Advanced Synthesis of Iclaprim Intermediates for Commercial Scale-up
Introduction to Advanced Iclaprim Intermediate Manufacturing
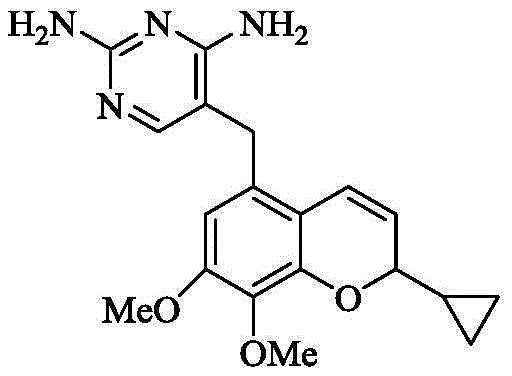
The pharmaceutical landscape for treating Acute Bacterial Skin and Skin Structure Infections (ABSSSI) has been significantly advanced by the development of dihydrofolate reductase inhibitors, specifically Iclaprim. As detailed in patent CN110724108B, a novel preparation method for critical phenol compounds and acetyl intermediates addresses longstanding manufacturing bottlenecks inherent in earlier synthetic strategies. This technology enables the efficient synthesis of the core benzopyran scaffold required for this potent antibiotic candidate, which is currently under investigation for Hospital Acquired Bacterial Pneumonia. By optimizing reaction conditions and reagent selection, the disclosed process offers a viable pathway for industrial production that surpasses previous limitations in yield and purity standards. Understanding the structural nuances of the final active pharmaceutical ingredient is essential for appreciating the complexity of the intermediate synthesis required to build it effectively.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Conventional synthetic routes, such as those described in prior art CN 101115743, suffer from excessive step counts and reliance on purification techniques incompatible with large-scale manufacturing economics. The traditional approach involves protecting group manipulations, followed by Friedel-Crafts acetylation, aldol condensation, and multiple demethylation steps that cumulatively result in a dismal overall yield of approximately 4.84 percent. Furthermore, the necessity for column chromatography at multiple stages, particularly during the conversion of compound 9 to compound 13, creates a severe bottleneck for cost-effective production and waste management. These operational complexities not only inflate the cost of goods but also introduce significant variability in product quality, making regulatory approval for commercial supply chains increasingly difficult to secure for generic manufacturers.
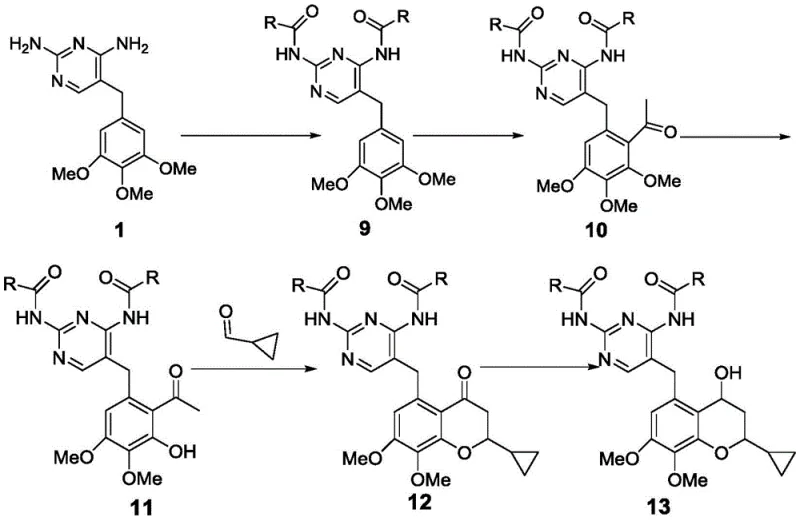
The Novel Approach
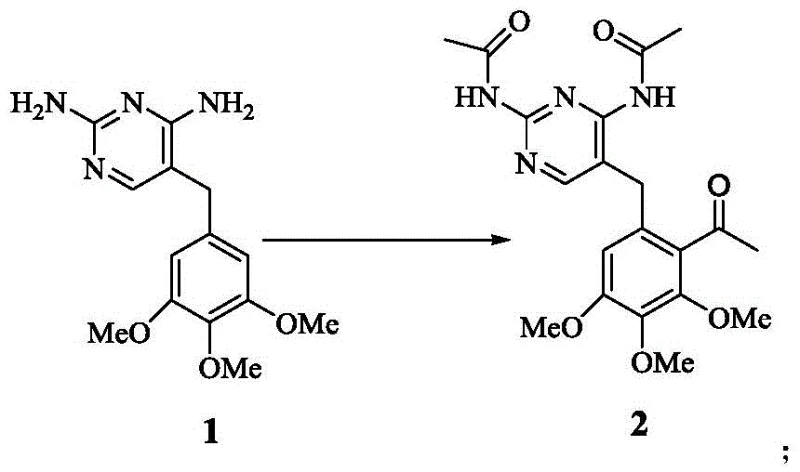
In stark contrast, the methodology presented in CN110724108B utilizes a streamlined strategy centered on direct Lewis acid-catalyzed acetylation and selective demethylation to bypass these inefficiencies. This approach eliminates the need for cumbersome protection-deprotection sequences and avoids the use of expensive transition metal catalysts like cerium chloride found in other prior art routes. The core innovation lies in the direct transformation of trimethoprim derivatives into acetylated intermediates using tin tetrachloride, achieving high regioselectivity without extensive purification requirements. Subsequent selective demethylation using boron tribromide or boron trichloride allows for precise control over the substitution pattern on the aromatic ring, ensuring the correct positioning of hydroxyl groups for downstream cyclization.
Mechanistic Insights into Lewis Acid Catalyzed Functionalization
The mechanistic underpinning of this synthesis relies heavily on the coordination chemistry of Lewis acids with electron-rich aromatic systems and ether functionalities to drive regioselective transformations. During the acetylation phase, tin tetrachloride acts as a potent electrophile activator, facilitating the attack of the acetylating agent on the specific position of the benzyl-pyrimidine scaffold with high efficiency. This catalytic cycle ensures that the acetyl group is introduced ortho to the methoxy groups with high fidelity, minimizing the formation of regioisomeric impurities that are notoriously difficult to separate on a large scale. Following this, the selective demethylation proceeds through the formation of a stable boron-ether complex, which upon hydrolysis yields the desired phenol while leaving other methoxy groups intact. This level of chemoselectivity is critical for maintaining the integrity of the pyrimidine diamine moiety throughout the synthesis, preventing degradation of the sensitive amine groups.
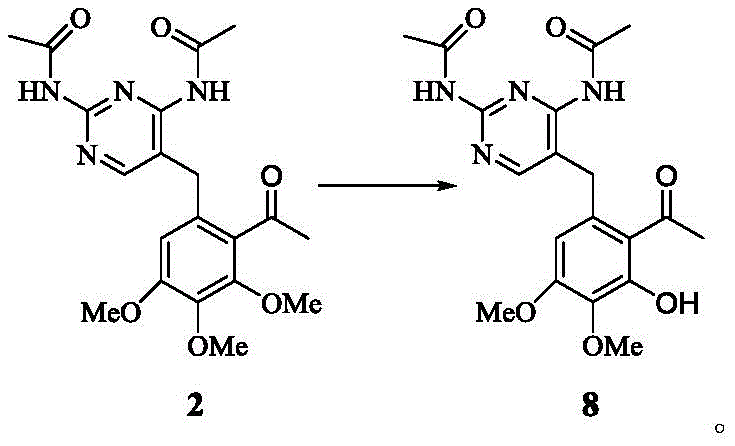
How to Synthesize Phenol Compound Formula 8 Efficiently
The synthesis of the target phenol compound involves a robust two-stage process beginning with acetylation followed by selective demethylation under strictly controlled thermal conditions. Operators must ensure precise stoichiometry of the Lewis acid catalysts to prevent over-reaction or decomposition of the sensitive pyrimidine ring system. The detailed standardized synthesis steps, including specific solvent volumes, temperature ramps, and workup procedures, are outlined in the guide below to ensure reproducibility and safety.
- Perform acetylation on trimethoprim or acetamide derivatives using tin tetrachloride and acetic anhydride in chloroform to obtain the acetyl compound.
- Conduct selective demethylation of the acetyl compound using boron tribromide or boron trichloride in dichloromethane at controlled low temperatures.
- Execute post-treatment involving extraction, pH adjustment, and recrystallization to isolate the high-purity phenol compound.
Commercial Advantages for Procurement and Supply Chain Teams
This patented methodology offers substantial strategic benefits for procurement teams looking to optimize the cost structure of antibiotic intermediate manufacturing without compromising quality. By eliminating the need for column chromatography and reducing the total number of synthetic steps, the process drastically simplifies the production workflow and reduces the consumption of silica gel and organic solvents. The use of commercially available and economical reagents such as tin tetrachloride and acetic anhydride further contributes to a lower raw material cost base compared to routes requiring specialized catalysts.
- Cost Reduction in Manufacturing: The elimination of chromatographic purification steps represents a major driver for cost savings, as it removes the need for expensive stationary phases and reduces solvent recovery loads significantly. Additionally, the higher yields achieved in each step mean that less starting material is required to produce the same amount of final product, directly improving the material efficiency of the plant. The simplified post-treatment involving crystallization rather than complex separations allows for faster batch turnover times and reduced labor costs associated with purification.
- Enhanced Supply Chain Reliability: Relying on widely available commodity chemicals like acetic anhydride and common Lewis acids ensures that the supply chain is not vulnerable to shortages of exotic or single-source reagents. The robustness of the reaction conditions, which tolerate standard industrial solvents like dichloromethane and chloroform, means that production can be easily transferred between different manufacturing sites without requalification issues. This flexibility is crucial for maintaining continuous supply in the event of regional disruptions or equipment maintenance at a primary facility.
- Scalability and Environmental Compliance: The shift from chromatography to crystallization-based purification makes the process inherently more scalable, allowing for seamless transition from pilot plant to multi-ton commercial production. Reduced solvent usage and the avoidance of heavy metal catalysts simplify waste stream treatment, helping facilities meet increasingly stringent environmental regulations regarding effluent discharge. The ability to recycle solvents more effectively due to fewer contaminants further enhances the environmental profile and sustainability metrics of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route in an industrial setting. These answers are derived directly from the experimental data and claims within the patent documentation to provide accurate guidance for technical teams. Understanding these details is vital for assessing the feasibility of adopting this technology for your specific production needs.
Q: What are the primary advantages of the new synthesis route over conventional methods?
A: The new route eliminates the need for column chromatography, significantly reduces step count, and uses more economical reagents compared to prior art methods which suffered from low yields around 4.84 percent.
Q: Which Lewis acids are preferred for the selective demethylation step?
A: Boron tribromide and boron trichloride are the preferred Lewis acids, typically used as solutions in methylene chloride to ensure precise control over the reaction temperature and selectivity.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for industrial suitability by replacing complex purification steps with simple crystallization and extraction, thereby enhancing scalability and supply chain reliability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Iclaprim Intermediate Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, leveraging deep expertise in complex pharmaceutical intermediate manufacturing to bring innovative patents like CN110724108B to life. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to reactor is seamless and efficient. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of Iclaprim intermediate meets the exacting standards required for global regulatory submissions.
We invite you to contact our technical procurement team to discuss how we can tailor this synthesis route to your specific volume and cost requirements. Request a Customized Cost-Saving Analysis today to understand the potential economic impact of switching to this optimized process. Our experts are ready to provide specific COA data and route feasibility assessments to support your supply chain strategy.
