Scalable Synthesis of Iclaprim Intermediate via Acetic Acid-Mediated Reduction
The pharmaceutical landscape is constantly evolving, driven by the urgent need for efficient and cost-effective synthesis routes for critical active pharmaceutical ingredients (APIs). A prime example of this innovation is found in Chinese Patent CN110498812B, which details a groundbreaking preparation method for a key intermediate compound of Iclaprim, a dihydrofolate reductase inhibitor developed for treating acute bacterial skin and skin tissue infections. This patent addresses a significant bottleneck in the existing supply chain by introducing a novel reduction strategy that replaces expensive and difficult-to-recycle reagents with commonplace, economical alternatives. For R&D directors and procurement managers alike, understanding the nuances of this synthetic pathway is crucial for optimizing the production of high-purity pharmaceutical intermediates. The technology described offers a robust solution for the commercial scale-up of complex pharmaceutical intermediates, ensuring a steady supply of high-quality materials essential for downstream drug formulation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
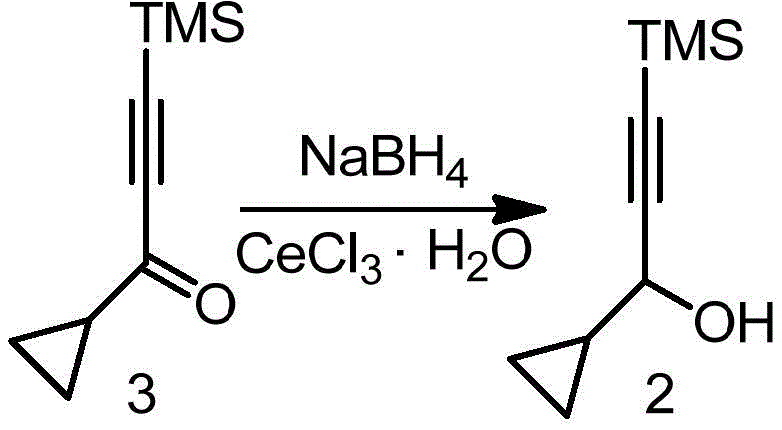
Prior to this innovation, the standard literature method for synthesizing the critical alcohol intermediate involved the use of cerium chloride heptahydrate as a Lewis acid promoter during the sodium borohydride reduction step. As illustrated in the reaction scheme below, this traditional approach, referenced in WO/1997020839, relies heavily on the presence of rare earth metal salts to facilitate the conversion of the ketone precursor to the desired alcohol. However, this dependency introduces severe logistical and economic drawbacks for industrial manufacturers. Cerium chloride heptahydrate is not only a costly reagent but also presents significant challenges in terms of waste management and recycling, as the metal salts are difficult to recover from the reaction mixture. Furthermore, the stoichiometric requirement for this heavy metal salt is substantial, often necessitating nearly three kilograms of cerium chloride for every kilogram of product produced, which drastically inflates the raw material costs and complicates the environmental compliance profile of the manufacturing process.
The Novel Approach
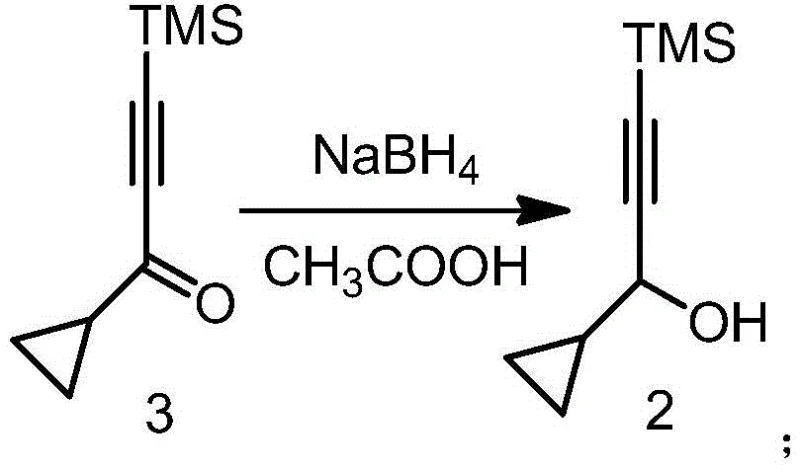
In stark contrast to the legacy methods, the novel approach disclosed in CN110498812B utilizes acetic acid as the promoting agent in conjunction with sodium borohydride, effectively eliminating the need for expensive cerium salts. This methodological shift represents a paradigm change in cost reduction in pharmaceutical intermediate manufacturing, as acetic acid is a ubiquitous, low-cost commodity chemical that is easy to handle and dispose of. The reaction proceeds smoothly in common organic solvents such as tetrahydrofuran or methanol, maintaining high selectivity for the carbonyl group while preserving the sensitive alkyne functionality. By replacing the heavy metal promoter with a simple carboxylic acid, the process not only achieves superior yields—often exceeding 80%—but also simplifies the downstream purification workflow. This streamlined protocol allows for the direct isolation of the product via vacuum distillation, bypassing the need for resource-intensive chromatographic separation techniques that are often required when metal contaminants are present.
Mechanistic Insights into Acetic Acid-Mediated Ketone Reduction
The core of this technological advancement lies in the precise modulation of the reducing environment through the addition of acetic acid. In a standard sodium borohydride reduction, the hydride ion acts as a nucleophile attacking the electrophilic carbon of the carbonyl group. However, in the presence of acetic acid, the reaction dynamics are subtly altered to enhance efficiency without compromising chemoselectivity. The acetic acid likely serves to activate the carbonyl oxygen through hydrogen bonding or transient protonation, thereby increasing the electrophilicity of the carbonyl carbon and facilitating a more rapid attack by the borohydride species. Crucially, the conditions are controlled—typically at mild temperatures between 10°C and 25°C—to ensure that the reduction is selective for the ketone moiety. This selectivity is paramount because the substrate contains a trimethylsilyl-protected alkyne group, which must remain intact for subsequent coupling reactions in the total synthesis of Iclaprim. The avoidance of harsh Lewis acids prevents potential side reactions such as desilylation or alkyne hydration, which are common pitfalls in less optimized protocols.
Furthermore, the mechanistic pathway ensures a clean impurity profile, which is a critical concern for R&D directors focused on regulatory compliance. The use of acetic acid generates benign byproducts, primarily sodium acetate and boron species that are easily removed during the aqueous workup. Unlike the cerium-mediated process, which can leave trace metal residues requiring rigorous scrubbing to meet ppm-level specifications, this acid-mediated reduction results in an organic phase that is relatively free of inorganic contaminants. The post-treatment involves a straightforward quench with dilute hydrochloric acid, followed by extraction and washing, which effectively partitions the product into the organic layer while leaving water-soluble salts behind. This inherent cleanliness of the reaction mechanism translates directly into higher overall process efficiency and reduced risk of batch failure due to out-of-specification impurity levels.
How to Synthesize 1-Cyclopropyl-3-(trimethylsilyl)prop-2-yn-1-ol Efficiently
Implementing this synthesis route requires careful attention to reagent ratios and temperature control to maximize yield and purity. The process begins by dissolving the ketone starting material and acetic acid in a suitable solvent like tetrahydrofuran, followed by the controlled addition of sodium borohydride. The reaction is exothermic, so maintaining the temperature within the optimal range of 0°C to 50°C is essential to prevent thermal runaway while ensuring complete conversion. Detailed operational parameters, including specific molar ratios and workup procedures, are critical for reproducibility on a manufacturing scale. For a comprehensive, step-by-step guide on executing this transformation with precision, please refer to the standardized protocol outlined below.
- Dissolve intermediate 3 (1-cyclopropyl-3-(trimethylsilyl)prop-2-yn-1-one) and acetic acid in an organic solvent such as tetrahydrofuran.
- Add sodium borohydride to the mixture in portions while stirring at a temperature between 0°C and 50°C.
- Quench the reaction with hydrochloric acid, extract with petroleum ether, and purify via vacuum distillation to obtain the alcohol intermediate with >98% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this acetic acid-mediated reduction process offers tangible strategic benefits that extend beyond simple chemistry. The primary advantage is the drastic simplification of the supply chain for raw materials; by removing the dependency on cerium chloride heptahydrate, manufacturers eliminate exposure to the price volatility and supply constraints often associated with rare earth elements. Acetic acid and sodium borohydride are globally available commodity chemicals with stable pricing and robust supply networks, ensuring continuity of supply even during market disruptions. This shift significantly de-risks the production schedule for the Iclaprim intermediate, allowing for more reliable long-term planning and inventory management without the fear of sudden raw material shortages.
- Cost Reduction in Manufacturing: The economic impact of switching to this new method is profound, driven principally by the replacement of a high-cost reagent with a low-cost alternative. In the conventional process, the mass ratio of cerium chloride to product is nearly 3:1, meaning a significant portion of the raw material budget is spent on a reagent that ends up as waste. In contrast, the new method utilizes acetic acid, which is orders of magnitude cheaper and required in much lower molar equivalents relative to the substrate. Additionally, the elimination of chromatographic purification steps reduces the consumption of silica gel and solvents, further driving down the variable costs per kilogram. These cumulative savings result in a substantially lower cost of goods sold (COGS), enhancing the overall profitability of the API manufacturing process.
- Enhanced Supply Chain Reliability: From a logistics perspective, the simplified reagent profile improves the agility of the supply chain. Sourcing cerium salts often involves complex vendor qualification processes and longer lead times due to their specialized nature. By contrast, acetic acid and sodium borohydride can be sourced from multiple Tier-1 chemical suppliers with short lead times and high availability. This flexibility allows procurement teams to negotiate better terms and maintain leaner inventory levels, as the risk of stockouts is minimized. Furthermore, the reduced hazard profile of the reagents simplifies storage and transportation requirements, lowering the logistical overhead and insurance costs associated with handling hazardous materials.
- Scalability and Environmental Compliance: The environmental footprint of the new process is significantly smaller, aligning with modern green chemistry principles and stringent regulatory standards. The absence of heavy metal waste streams simplifies effluent treatment, reducing the burden on wastewater treatment facilities and lowering disposal costs. The process is inherently scalable, as demonstrated by the successful execution of the reaction in standard solvents like THF and methanol without the need for exotic conditions. This scalability ensures that the transition from pilot plant to commercial production is seamless, allowing manufacturers to rapidly ramp up capacity to meet market demand for the final antibiotic drug without encountering the bottlenecks typical of metal-mediated chemistries.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this synthesis route, we have compiled answers to common questions regarding the process parameters and outcomes. These insights are derived directly from the experimental data provided in the patent, ensuring accuracy and relevance for process development scientists. Understanding these details is key to making informed decisions about technology transfer and process validation.
Q: Why is acetic acid preferred over cerium chloride for this reduction?
A: Acetic acid is significantly cheaper and easier to handle than cerium chloride heptahydrate. It eliminates the need for expensive Lewis acids and simplifies the post-treatment process, avoiding complex recycling steps associated with heavy metal salts.
Q: What is the expected purity of the intermediate produced by this method?
A: The patented process consistently yields the intermediate alcohol with a purity greater than 98% after simple vacuum distillation, without the need for column chromatography.
Q: Can this process be scaled for industrial manufacturing?
A: Yes, the method uses common solvents like THF or methanol and operates at mild temperatures (10-25°C), making it highly suitable for large-scale commercial production with minimal safety risks.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Iclaprim Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust and scalable synthetic routes in the fast-paced pharmaceutical industry. Our team of expert chemists has thoroughly analyzed the technology disclosed in CN110498812B and is fully equipped to implement this advanced acetic acid-mediated reduction process. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are designed to handle complex organic syntheses with stringent purity specifications, utilizing rigorous QC labs to verify that every batch of intermediate meets or exceeds the >98% purity benchmark established in the patent literature.
We invite you to collaborate with us to leverage this cost-effective technology for your Iclaprim supply chain. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to discuss your project needs,索取 specific COA data, and review our detailed route feasibility assessments. Let us help you optimize your manufacturing strategy with high-quality intermediates delivered on time and within budget.
