how this optimized synthesis route can enhance your supply chain efficiency and reduce your overall manufacturing costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our veratraldehyde can serve as a reliable foundation for your next generation of therapeutic agents.
