Revolutionizing Methyl Heptenone Production with Advanced Rhodium Catalysis for Commercial Scale
Revolutionizing Methyl Heptenone Production with Advanced Rhodium Catalysis for Commercial Scale
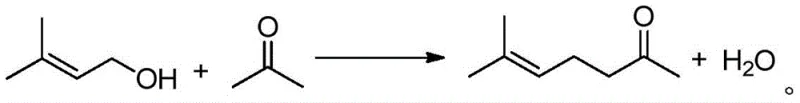
The global demand for high-purity methyl heptenone, a critical intermediate in the synthesis of vitamins A, E, and K1 as well as premium fragrances like linalool and citral, necessitates a paradigm shift in manufacturing technologies. Patent CN110845312A introduces a groundbreaking methodology that transcends the limitations of historical synthetic routes by employing a highly efficient rhodium-catalyzed selective dehydrogenation coupling reaction. This innovation leverages readily available 3-methyl-2-buten-1-ol and acetone as feedstocks, transforming them into valuable methyl heptenone in a single operational step. For R&D directors and procurement strategists, this patent represents a significant leap forward in atom economy and process safety, effectively addressing the long-standing challenges of waste generation and harsh reaction conditions that have plagued the industry for decades. The technical breakthrough lies not merely in the yield improvements but in the fundamental restructuring of the synthetic pathway to align with modern green chemistry principles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of methyl heptenone has relied on several established yet fundamentally flawed methodologies, each carrying substantial operational burdens and environmental liabilities. The earliest industrialized route, the acetylene-acetone method, involves the addition of acetylene to acetone followed by partial hydrogenation and a Carroll rearrangement. As illustrated in the reaction scheme below, this multi-step sequence is notoriously inefficient, requiring the consumption of large quantities of calcium carbide to generate acetylene, which inevitably produces massive amounts of solid carbide waste residue.

Furthermore, the Carroll rearrangement step generates stoichiometric amounts of ethanol by-products, severely compromising the atom economy of the entire process. Similarly, the isoprene method, which proceeds via hydrochlorination to isopentenyl chloride followed by condensation with acetone, suffers from poor selectivity and significant difficulties in catalyst recovery. The reliance on phase transfer catalysis in this route often leads to complex separation challenges and the generation of chlorinated organic waste, rendering it environmentally unsustainable and economically unviable for modern strict regulatory environments. These legacy processes collectively impose heavy costs on supply chain managers due to complex waste treatment requirements and low overall material throughput.
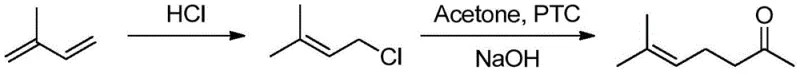
The Novel Approach
In stark contrast to these cumbersome legacy pathways, the novel approach detailed in the patent utilizes a direct, one-step coupling strategy that dramatically simplifies the manufacturing landscape. By bypassing the need for hazardous acetylene gas, corrosive hydrogen chloride, or unstable methoxypropene intermediates, this new method significantly reduces the inherent safety risks associated with raw material handling and storage. The transition from multi-step sequences involving isolation of unstable intermediates to a telescoped one-pot reaction minimizes equipment footprint and reduces the potential for yield loss during transfer operations. This streamlining of the synthetic route directly translates to enhanced process reliability and a substantial reduction in the complexity of downstream purification units, offering a compelling value proposition for facilities looking to optimize their capital expenditure and operational efficiency.
Mechanistic Insights into Rhodium-Catalyzed Selective Dehydrogenation Coupling
The core scientific innovation driving this process is the sophisticated interplay between the rhodium catalyst, the base promoter, and the substrate molecules, which orchestrates a precise sequence of dehydrogenation, condensation, and selective hydrogenation events. The mechanism initiates with the activation of 3-methyl-2-buten-1-ol by the rhodium center, facilitating a dehydrogenation event that generates the reactive 3-methyl-2-butenal intermediate alongside a rhodium-hydride species. This in-situ generated aldehyde then undergoes a base-catalyzed aldol condensation with acetone, forming 6-methyl-3,5-heptadien-2-one while releasing a molecule of water. The elegance of this system is further demonstrated in the final step, where the rhodium-hydride species selectively reduces the specific double bond at the 3-position of the diene intermediate.
This selectivity is critically governed by steric hindrance and can be finely tuned through the addition of organic base auxiliaries such as quinoline or isoquinoline. These additives serve a dual purpose: they modulate the hydrogenation activity of the rhodium-hydride species to prevent over-reduction of the desired product, and they synergistically enhance the rate of the aldol condensation step. For technical teams, understanding this mechanistic nuance is vital for optimizing reaction parameters, as the precise balance between the inorganic base (like LiOH) and the organic auxiliary determines the final impurity profile. This level of control ensures that the resulting methyl heptenone meets stringent purity specifications required for high-end fragrance and pharmaceutical applications without the need for extensive chromatographic purification.
How to Synthesize Methyl Heptenone Efficiently
Implementing this advanced synthesis route requires careful attention to catalyst loading, atmospheric control, and thermal management to maximize the benefits of the rhodium-mediated transformation. The process is designed to be robust, utilizing standard high-pressure reactor equipment that is commonly available in fine chemical manufacturing facilities, thereby lowering the barrier to adoption for existing production sites. Operators must ensure strict exclusion of oxygen and moisture during the catalyst preparation phase to maintain the activity of the sensitive rhodium complexes, typically handled within an inert glove box environment before charging the autoclave. The following guide outlines the standardized operational framework derived from the patent examples, providing a clear roadmap for scaling this technology from laboratory validation to pilot plant execution.
- Prepare the reaction system by loading anhydrous lithium hydroxide into a high-pressure autoclave and purging with nitrogen to ensure an inert atmosphere.
- In a glove box, mix the rhodium catalyst precursor, 3-methyl-2-buten-1-ol, and acetone, then transfer the mixture to the autoclave under nitrogen protection.
- Heat the reaction mixture to 60-120°C under 0.5-1.0 MPa pressure for 3-6 hours, followed by distillation to recover acetone and isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this rhodium-catalyzed technology offers transformative benefits that extend far beyond simple yield improvements, fundamentally altering the cost structure and risk profile of methyl heptenone sourcing. The elimination of hazardous and difficult-to-handle raw materials like acetylene gas and calcium carbide removes significant regulatory compliance burdens and safety insurance costs from the operational ledger. Furthermore, the drastic reduction in three wastes (waste water, waste gas, and solid residue) simplifies environmental permitting and lowers the ongoing expenses associated with waste treatment and disposal, contributing to a more sustainable and socially responsible supply chain. These factors collectively enhance the long-term viability of the production site, ensuring continuity of supply even as environmental regulations become increasingly stringent globally.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the consolidation of multiple reaction steps into a single unit operation, which significantly lowers energy consumption and labor requirements. By removing the need for expensive and complex purification steps associated with removing ethanol by-products or chlorinated impurities found in older methods, the overall processing cost is substantially decreased. Additionally, the high atom utilization rate means that a greater proportion of the purchased raw materials ends up in the final saleable product, minimizing raw material waste and maximizing the return on investment for every kilogram of feedstock procured.
- Enhanced Supply Chain Reliability: Sourcing stability is greatly improved by relying on commodity chemicals like acetone and 3-methyl-2-buten-1-ol, which are produced on a massive global scale with stable pricing dynamics, unlike niche intermediates required by the Saucy-Marbet or isoprene routes. The mild reaction conditions (60°C-120°C) reduce the thermal stress on equipment, leading to longer reactor lifecycles and fewer unplanned maintenance shutdowns that could disrupt delivery schedules. This robustness ensures that suppliers can maintain consistent output levels, providing downstream customers with the predictability they need for their own production planning and inventory management.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively with standard autoclave configurations that can be easily multiplied for capacity expansion without requiring exotic high-pressure infrastructure. The minimal generation of hazardous by-products aligns perfectly with modern green chemistry mandates, allowing manufacturers to market their products as sustainably produced, which is an increasingly valuable attribute in the European and North American markets. This alignment with environmental standards future-proofs the supply chain against potential regulatory bans on older, dirtier technologies, securing the long-term availability of this critical intermediate.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology, drawing directly from the experimental data and comparative analysis provided in the patent documentation. These insights are intended to clarify the operational feasibility and strategic advantages for stakeholders evaluating this method for potential integration into their manufacturing portfolios. Understanding these details is crucial for making informed decisions about technology licensing or supplier qualification in the competitive landscape of fine chemical intermediates.
Q: What are the primary advantages of this rhodium-catalyzed method over the traditional acetylene-acetone route?
A: Unlike the traditional acetylene-acetone method which generates significant calcium carbide waste and requires harsh Carroll rearrangement conditions, this novel approach utilizes a one-step selective dehydrogenation coupling. It offers superior atom economy, operates under much milder temperatures and pressures, and achieves conversion rates exceeding 99% with selectivity up to 99.73%, drastically reducing downstream purification costs.
Q: How does the catalyst system ensure high selectivity for methyl heptenone?
A: The system employs a specific rhodium complex, such as [Rh(COD)2]OTf, in conjunction with a base like lithium hydroxide. The mechanism involves the selective activation of the alcohol to an aldehyde, followed by aldol condensation. Crucially, the steric hindrance and optional organic base additives (like quinoline) modulate the [RhH2] species to selectively hydrogenate only the specific double bond required, preventing over-reduction and ensuring high purity of the final enone product.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the process is explicitly designed for scalability. It eliminates the need for hazardous acetylene gas handling and complex multi-step sequences found in older methods like the isoprene or Saucy-Marbet routes. The use of commercially available raw materials (3-methyl-2-buten-1-ol and acetone) and the robustness of the rhodium catalyst system under moderate pressure conditions make it highly viable for commercial scale-up of complex fragrance intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Methyl Heptenone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced catalytic processes like the one described in CN110845312A requires a partner with deep technical expertise and proven scale-up capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this rhodium-catalyzed route are fully realized in a commercial setting. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of methyl heptenone meets the exacting standards required for vitamin and fragrance synthesis. Our commitment to quality assurance means that clients can rely on us for consistent product performance, batch after batch.
We invite global partners to engage with our technical procurement team to discuss how this innovative manufacturing route can be tailored to your specific volume requirements and quality targets. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic advantages specific to your supply chain context. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to evaluate the tangible benefits of partnering with a leader in next-generation chemical synthesis.
