Scalable Manufacturing of 2-(Difluoromethyl)pyridine-3-alcohol via Novel Fluorination Route
The landscape of organofluorine chemistry continues to evolve, driven by the critical demand for fluorinated heterocycles in modern drug discovery and agrochemical development. Patent CN110981793B introduces a groundbreaking synthetic methodology for producing 2-(difluoromethyl)pyridine-3-alcohol, a valuable building block characterized by its unique lipophilicity and metabolic stability profiles. This specific compound addresses a significant gap in the literature, as prior to this disclosure, no complete and efficient route had been reported for its preparation. The introduction of the difluoromethyl group onto the pyridine scaffold is particularly strategic, as it serves as a bioisostere for hydroxyl or methoxy groups while enhancing membrane permeability and binding affinity in biological systems. By establishing a reliable pathway to this molecule, the patent provides a foundational technology for the creation of next-generation therapeutics and crop protection agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fluorinated pyridine derivatives has been fraught with challenges, often requiring harsh conditions, expensive reagents, or convoluted multi-step sequences that degrade overall yield. Traditional approaches to installing fluorine atoms frequently involve electrophilic fluorination agents that lack selectivity or nucleophilic sources that require activated substrates, leading to complex impurity profiles that are difficult to purge. Furthermore, the specific functionalization of the 2-position of a pyridine ring with a difluoromethyl group while maintaining a hydroxyl group at the 3-position presents significant regioselectivity issues. Existing methods often suffer from poor atom economy and generate substantial hazardous waste, making them economically unviable for large-scale commercial manufacturing. The lack of a direct route previously forced process chemists to rely on inefficient workaround strategies that increased lead times and production costs significantly.
The Novel Approach
In stark contrast to these historical limitations, the methodology disclosed in the patent offers a streamlined, two-step sequence that maximizes efficiency and minimizes operational complexity. The strategy leverages the high reactivity of diethylaminosulfur trifluoride (DAST) to convert an aldehyde precursor directly into the desired difluoromethyl moiety under controlled conditions. This is followed by a straightforward acidic demethylation to reveal the phenolic hydroxyl group, completing the synthesis in just two distinct chemical transformations. This approach not only reduces the number of unit operations but also utilizes commercially available starting materials, thereby simplifying the supply chain logistics. The robustness of this route allows for precise control over reaction parameters, ensuring consistent quality and high purity of the final product, which is essential for regulatory compliance in the pharmaceutical industry.
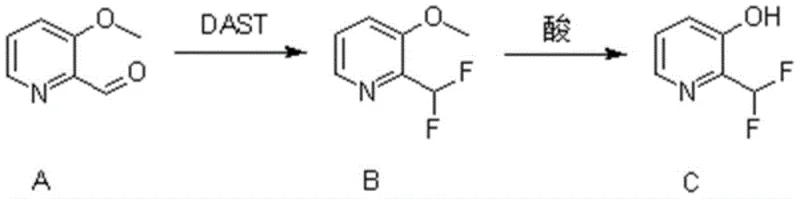
Mechanistic Insights into DAST-Mediated Fluorination and Acidic Demethylation
The core of this synthetic innovation lies in the mechanistic precision of the DAST-mediated fluorination step, which transforms the carbonyl functionality of 3-methoxypyridine-2-carbaldehyde into a difluoromethyl group. The reaction initiates at cryogenic temperatures, typically around -78°C, to manage the exothermic nature of the interaction between the aldehyde and the sulfur-fluorine reagent. As the temperature is gradually raised to a range of 5-40°C, the oxygen of the carbonyl group attacks the sulfur atom of DAST, forming an unstable intermediate that subsequently undergoes fluoride displacement. This nucleophilic substitution effectively replaces the carbonyl oxygen with two fluorine atoms, a transformation that is thermodynamically driven by the formation of strong sulfur-oxygen bonds. The careful modulation of temperature and stoichiometry, specifically using a 1:3 molar ratio of substrate to reagent, ensures complete conversion while suppressing potential side reactions such as elimination or rearrangement.
Following the fluorination, the second critical mechanistic phase involves the acid-catalyzed cleavage of the methyl ether to generate the free phenol. This step utilizes a mixed acid system comprising acetic acid and hydrobromic acid, heated to temperatures between 80-115°C. The protonation of the methoxy oxygen by the strong acid renders the methyl group susceptible to nucleophilic attack by bromide ions or water, leading to the scission of the carbon-oxygen bond. This demethylation process is highly effective for pyridine systems, where the electron-withdrawing nature of the ring can sometimes deactivate the ether towards cleavage. The use of hydrobromic acid is particularly advantageous due to the high nucleophilicity of the bromide ion, which facilitates the SN2-type displacement of the methyl group. This rigorous acidic treatment ensures that the final product, 2-(difluoromethyl)pyridine-3-alcohol, is obtained with minimal residual starting material or partially demethylated impurities.
How to Synthesize 2-(Difluoromethyl)pyridine-3-alcohol Efficiently
To achieve optimal results in the laboratory or pilot plant, strict adherence to the patented protocol is essential, particularly regarding temperature gradients and reagent addition rates. The process begins with the dissolution of the aldehyde precursor in an anhydrous solvent like dichloromethane, followed by the controlled addition of DAST under an inert atmosphere to prevent moisture interference. Detailed standardized operating procedures for scaling this reaction, including specific quenching and purification techniques, are critical for maintaining safety and yield consistency. For a comprehensive breakdown of the exact experimental conditions and workup procedures, please refer to the step-by-step guide below.
- Dissolve 3-methoxypyridine-2-carbaldehyde in dichloromethane and react with diethylaminosulfur trifluoride (DAST) under nitrogen protection at controlled temperatures to form the difluoromethyl intermediate.
- Subject the resulting 2-(difluoromethyl)-3-methoxypyridine to acidic hydrolysis using a mixture of acetic acid and hydrobromic acid at elevated temperatures.
- Neutralize the reaction mixture, extract with ethyl acetate, and purify via column chromatography to isolate the final 2-(difluoromethyl)pyridine-3-alcohol product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers profound benefits for procurement managers and supply chain directors looking to optimize their sourcing strategies for fluorinated intermediates. The reduction of the synthesis to merely two steps fundamentally alters the cost structure of the molecule by drastically cutting down on labor, energy consumption, and solvent usage associated with intermediate isolations. By eliminating the need for complex transition metal catalysts or exotic reagents, the process relies on commodity chemicals that are readily available in the global market, thereby reducing supply chain vulnerability. This simplicity translates directly into a more resilient supply chain, as the risk of bottlenecks caused by specialized reagent shortages is significantly minimized. Furthermore, the high yields reported in the patent examples suggest that material throughput will be maximized, allowing manufacturers to meet large-volume demands without excessive raw material overhead.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by its exceptional step economy, which inherently lowers the cost of goods sold (COGS) by reducing the number of processing stages. By avoiding the use of precious metal catalysts, the process eliminates the costly and time-consuming steps associated with metal scavenging and residual metal testing, which are mandatory for pharmaceutical grade materials. The use of standard solvents like dichloromethane and ethyl acetate further ensures that solvent recovery and recycling can be implemented efficiently, contributing to substantial operational savings. Additionally, the high conversion rates observed mean that less raw material is wasted, optimizing the overall material balance and reducing disposal costs for chemical waste.
- Enhanced Supply Chain Reliability: The reliance on 3-methoxypyridine-2-carbaldehyde as a starting material provides a significant logistical advantage, as this compound is structurally simple and can be sourced from multiple established vendors globally. The robustness of the reaction conditions, which do not require ultra-high pressures or cryogenic maintenance beyond the initial addition phase, allows for production in a wider range of manufacturing facilities, diversifying the potential supplier base. This flexibility ensures continuity of supply even in the face of regional disruptions or capacity constraints at specific sites. Moreover, the stability of the intermediates allows for potential campaign manufacturing, where the difluoromethyl intermediate can be stockpiled if necessary, adding another layer of security to the supply chain.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory experiments to multi-kilogram or ton-scale production without requiring fundamental changes to the reaction engineering. The waste profile is relatively benign compared to alternative fluorination methods, as the primary byproducts are sulfur-containing species that can be managed through standard neutralization and disposal protocols. The absence of heavy metals simplifies environmental compliance and reduces the burden on wastewater treatment facilities. This alignment with green chemistry principles not only mitigates regulatory risk but also enhances the sustainability profile of the final product, a factor increasingly valued by downstream customers in the pharmaceutical and agrochemical sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this fluorinated pyridine derivative. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation to ensure accuracy and relevance for industry professionals. Understanding these details is crucial for evaluating the feasibility of integrating this intermediate into your specific development pipeline.
Q: What are the key advantages of this synthetic route for 2-(difluoromethyl)pyridine-3-alcohol?
A: The primary advantage is the brevity and efficiency of the two-step process, which avoids complex multi-step sequences often required for introducing difluoromethyl groups onto heterocyclic rings. Additionally, the use of readily available starting materials like 3-methoxypyridine-2-carbaldehyde simplifies procurement.
Q: How does the process ensure high purity for pharmaceutical applications?
A: The process utilizes specific temperature controls during the DAST fluorination step (-78°C initiation) to minimize side reactions, followed by a robust acidic workup that effectively removes methoxy protecting groups, resulting in a clean product profile suitable for downstream API synthesis.
Q: Is this method suitable for large-scale commercial production?
A: Yes, the methodology is designed for scalability, utilizing standard organic solvents like dichloromethane and common industrial acids. The operational simplicity and high yields reported in the patent examples indicate strong potential for kilogram-to-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(Difluoromethyl)pyridine-3-alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of high-quality fluorinated intermediates in accelerating drug discovery timelines. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and reliability. We are committed to delivering materials that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to replicate and optimize the patented route described above allows us to offer a consistent and dependable supply of 2-(difluoromethyl)pyridine-3-alcohol to support your R&D and manufacturing goals.
We invite you to contact our technical procurement team to discuss your specific requirements and explore how we can support your project with a Customized Cost-Saving Analysis. Whether you need small quantities for initial screening or bulk volumes for clinical trials, we are prepared to provide specific COA data and route feasibility assessments tailored to your timeline. Partner with us to leverage our technical expertise and secure a stable supply chain for your critical fluorinated building blocks.
