Scalable Microbial Production of Valienamine and Validamine for Pharmaceutical Intermediates
Scalable Microbial Production of Valienamine and Validamine for Pharmaceutical Intermediates
The pharmaceutical and agrochemical industries are constantly seeking more efficient, sustainable, and cost-effective routes to produce high-value cyclic alcohol intermediates such as valienamine and validamine. Patent CN1563397A introduces a groundbreaking microbial preparation method utilizing a novel strain of Stenotrophomonas maltophilia (CCTCC No.M 204024) to decompose validamycin or valienolamine into these critical compounds. This biological approach represents a significant departure from traditional chemical synthesis or less efficient microbial degradation methods, offering a robust pathway for the commercial scale-up of complex pharmaceutical intermediates. By leveraging the unique metabolic capabilities of this specific bacterial strain, manufacturers can achieve high decomposition rates and molar yields, addressing the growing global demand for glycosidase inhibitors and their precursors. The technology outlined in this patent provides a foundational blueprint for establishing a reliable supply chain for these essential building blocks, ensuring consistency and quality in downstream drug manufacturing processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of valienamine and validamine has relied on various microbial strains, including Pseudomonas denitrificans, Flavobacterium saccharophilum, and Agrobacterium radiobacter, yet these methods have been plagued by significant inefficiencies and operational constraints. Many of these conventional strains exhibit a very weak capacity for decomposing validamycin, often failing to grow effectively when validamycin is the sole carbon source, which severely limits substrate loading and overall productivity. Furthermore, the metabolic pathways in these older strains often result in incomplete degradation or the accumulation of unwanted byproducts, necessitating complex and costly downstream purification steps to isolate the target amines. The reliance on multiple bacterial species for different stages of degradation also complicates the fermentation process control, leading to batch-to-batch variability that is unacceptable for GMP-compliant pharmaceutical manufacturing. Consequently, the industry has faced challenges in securing a consistent, high-volume supply of these intermediates, driving up costs and creating bottlenecks in the production of final active pharmaceutical ingredients.
The Novel Approach
In stark contrast to previous methodologies, the utilization of Stenotrophomonas maltophilia CCTCC No.M 204024 offers a streamlined and highly efficient solution for the bioconversion of validamycin. This novel strain possesses a superior enzymatic system capable of rapidly hydrolyzing the glycosidic linkages in validamycin, allowing it to thrive even when validamycin is the exclusive carbon source in the fermentation medium. The patent data indicates that this strain can achieve validamycin decomposition rates ranging from 70% to 90%, with molar yields of valienamine reaching impressive levels of 40% to 80% under optimized conditions. This high efficiency translates directly into reduced raw material waste and higher throughput per fermentation batch, fundamentally altering the economic viability of producing these cyclic alcohols. Moreover, the process flexibility allows for the use of either whole cells or extracted enzymes, providing manufacturers with the option to optimize for either continuous fermentation or batch enzymatic conversion depending on their specific facility capabilities and production goals.
Mechanistic Insights into Microbial Decomposition and Hydrolysis
The core of this innovative process lies in the specific biochemical interaction between the Stenotrophomonas maltophilia enzymes and the validamycin substrate, which facilitates the cleavage of glycosidic bonds to release valienamine and validamine. The fermentation environment is carefully controlled within a temperature range of 20°C to 40°C and a pH of 6.0 to 8.0, conditions that maximize the activity of the intracellular or extracellular lyases responsible for the degradation. Unlike harsh chemical hydrolysis which might require extreme pH or temperature and lead to racemization or degradation of the sensitive cyclohexene ring, this biological catalyst operates under mild physiological conditions, preserving the stereochemical integrity of the products. The metabolic pathway effectively converts the complex aminoglycoside structure of validamycin into the simpler, high-value cyclitols through a series of enzymatic steps that are highly specific, thereby minimizing the formation of structural isomers or impurities that are difficult to separate. This specificity is crucial for pharmaceutical applications where the optical purity of the intermediate dictates the efficacy and safety of the final drug product.
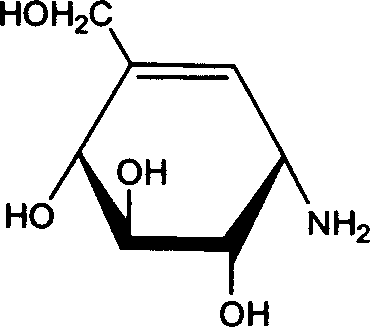
Understanding the molecular architecture of the target products is essential for optimizing the purification strategy, as the presence of multiple hydroxyl and amino groups influences their solubility and ion-exchange behavior. Valienamine, characterized by its primary amino group, carbon-carbon double bond, and three hydroxyl groups, exhibits distinct physicochemical properties that allow for its separation from the fermentation broth using ion-exchange chromatography. The presence of the double bond in valienamine distinguishes it from validamine, which is fully saturated, enabling analysts to monitor the reaction progress and product ratio using techniques like thin-layer chromatography and gas phase analysis. The ability of the microorganism to produce both compounds simultaneously, yet in separable forms, simplifies the downstream processing workflow compared to synthetic routes that might require protecting group strategies. This mechanistic clarity ensures that process engineers can design robust isolation protocols that consistently deliver high-purity valienamine and validamine suitable for sensitive downstream coupling reactions.
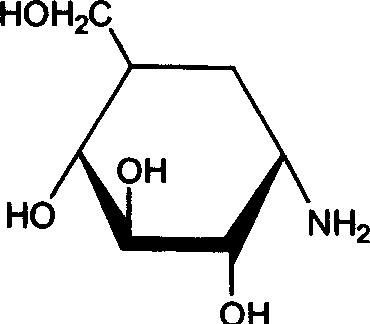
How to Synthesize Valienamine Efficiently
The synthesis of valienamine via this microbial route involves a carefully orchestrated sequence of medium preparation, inoculation, fermentation, and purification, all of which are detailed in the patent examples to ensure reproducibility. The process begins with the formulation of a nutrient-rich medium containing validamycin as the substrate, supplemented with inorganic salts like ammonium sulfate and phosphates to support robust bacterial growth and enzyme expression. Following sterilization, the medium is inoculated with the Stenotrophomonas maltophilia seed culture, and the fermentation is allowed to proceed under aerated and stirred conditions for a period ranging from 100 to 180 hours, depending on the specific substrate concentration and desired conversion. Detailed standardized synthesis steps see the guide below.
- Prepare the fermentation medium containing validamycin as the primary carbon source, along with ammonium sulfate, potassium chloride, and phosphate buffers, adjusting the initial pH to between 6.0 and 8.0.
- Inoculate the sterilized medium with Stenotrophomonas maltophilia (CCTCC No.M 204024) seed culture and maintain fermentation temperatures between 20°C and 40°C with adequate aeration and stirring.
- Harvest the fermentation broth after 100 to 180 hours, separate the biomass via centrifugation, and purify the supernatant using ion exchange resin and chromatographic techniques to isolate the target amines.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this microbial fermentation technology offers substantial strategic benefits, primarily driven by the simplification of the manufacturing process and the use of renewable biological resources. By shifting away from multi-step chemical synthesis which often relies on expensive reagents and generates significant hazardous waste, this bio-based route aligns with modern green chemistry principles and reduces the environmental compliance burden on the manufacturing facility. The ability to use validamycin, a widely available agricultural antibiotic, as a starting material ensures a stable and cost-effective raw material supply, mitigating the risks associated with volatile petrochemical feedstock markets. Furthermore, the high decomposition efficiency of the new strain means that less substrate is required to produce the same amount of product, directly contributing to cost reduction in pharmaceutical intermediate manufacturing through improved material utilization rates. These factors combined create a more resilient supply chain capable of meeting fluctuating market demands without compromising on quality or delivery timelines.
- Cost Reduction in Manufacturing: The elimination of complex chemical synthesis steps and the use of a highly efficient biological catalyst significantly lower the operational expenditures associated with producing valienamine and validamine. Since the process avoids the need for expensive transition metal catalysts or harsh organic solvents, the costs related to reagent procurement, solvent recovery, and hazardous waste disposal are drastically reduced. The high molar yields reported in the patent examples suggest that the process is materially efficient, meaning that a greater proportion of the input validamycin is converted into saleable product rather than lost as byproducts. This efficiency translates into a lower cost of goods sold (COGS), allowing suppliers to offer more competitive pricing to downstream pharmaceutical clients while maintaining healthy profit margins. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, further enhancing the overall economic attractiveness of the method.
- Enhanced Supply Chain Reliability: Utilizing a robust microbial strain that can grow on validamycin as a sole carbon source simplifies the upstream supply requirements, as validamycin is a mature, commercially available commodity with a stable production history. The fermentation process is inherently scalable, allowing manufacturers to increase production capacity by simply increasing fermenter volume or running parallel batches, ensuring that supply can be ramped up quickly to meet urgent client needs. This scalability reduces the lead time for high-purity pharmaceutical intermediates, as the biological conversion is faster and more direct than traditional multi-step organic synthesis which often suffers from bottleneck steps. The consistency of the biological system, once optimized, ensures batch-to-batch reproducibility, which is critical for maintaining long-term contracts with major pharmaceutical companies that require strict adherence to quality specifications.
- Scalability and Environmental Compliance: The fermentation process described is well-suited for large-scale industrial application, utilizing standard stirred-tank reactors and aeration systems that are common in modern biopharmaceutical facilities. Because the process is bio-based, the waste stream is primarily organic and biodegradable, significantly easing the burden on wastewater treatment systems and reducing the environmental footprint of the manufacturing site. This alignment with environmental, social, and governance (ESG) goals is increasingly important for multinational corporations seeking sustainable suppliers, potentially opening up new market opportunities for producers of these intermediates. The straightforward downstream processing involving ion exchange and chromatography is also easily adaptable to large-scale preparative columns, ensuring that the purification stage does not become a bottleneck as production volumes increase.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the microbial production of valienamine and validamine, based on the specific data and embodiments provided in the patent literature. These answers are designed to clarify the feasibility of the process for potential partners and to highlight the key differentiators of this technology compared to existing methods. Understanding these details is crucial for making informed decisions about integrating this supply route into your broader procurement strategy.
Q: What is the primary advantage of using Stenotrophomonas maltophilia for valienamine production?
A: The strain CCTCC No.M 204024 exhibits a significantly higher decomposition capacity for validamycin compared to conventional strains like Pseudomonas or Flavobacterium, allowing it to grow on validamycin as a sole carbon source with high conversion rates.
Q: What are the typical purification methods for the fermentation broth?
A: The process utilizes a combination of macro-porous weakly acidic resin adsorption followed by elution with ammoniacal liquor, and further refinement using strongly basic chromatographic resin to achieve high purity separation of valienamine and validamine.
Q: Can this process be scaled for industrial manufacturing?
A: Yes, the patent describes conditions suitable for deep ventilation and stirred tank fermentation, indicating strong potential for commercial scale-up from laboratory shake flasks to large-scale bioreactors without complex chemical synthesis steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Valienamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates like valienamine and validamine play in the development of next-generation glycosidase inhibitors and therapeutic agents. Our team of expert process chemists and fermentation specialists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising laboratory results of patent CN1563397A can be successfully translated into industrial reality. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which employ advanced analytical techniques to verify the identity and potency of every batch. By partnering with us, you gain access to a supply chain that is not only technically robust but also deeply aligned with your requirements for reliability, quality, and regulatory compliance.
We invite you to engage with our technical procurement team to discuss how we can tailor this microbial fermentation process to your specific volume and quality needs. Whether you require a Customized Cost-Saving Analysis for your current supply chain or need to evaluate the feasibility of switching to this bio-based route, we are ready to provide the data and support you need. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can drive value and efficiency in your pharmaceutical manufacturing operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
