Advanced Synthetic Route for 2-(Difluoromethyl)pyridine-3-alcohol: Commercial Scalability and Process Optimization
Introduction to Novel Fluorinated Pyridine Synthesis
The landscape of organic chemical synthesis is constantly evolving to meet the rigorous demands of the pharmaceutical and agrochemical industries, particularly regarding the incorporation of fluorine atoms to enhance metabolic stability and bioavailability. Patent CN110981793A discloses a groundbreaking synthetic method for 2-(difluoromethyl)pyridine-3-alcohol, a valuable building block that addresses the historical lack of efficient routes for this specific structural motif. This technology represents a significant leap forward in fine chemical manufacturing, offering a streamlined pathway that bypasses the complexities often associated with introducing difluoromethyl groups onto heteroaromatic rings. By leveraging a direct fluorination strategy followed by a robust demethylation protocol, this process ensures high yields and operational simplicity, making it an ideal candidate for industrial scale-up. For R&D directors and procurement specialists alike, understanding the nuances of this patent is crucial for securing a reliable supply chain of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fluorinated pyridine derivatives has been plagued by inefficiencies that hinder commercial viability and increase production costs. Traditional routes often involve multi-step sequences that require harsh reagents, extreme temperatures, or expensive transition metal catalysts which introduce significant impurity profiles. Furthermore, many existing methods struggle with regioselectivity, leading to difficult separations and reduced overall yields that make large-scale production economically unfeasible. The absence of a complete, reported route for 2-(difluoromethyl)pyridine-3-alcohol prior to this invention highlights the technical barriers that have limited access to this critical intermediate. These conventional limitations not only extend lead times for drug development projects but also create supply chain vulnerabilities due to the reliance on niche reagents and complex purification protocols that are difficult to standardize across different manufacturing sites.
The Novel Approach
In stark contrast to these legacy challenges, the novel approach detailed in the patent utilizes a concise two-step strategy that maximizes atom economy and minimizes waste generation. The process begins with the direct conversion of 3-methoxypyridine-2-carbaldehyde using diethylaminosulfur trifluoride (DAST), a reagent known for its efficacy in transforming carbonyl groups into difluoromethylene moieties. This is followed by a straightforward acid-mediated cleavage of the methoxy group to reveal the target phenol functionality. This methodology eliminates the need for protecting group manipulations or exotic catalysts, thereby simplifying the operational workflow significantly. The result is a robust synthetic pathway that offers superior control over reaction parameters, ensuring consistent quality and facilitating the commercial scale-up of complex pharmaceutical intermediates with greater ease and reliability than previously possible.
Mechanistic Insights into DAST-Mediated Fluorination and Acid Hydrolysis
The core of this synthetic innovation lies in the precise mechanistic execution of the fluorination step, where the aldehyde functionality is transformed into a difluoromethyl group. The reaction initiates with the nucleophilic attack of the carbonyl oxygen on the sulfur atom of DAST, forming a reactive intermediate that subsequently undergoes fluoride displacement. This process effectively replaces the carbonyl oxygen with two fluorine atoms, a transformation that is critical for modulating the lipophilicity and metabolic profile of the final molecule. The use of dichloromethane as a solvent provides an optimal medium for this exothermic reaction, allowing for precise temperature control from cryogenic conditions up to moderate heating. Understanding this mechanism is vital for R&D teams aiming to replicate the high yields reported, as slight deviations in stoichiometry or temperature can impact the formation of side products.
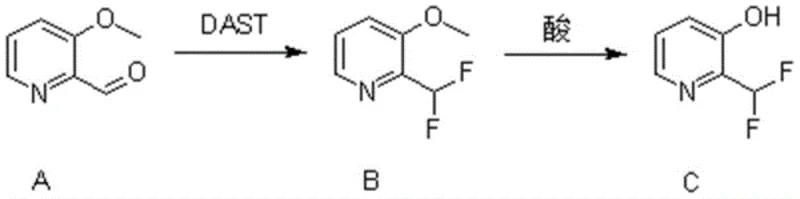
Following the fluorination, the second mechanistic phase involves the cleavage of the aryl methyl ether to generate the free hydroxyl group. This is achieved through the use of a mixed acid system comprising acetic acid and hydrobromic acid, which acts as a potent source of protons and nucleophilic bromide ions. The elevated temperatures facilitate the protonation of the methoxy oxygen, rendering the methyl group susceptible to nucleophilic attack and subsequent departure as a volatile species. This demethylation step is remarkably tolerant of the sensitive difluoromethyl group, demonstrating the chemoselectivity of the process. For quality control purposes, this mechanism ensures that the final product maintains structural integrity without defluorination, a common issue in harsh acidic environments, thus guaranteeing the high purity required for downstream applications in medicinal chemistry.
How to Synthesize 2-(Difluoromethyl)pyridine-3-alcohol Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the optimized conditions outlined in the patent to ensure safety and maximum yield. The process is designed to be scalable, moving seamlessly from gram-scale experiments to multi-kilogram production batches with minimal adjustment. Operators must focus on the controlled addition of reagents, particularly during the exothermic fluorination stage, to maintain thermal stability. The following guide summarizes the critical operational phases derived from the patent examples, serving as a foundational reference for process engineers looking to adopt this technology.
- Dissolve 3-methoxypyridine-2-carbaldehyde in dichloromethane and react with diethylaminosulfur trifluoride (DAST) at controlled temperatures to form the difluoromethyl intermediate.
- Treat the resulting 2-(difluoromethyl)-3-methoxypyridine with a mixed acid system of acetic acid and hydrobromic acid under heating to effect demethylation.
- Isolate the final 2-(difluoromethyl)pyridine-3-alcohol product through alkaline workup, extraction, and purification to achieve high purity specifications.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this synthetic route offers compelling advantages that directly address the pain points of cost containment and supply continuity. By reducing the number of synthetic steps and utilizing commercially available starting materials, manufacturers can significantly lower the cost of goods sold (COGS) while mitigating the risks associated with complex supply chains. The elimination of transition metal catalysts further reduces the burden on downstream processing, as there is no need for expensive metal scavenging resins or extensive testing for heavy metal residues. This streamlined approach translates into faster turnaround times for custom synthesis projects and enhances the overall agility of the supply chain in responding to market demands for fluorinated building blocks.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the reduction in unit operations and the use of cost-effective reagents like DAST and mineral acids. By avoiding multi-step sequences that typically accumulate yield losses and waste disposal costs, the overall production efficiency is drastically improved. Furthermore, the simplified workup procedures reduce solvent consumption and energy usage, contributing to substantial cost savings in utility and waste management sectors. These factors combine to create a highly competitive pricing structure for the final intermediate, making it an attractive option for budget-conscious procurement strategies without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on readily accessible raw materials such as 3-methoxypyridine-2-carbaldehyde ensures a stable supply base that is less susceptible to geopolitical disruptions or raw material shortages. Unlike routes dependent on bespoke or scarce catalysts, this method leverages commodity chemicals that are produced at scale globally. This inherent stability allows for better forecasting and inventory management, reducing the likelihood of stockouts that could delay critical drug development timelines. Consequently, partners can rely on a consistent flow of high-quality intermediates, fostering long-term strategic relationships built on dependability and trust.
- Scalability and Environmental Compliance: The robust nature of the reaction conditions facilitates easy scale-up from laboratory benchtops to industrial reactors, ensuring that production capacity can be expanded rapidly to meet surging demand. Additionally, the process generates fewer hazardous byproducts compared to traditional fluorination methods, aligning with increasingly stringent environmental regulations and sustainability goals. The ability to manage waste streams effectively and minimize the environmental footprint of the synthesis adds significant value for companies committed to green chemistry principles. This compliance advantage not only mitigates regulatory risk but also enhances the corporate social responsibility profile of the supply chain.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this synthesis method, we have compiled answers to common inquiries based on the patent data. These insights are intended to assist technical decision-makers in evaluating the feasibility of integrating this intermediate into their existing pipelines. The responses reflect the specific conditions and outcomes observed in the documented examples, providing a realistic expectation of performance.
Q: What are the critical reaction conditions for the DAST fluorination step?
A: The fluorination step requires precise temperature control, initiating at -78°C and warming to between 5°C and 40°C over a period of 3 to 30 hours, using a molar excess of DAST to ensure complete conversion of the aldehyde group.
Q: How is the methoxy group removed in the second step?
A: Demethylation is achieved using a robust mixed acid system comprising acetic acid and 37% hydrobromic acid in a 1:2 volume ratio, heated to temperatures between 80°C and 115°C for extended periods to cleave the ether bond efficiently.
Q: Why is this synthetic route preferred for commercial manufacturing?
A: This route is preferred due to its brevity (only two steps), the use of readily available starting materials, and the avoidance of complex transition metal catalysts, which simplifies downstream purification and reduces heavy metal contamination risks.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(Difluoromethyl)pyridine-3-alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality fluorinated intermediates play in the development of next-generation therapeutics and agrochemicals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with precision and speed. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify every batch. By partnering with us, you gain access to a supply chain that prioritizes consistency, safety, and technical excellence.
We invite you to contact our technical procurement team to discuss your specific requirements for 2-(difluoromethyl)pyridine-3-alcohol and other fine chemical intermediates. Let us provide you with a Customized Cost-Saving Analysis tailored to your project needs, demonstrating how our optimized processes can enhance your bottom line. Reach out today to request specific COA data and route feasibility assessments, and discover how NINGBO INNO PHARMCHEM can become your trusted partner in chemical innovation.
