Advancing Asymmetric Synthesis: Enamine-Based Pd-Catalyzed Allylic Alkylation for Commercial Scale-up
The chemical industry is constantly evolving towards safer, more efficient synthetic methodologies, and patent CN101139270B represents a significant breakthrough in the realm of asymmetric synthesis. This specific intellectual property outlines a novel method for utilizing enamines as nucleophiles in palladium-catalyzed asymmetric allylic alkylation reactions, effectively addressing long-standing challenges associated with traditional ketone functionalization. By shifting away from the reliance on unstable enolates generated by strong bases, this technology offers a pathway to high-value chiral intermediates with exceptional stereocontrol and operational simplicity. For R&D directors and process chemists, the implications are profound, as it replaces hazardous, cryogenic-dependent protocols with a robust room-temperature procedure that maintains high enantioselectivity. This report analyzes the technical merits of this innovation, demonstrating how it serves as a reliable pharmaceutical intermediates supplier solution for complex molecule construction.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the asymmetric allylic substitution of simple ketones has been plagued by significant operational hurdles and safety concerns inherent to the generation of reactive nucleophiles. Conventional literature, such as the work by Braun et al., typically necessitates the use of extremely strong bases like butyllithium or lithium diisopropylamide (LDA) to deprotonate ketones into their corresponding enolates. These reagents are not only prohibitively expensive but also pose severe safety risks due to their pyrophoric nature and sensitivity to moisture and air. Furthermore, the resulting enol ether intermediates are notoriously unstable, requiring严苛 conditions for their formation and often needing to be prepared immediately prior to use to prevent decomposition. This instability forces manufacturing processes into complex, multi-step sequences involving cryogenic temperatures to maintain integrity, which drastically increases energy consumption and equipment costs while limiting the feasibility of cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
In stark contrast, the methodology described in CN101139270B introduces enamines as a superior class of nucleophiles that circumvent the instability issues of traditional enolates. By pre-forming the enamine through the condensation of a simple ketone with a secondary amine in the presence of a dehydrating agent, the process generates a stable, isolable species that can be handled safely without inert atmosphere gloveboxes or extreme cooling. This novel approach allows the subsequent palladium-catalyzed coupling to proceed under mild conditions, often at room temperature, with reaction times as short as 1 hour. The elimination of strong bases and the stabilization of the nucleophilic species streamline the entire workflow, transforming a hazardous, multi-variable operation into a straightforward addition sequence. This shift not only enhances safety profiles but also opens the door for substantial cost savings by reducing the need for specialized cryogenic reactors and expensive stoichiometric bases.
Mechanistic Insights into Enamine-Mediated Pd-Catalyzed Alkylation
The core of this technological advancement lies in the unique reactivity profile of enamines within the palladium catalytic cycle. Unlike hard enolate nucleophiles that require strict counter-ion management, enamines act as soft nucleophiles that interact efficiently with the pi-allyl palladium complex formed from the allylic acetate substrate. The reaction initiates with the oxidative addition of the Pd(0) species to the allylic acetate, generating a cationic pi-allyl palladium intermediate coordinated by the chiral ligand. The enamine then attacks this electrophilic complex in a highly stereocontrolled manner, dictated by the chiral environment of the ligand, leading to the formation of the C-C bond with high fidelity. Following the nucleophilic attack, a simple hydrolysis step using cold saturated ammonium chloride cleaves the enamine moiety to reveal the desired chiral ketone product, regenerating the secondary amine which can potentially be recycled. This mechanism ensures that the stereochemical information encoded in the chiral ligand is effectively transferred to the product, achieving enantiomeric excess values up to 99% ee under optimized conditions.
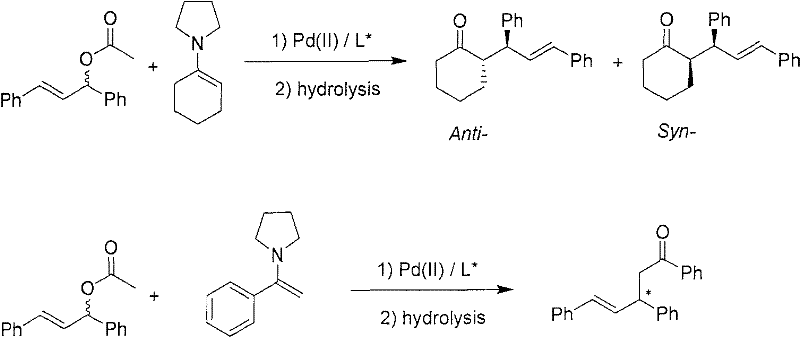
Controlling the impurity profile is another critical aspect where this mechanism excels, particularly regarding the ratio of anti to syn diastereomers. The patent data indicates that by tuning the solvent system—switching from dichloromethane to toluene or THF—and adjusting the temperature, the diastereoselectivity can be modulated effectively. For instance, running the reaction in THF at -15°C yielded an anti/syn ratio of 61/39 with exceptional optical purity, whereas room temperature reactions in DCM showed different selectivity profiles. This tunability allows process chemists to optimize for the specific isomer required for downstream API synthesis, minimizing the burden on purification units. The hydrolysis step is equally crucial, as it must be performed carefully to avoid racemization of the newly formed stereocenter, ensuring that the high-purity pharmaceutical intermediates produced meet the stringent quality standards required for clinical applications.
How to Synthesize Chiral Ketones Efficiently
Implementing this synthesis route requires a disciplined approach to reagent preparation and reaction monitoring to ensure consistent high-quality output. The process begins with the distinct preparation of the enamine nucleophile, where a simple ketone is condensed with a secondary amine like pyrrolidine using a dehydrating agent such as magnesium sulfate or titanium tetrachloride. Once the stable enamine is secured, the catalytic system is assembled by dissolving a palladium precursor and the chosen chiral ligand in a dry solvent, allowing the active species to form before introducing the electrophile. The detailed standardized synthesis steps below outline the precise molar ratios, addition sequences, and workup procedures necessary to replicate the high yields and selectivity reported in the patent documentation.
- Prepare the enamine nucleophile by reacting a simple ketone with a secondary amine (e.g., pyrrolidine) and a dehydrating agent like magnesium sulfate in a non-polar solvent.
- Generate the active palladium catalyst in situ by mixing a Pd(II) source with a chiral ligand in a solvent such as dichloromethane or toluene at room temperature.
- Add the substituted allyl acetate substrate followed by the prepared enamine, stir for approximately 1 hour, and quench with cold saturated ammonium chloride solution to hydrolyze the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this enamine-based methodology offers tangible strategic benefits that extend beyond mere technical elegance. The primary advantage lies in the drastic simplification of the raw material portfolio, as the process eliminates the need for hazardous, high-cost organometallic bases that often suffer from volatile pricing and strict transportation regulations. By replacing these dangerous reagents with stable amines and common dehydrating agents, the supply chain becomes more resilient and less prone to disruptions caused by regulatory changes or supplier shortages. Furthermore, the ability to conduct reactions at or near room temperature significantly reduces the energy load on manufacturing facilities, removing the dependency on large-scale cryogenic cooling systems that are capital-intensive to install and maintain. This operational flexibility translates directly into improved margin potential and a more predictable production schedule.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the removal of expensive stoichiometric bases and the reduction of energy consumption associated with cryogenic processing. Traditional methods requiring butyllithium or LDA incur high costs not only for the reagents themselves but also for the specialized containment and quenching systems needed to handle them safely. By utilizing enamines, the process operates under much milder thermal conditions, often completing within 1 hour at room temperature, which maximizes reactor throughput and minimizes utility costs. Additionally, the simplified workup procedure using aqueous ammonium chloride reduces the volume of organic waste generated, lowering disposal fees and environmental compliance costs significantly.
- Enhanced Supply Chain Reliability: From a logistics perspective, the shift to stable enamine nucleophiles mitigates the risks associated with transporting and storing pyrophoric materials. Strong bases like LDA often have limited shelf lives and require dedicated storage infrastructure, creating bottlenecks in the supply chain. In contrast, the secondary amines and ketones used to generate enamines are commodity chemicals with robust global supply networks, ensuring continuous availability even during market fluctuations. This stability allows for better inventory planning and reduces the lead time for high-purity pharmaceutical intermediates, enabling faster response to customer demand without compromising on safety or quality standards.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety risks, but this methodology inherently de-risks the scale-up phase by avoiding exothermic hazards associated with strong base additions. The mild reaction conditions facilitate a smoother transition from laboratory benchtop to pilot plant and finally to commercial production, as heat transfer and mixing requirements are less demanding than those for cryogenic reactions. Moreover, the reduced use of hazardous reagents aligns with green chemistry principles, simplifying the permitting process for new manufacturing lines and enhancing the company's sustainability profile. This environmental compliance is increasingly becoming a key differentiator in securing contracts with major multinational corporations that prioritize responsible sourcing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this enamine-based palladium catalysis technology. These insights are derived directly from the experimental data and comparative analysis found within the patent specification, providing clarity on performance metrics and operational parameters. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why are enamines preferred over traditional enolates in this Pd-catalyzed reaction?
A: Enamines serve as stable, convenient nucleophiles that eliminate the need for hazardous strong bases like butyllithium or LDA. This avoids the formation of unstable enol ether intermediates, significantly improving operational safety and simplifying the workflow by removing the requirement for cryogenic conditions and immediate usage.
Q: What level of stereoselectivity can be achieved with this method?
A: The patent data demonstrates that this method achieves exceptional stereoselectivity, with enantiomeric excess (ee) values reaching up to 99% under optimized conditions (e.g., in THF at -15°C). This high level of control is critical for producing high-purity pharmaceutical intermediates.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process is highly scalable due to its mild reaction conditions, primarily operating at room temperature with short reaction times (around 1 hour). The elimination of pyrophoric reagents and the use of standard workup procedures like ammonium chloride hydrolysis make it robust for commercial production environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Ketones Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the enamine-mediated allylic alkylation described in CN101139270B. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. We understand that for R&D directors, consistency is key, and our process engineering teams are dedicated to optimizing these chiral syntheses to deliver high-purity pharmaceutical intermediates that meet the exacting standards of the global market.
We invite you to collaborate with us to leverage this cutting-edge chemistry for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this safer, more efficient route can improve your bottom line. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage through superior chemical manufacturing solutions.
